DIAGNOSIS
AND TREATMENT OF PROSTATIC ABSCESS
(
Download pdf )
PAULO OLIVEIRA, JUAREZ A. ANDRADE, HELDER C. PORTO, JOSÉ E. PEREIRA FILHO, ANTÔNIO F. J. VINHAES
Section of Urology, São Rafael Hospital, Salvador, Bahia, Brazil
ABSTRACT
Objectives:
Present and discuss the pathogenesis, diagnostic methods and treatment
of the prostatic abscess.
Materials and methods: We have retrospectively
studied the medical records of 9 patients diagnosed and treated for prostatic
abscess, between March 1998 and December 2000, assessing age, context,
associated diseases, and diagnostic and therapeutic methods. We have compared
the data found with those described in literature, based on Medline data.
Results: Mean age was 52.6 years. Three
patients had previous diabetes mellitus diagnosis, and one was infected
by HIV virus. Transrectal ultrasound of the prostate confirmed the diagnosis
of prostatic abscess in all 7 cases in which it was performed. All cases
received antibiotic treatment, and 77.8% needed concomitant surgical treatment.
Two cases of microabscess were treated only with antibiotics. Four patients
were submitted to perineal catheter drainage, 2 were submitted to transurethral
resection of the prostate (TURP), and one patient required both procedures.
Mean hospitalization time was 11.2 days, and most frequent bacterial agent
was S. aureus. All patients were discharged from the hospital, and there
was no death in this series.
Conclusions: Prostatic abscess should be
treated with broad-spectrum antibiotics and surgical drainage (perineal
puncture or TURP). Microabscess may heal without surgery.
Key words:
prostate; infection; abscess; diagnosis; therapeutics
Int Braz J Urol. 2003; 29: 30-4
INTRODUCTION
The
prostatic abscess is difficult to diagnose, because at the symptoms onset
it may mimic several other diseases of the lower urinary tract. It is
uncommon, and rarely diagnosed, and it has suffered a great shift in its
mortality rate, and in the types of etiologic agents observed since the
discovery and use of penicillin (1). In the forties, mortality ranged
from 6% to 30%, and major microorganism involved was Neisseria gonorrhea.
More recent data suggests a mortality rate from 3% to 16% (2), enterobacteria
being the most common agents. Among these, Escherichia coli has the highest
prevalence, in about 70% of the cases (3).
In this paper we present data about 9 patients
diagnosed with prostatic abscess, discussing clinical findings, diagnostic
criteria, and treatment results.
MATERIALS AND METHODS
We
have retrospectively studied a series of 9 patients admitted to, and treated
in, our facility, between March 1998 and December 2000, with diagnosis
of prostatic abscess. The data was collected in the medical records. Literature
review was based on Medline data.
Age ranged from 37 to 73 years (mean 52.7).
Four patients had risk factors. One was an intravenous drug user with
7 years asymptomatic HIV infection and the prostatic abscess was concomitant
to a perirenal abscess. Three had diabetes, and one presented the prostatic
abscess after a prostatic biopsy. All were treated with parenteral antibiotics.
Surgical treatment was indicated in cases where there was no clinical
improvement with antibiotic therapy, and after confirming the diagnosis
of prostatic abscess. The criteria of hospital discharge were absence
of fever for at least 48 hours, and leucogram normalization.
RESULTS
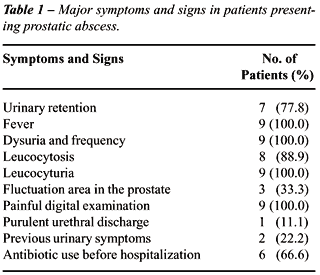
Symptoms
consistent with prostatitis initiated the clinical context, and 7 patients
(those with the largest abscess) progressed with urinary retention. Two
patients presented previously lower urinary tract symptoms, but there
was a worsening of the symptoms, suggesting acute prostatitis. Besides
4 patients with previous diseases altering the immune system (case 1 -
HIV infection and cases 4, 6 and 7 - diabetes), 2 also presented possible
primary focuses of bacterial haematogenous dissemination (case 1 presented
concomitant perirenal abscess with the same etiologic agent, and case
4 had a cutaneous injury 15 days before the symptoms’ onset). One
patient had history of urinary tract manipulation (transrectal ultrasound-guided
biopsy of the prostate) and diabetes (case 6).
In 7 patients (77,8%), the diagnosis was
confirmed by transrectal ultrasound (Figure-1). One of the patients also
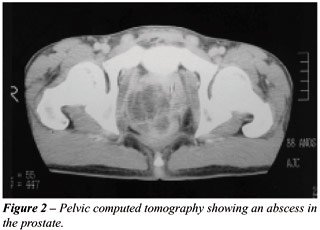
performed a computed tomography that confirmed the presence of the abscess
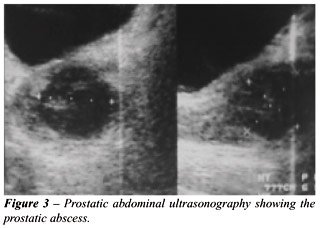
(Figure-2). For 2 patients, the diagnosis was performed by abdominal ultrasound
while still in the emergency unit (Figure-3). One patient did not perform
ultrasonography and the diagnosis was done during an adenomectomy surgery.

All patients were treated with parenteral
antibiotics during the hospital stay, with ciprofloxacin 400 mg IV bid,
in some cases associated to amikacin 500 mg IV bid. Two patients also
received metronidazole 500 mg IV qid. In 2 cases it was necessary to alter
the antibiotic regimen to ceftriaxone due to absence of clinical improvement.
Seven patients (77,8%) required adjuvant surgical treatment, and 2 were
submitted to transurethral resection of the prostate (TURP), 4 to perineal
pucture/drainage of the prostate, maintaining a silicone catheter for
drainage, and 1 performed both procedures due to the periprostatic extension
of the abscess. Two patients had extensions of the abscess to the space
between the prostate and the rectum (Figure-4). The patient presenting
concomitant perirenal abscess had the perirenal space drained by computed
tomography-guided percutaneous route, with good outcome.

Two patients (22,2%) presented microabscess
and were treated exclusively with antibiotics, showing good results.
All patients had good outcomes, with no
occurrence of sepsis or deaths in this series. Mean hospital stay was
11.2 days, and patients were discharged with oral antibiotics prescribed
until totaling at least 21 treatment days, if not presenting residual
abscess in clinical examination or control transrectal ultrasound.
In 7 patients surgically treated, there
was Staphylococcus aureus growing in the material collected from the abscess
in 4 (57,1%) cases, Escherichia coli in one case, Aeromonas aerophyla
in one case, and Klebsiella pneumoniae in one case.
Major clinical data is presented in Tables-1
and 2.

DISCUSSION
When
not adequately treated, the prostatic abscess may progress to sepsis and
death. Thus, an accurate diagnostic and an efficient treatment are both
required. Most published data about prostatic abscess are case reports,
and there is no standardization of the diagnostic and therapeutic routine.
In review articles, the summary of several individual experiences permits
delineating some lines of action in cases of prostatic abscess (1,2).
Various factors have influenced the shift
of the epidemiological profile of prostatic abscess, such as routine and
widespread use of broad-spectrum antibiotics to patients with lower urinary
tract symptoms, without the investigation required (4); better control
of chronic diseases allowing an increase in population longevity; therapeutic
advances such as hemodialysis, organ transplants, chemotherapy, and immunosuppressive
drugs, promoting longer survival, but also exposing to the risks of immunosuppression
(4-7).
Finding spontaneous abscess drainage to
the urethra (4), and peritonitis (8), is sporadic today. It is thought
that the retrograde flow of contaminated urine within the prostate during
micturition is the most prevalent pathogenic factor (9). Some authors
suggest that prostatic abscess is a complication of bacterial prostatitis,
acute or chronic, but the actual incidence and frequency of these events
is not known (10). Bacterial haematogenous spread from distant foci was
also described, such as from respiratory (bronchitis, otitis), digestive
(appendicitis, diverticulitis), and urinary tracts (perirenal abscess),
and from the skin (furuncles, abrasions). In these cases, germs like S.
aureus, M. tuberculosis, E. coli e Candida sp. may be found.
In this series, patients presented a mean
age comparable to that found in literature, where the most common age
group is between the fifth and the sixth decades. However, cases in diverse
age groups were described, including neonates (2). A higher prevalence
of S. aureus was observed, opposed to the findings in literature indicating
E. coli as the most prevalent bacteria (1). Only for 2 patients we have
found a clear explanation for this fact, with the primary foci in the
skin (injections drug use and cutaneous abscess).
Initially the disease manifests as dysuria,
urgence, and frequency in 96% of the cases (1), fever in 30% to 72% (1,2,7),
and urinary retention in 1/3 of the patients (2,7). We have observed a
higher frequency of fever, urinary retention, dysuria and frequency in
all patients (Table-1). There are reports of cases disclosed only at necropsy
in children, and of 2 cases that did not present any symptom in a series
of 269 cases (2). The most typical sign of prostatic abscess is fluctuation
areas in the prostate by digital examination, although the results diverge
between 16% (2) and 88% (1). This finding was observed in 33.3% of our
patients. However, all presented painful prostate at digital exam, and
leucocytosis and leucocyturia as well.
The diagnostic study of choice to assist
the treatment and follow-up of patients with prostatic abscess is transrectal
ultrasonography of the prostate. The most common finding is presence of
one or more hypoechogenic areas, of several sizes, containing thick liquid
primarily in the transition zone and in central zone of the prostate,
permeated by hyperechogenic areas and distortion of the anatomy of the
gland (7). In this series this finding was observed in 100% of the cases
for which the examination was performed. Differential diagnosis should
include prostatic cysts and neoplasia (10,11). Computed tomography adds
few benefits to transrectal ultrasonography for the diagnosis of prostatic
abscess, especially when there are extraprostatic collections (12,13).
Treatment implies in parenteral broad-spectrum
antibiotic administration and abscess drainage. This may be performed
by transrectal puncture (14) or transperineal ultrasound-guided, digital-guided
puncture/drainage by perineal route, transurethral incision of the prostate,
TURP, or open perineal drainage (15-18). There is a preference for minimally
invasive procedures that may be performed under local anesthesia or sedation,
and repeated if necessary. All methods have safety and efficiency reports
(1-3,5,9,15-18).
Sending material to culture (pus, urine,
blood, and/or a fragment of the prostate) is important in identifying
the etiologic agent, especially for immunosuppressed patients, considering
that they usually present uncommon germs (19,20).
Lack of uniformity in antibiotics prescription
occurs due to rareness of the disease, and there is no routine established
for these cases. Most of the times the antibiotic was introduced by the
on-call doctor in the emergency room.
Hospital stay period was prolonged, and
most patients needed surgical treatment (77,8%), showing that this disease
deserves hospital care. The diagnosis of prostatic abscess should be proposed
for patients presenting fever and persistent irritative voiding symptoms
despite antimicrobials use, for diabetics with protracted symptoms, for
those with lower urinary tract symptoms and fever progressing to urinary
retention, and after the performance of prostatic biopsy.
REFERENCES
- Weinberger M, Cytron S, Servadio C, Block C, Rosenfeld JB, Pitlik SD: Prostatic abscess in the antibiotics era. Rev Infect Dis. 1988; 10: 239-49.
- Granados EA, Caffaratti J, Farina L, Hocsman H: Prostatic abscess drainage: Clinical-sonography correlation. Urol Int. 1992; 48: 358-61.
- Meares EM, Jr.: Prostatic abscess. J Urol. 1996; 129: 1281-2.
- Gill SK, Gilson RJC, Rickards, D: Multiple prostatic abscesses presenting with urethral discharge. Genitourin Med. 1991; 67: 411-2.
- Barozzi L, Pavlica P, Menchi I, De Matteis M, Canepari M: Prostatic abscess: diagnosis and treatment. AJR. 1998; 170: 753-7.
- Cytron S, Weinberger M, Pitlik S, Servadio, C: Value of transrectal ultrasonography for diagnosis and treatment of prostatic abscess. Urology 1988; 32: 454-8.
- Granados EA, Riley G, Salvador J, Vicente J: Prostatic abscess: Diagnosis and treatment. J Urol. 1992; 148: 80-2.
- Mitchell RJ, Blake RJS: Spontaneous perforation of prostatic abscess with peritonitis. J Urol. 1972; 107: 622-3.
- Trauzzi SJ, Kay CJ, Kaufman DG, Lowe FC: Management of prostatic abscess in patients with human immunodeficience syndrome. Urology 1994; 43: 629-33.
- Jameson RM: Prostatic abscess and carcinoma of the prostate. Br J Urol. 1968; 40: 288-92.
- Rifkin MD: Ultrasonography of the lower genitourinary tract. Urol Clin North Am. 1985; 12: 645-56.
- Thornill BA, Morehouse HT, Coleman P, Tretin JCH: Prostatic abscess: CT and sonographic findings. AJR. 1987; 148: 899-900.
- Vaccaro JA, Belville WD, Kiesling Jr. VJ, Davis R: Prostatic abscess: Computerized tomography scanning as an aid to diagnosis and treatment. J Urol. 1986; 136: 1318-9.
- Chaabouni MN, Pfeifer P, Ferrandis P, Chokairi P, D’Ardalhon T, Dumas JP et al.: Place de ponction transrectale écho-guidée dans le traitement des abcès prostatiques. Ann Urol. 1994; 28: 24-7.
- Lopez VM, Castro VF, Pallas MP, Garcia JA, Gonzalez PC: Drenaje transperineal de un absceso prostático. Arch Esp de Urol. 1994; 47: 290-1.
- Kinahan TJ, Goldenberg SL, Ajzen AS, Cooperberg PL, English, RA: Transurethral resection of prostatic abscess under sonographic guidance. Urology. 1991; 37: 475-7.
- Bachor R, Gottfried HW, Hautmann, R: Minimal invasive therapy of prostatic abscess by transrectal ultrasound-guided perineal drainage. Eur Urol. 1995; 28: 320-4.
- Llanes JV, Carbonell CL, Toro AOA, Mas AG: Abscesos prostáticos: tratamiento percutáneo. Arch Esp de Urol. 1991; 44: 69-72.
- Sanjuan FG, Cidre MJ, Rodríguez RR, Elias MJP, Santos VG, Escudero JAR: Absceso prostático tuberculoso en síndrome de inmunodeficiencia adquirida. Arch Esp de Urol. 1997; 50: 393-5.
- Learmonth DJ, Philp NH: Salmonella prostatic abscess. Br J Urol. 1988; 61: 163.
_____________________
Received: August 7, 2002
Accepted after revision: January 9, 2003
_______________________
Correspondence address:
Dr. Paulo Oliveira
Serviço de Urologia, Hospital São Rafael
Av. São Rafael, 2152 / 6o. Andar / Sala 9
Salvador, BA, 41256-900, Brazil
Fax: + 55 71 399-6513
E-mail: paulourologia@hotmail.com