ASSESSMENT
OF LYMPHOCELE INCIDENCE FOLLOWING 450 RENAL TRANSPLANTATIONS
(
Download pdf )
VICTOR T. DUBEAUX, RENÉ M. OLIVEIRA, VICTOR J. MOURA, J. MONTEIRO S. PEREIRA, FABIO P. HENRIQUES
Section of Urology, Clementino Fraga Filho University Hospital, Federal University of Rio de Janeiro, Rio de Janeiro, RJ, Brazil
ABSTRACT
Objective:
To determine the incidence of lymphocele in the follow-up of patients
who underwent renal transplantation, as well as potential factors responsible
or associated to its development.
Materials and Methods: All records from
patients who were treated for lymphocele in our institution between May
1989 and December 2002 were reviewed, as well as their clinical outcome
following treatment.
Results: Among 450 patients who underwent
renal transplantation in the period, only 3 required treatment, with 2
of them treated due to the collection volume, and the other due to symptoms
(pain), representing an incidence of only 0.6%.
Comments: The occurrence of perirenal fluid
collections following renal transplantation is frequent. In cases where
treatment is required, this can generate an excessive morbidity for the
patient, which motivates the development of preventive methods, such as
minimally invasive therapy, for such cases.
Conclusion: Careful ligation of lymphatic
vessels both during graft preparation and during its implantation, added
to post-operative drainage can significantly contribute to reducing the
incidence of lymphocele following renal transplantation.
Key words: renal transplantation; lymphocele; prevalence;
predisposing factors; surgical procedures, operative
Int Braz J Urol. 2004; 30: 18-21
INTRODUCTION
Lymphoceles
are the fluid collections most frequently found following renal transplantation,
occurring in up to 22% of transplantations performed in the United States
every year (1). The risk of developing a lymphocele is associated to previous
episode of acute rejection, incomplete ligation of lymphatic vessels in
the graft, high doses of steroids and extensive perivascular dissection
in the recipient, among other causes (1).
The majority of cases develops during the
first year following surgery, and is asymptomatic, with no need of treatment
(2). However, symptomatic cases can evolve with increase in creatinine
levels and a fall in the urinary output due to the compression exerted
over the implanted kidney or ureter. It can manifest as a palpable mass,
with abdominal pain, or edema in lower limbs due to the compression of
the iliac vessels (3).
Therapeutic options include percutaneous
drainage, aspiration followed by infusion of a sclerosing agent, or internal
(peritoneal) drainage, by both laparoscopic and open surgery (4,5).
The objective of this work was to determine
the incidence of lymphocele in the follow-up of patients who underwent
renal transplantation, as well as potential factors that could be responsible
or associated to its development.
MATERIALS AND METHODS
After
reviewing the records of patients who underwent surgical intervention
for treatment of lymphocele between May 1989 and December 2002, we investigated
its incidence, and the treatment performed.
Patients’ age ranged from 27 to 45
years, with all of them being Caucasian and male.
Patients received an immunosuppressive regimen
that consisted of cyclosporine, fk 506 (tacrolimus), corticotherapy, mycofenolate
mofetil and rapamicine, at the discretion of our nephrologic team.
RESULTS
Among
the 450 patients who were transplanted until then, only 3 (0.6%) presented
a clinical picture of lymphocele requiring intervention post-operatively.
One case was symptomatic and the other 2 were incidentally diagnosed through
ultrasonography scans.
One patient who presented a symptomatic
lymphocele measuring 20 x 15 cm on the ultrasound underwent intervention,
as well as the other 2 who had voluminous collections, even if asymptomatic,
due to the volume detected by ultrasonography (10 x 8 cm and 15 x 12 cm),
and the suspected possibility of occurrence of subsequent infection.
Resolution was achieved by percutaneous
drainage in 2 cases. The third patient presented recurrence following
drainage and underwent surgical treatment with drainage of the collection
to the peritoneum by conventional approach, and did not present recurrence
in the post-operative period.
The collected material was submitted to
biochemical and microbiological analysis, revealing to be sterile and
presenting biochemical characteristics of lymph.
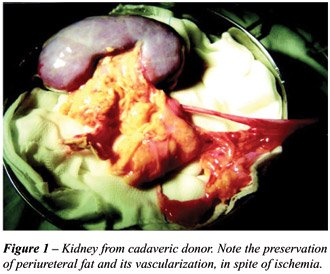
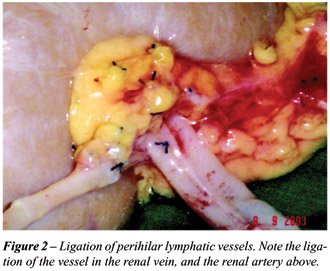
All patients were recipients of kidneys
from cadaveric donors, and had their implants prepared by the same team,
with rigorous ligation of lymphatic vessels, including the ureteral ones
(Figures-1 and 2). The implantation was performed in the right iliac fossa
in 2 cases and in the left fossa in 1 case, followed by open drainage
of the cavity for 48 to 72 hours, according to the output present post-operatively.
The ureteroneocystostomy was performed using the extravesical technique
(Lich-Gregoire) in all cases, without placement of double-J stent.
The arterial anastomoses were performed
in the internal iliac artery (termino-terminal) and common iliac vein
(termino-lateral).
Patients were follow-up for an average of
6 months (between 4 and 8 months) without recurrence and with no need
of further treatment.
DISCUSSION
The
development of lymphoceles following renal transplantation is a reality
that urologists who develop such activity must live with. The use of ultrasonography
increased the index of detection for these fluid collections, though the
majority of them have small size, and resolved spontaneously (2). While
the etiopathogeny of its formation remains unclear, many risk factors
for its development have already been postulated. It is believe that extensive
perivascular dissection along the route of iliac vessels, episodes of
acute rejection, cadaveric versus live donors, high doses of corticotherapy,
re-transplantation, and polycystic disease in the recipients, are factors
that contribute to the development of fluid collections. Nevertheless,
little has been done to prevent the occurrence of lymphocele, besides
preventing acute rejection (1,5).
We believe that the low incidence of lymphoceles
in this patient series results from the careful ligation of lymphatic
vessels in the graft, extending from the renal hilum and along the ureter,
which is not dissected, thus preventing a decrease in its vascularization.
Moreover, the recipient’s lymphatic vessels are also carefully ligated
along the area where the vascular anastomoses will be subsequently performed.
Additionally, open drainage of the graft
cavity, routinely performed in the post-operative period, until a decreased
in output, can also account for the low indexes of collection detected
in the late post-operative period, since the transudation is very high
during the first 24 to 48 h following surgery. Though many works report
such measure to be unnecessary, since the lymphatic dissection and ligation
are carefully performed, we believe that such procedure adds low morbidity
and contributes for the low rates of lymphocele.
An interesting fact detected in this review
was that all cases occurred after 1996, when the immunosuppressive regimens
were practically the same used nowadays, and our team had already been
trained for performing such procedure.
CONCLUSION
Lymphoceles are common complications following renal transplantation, and can lead to an increase in morbidity for this procedure. The use of a standardized dissection technique with rigorous ligation of the lymphatic vessels in the graft and drainage of the cavity can prevent or decrease its occurrence, regardless of the presence of predisposing factors to its formation. However, prospective randomized studies are required to establish the real need of this meticulous procedure during the organ preparation and the dissection in the surgical field.
REFERENCES
- Khauli RB, Stoff JS, Lovewell T, Ghavamian R, Baker S: Post transplant lymphoceles: a critical look into the risk factors, pathophysiology and management. J Urol. 1993; 150: 22-6.
- Pollak R, Veremis SA, Maddux MS, Mozes MF: The natural history of and therapy for perirenal fluid collections following renal transplantation. J Urol. 1988; 140: 716-20.
- Chin A, Ragavendra N, Hilborne L, Gritsch HA: Fibrin sealant sclerotherapy for treatment of lymphoceles following renal transplantation. J Urol. 2003; 170: 380-3.
- Shaver TR, Swanson SJ 3rd, Fernandez-Bueno C, Kocandrle V: The optimal treatment of lymphoceles following renal transplantation. Transpl Int. 1993; 6: 108-10.
- Fahlenkamp D, Raatz D, Schonberger B, Loening SA: Laparoscopic lymphocele drainage after renal transplantation. J Urol. 1993; 150: 316-8.
- Sansalone CV, Aseni P, Minetti E, Di Benedetto F, Rosseti O, Manoochehri F, et al.: Is lymphocele in renal transplantation an avoidable complication? Am J Surg. 2000; 179: 182-5.
- Huilgol AK, Sundar S, Karunagaran SG, Sudhakar S, Sreenivasa Pr MA, Ravindran T: Lymphoceles and their management in renal transplantation. Transplant Proc. 2003; 35: 323.
- Chandrasekaran D, Meyyappan RM, Rajaraman T: Instillation of povidone to treat and prevent lymphocele after renal transplantation. BJU Int. 2003; 91: 296.
- Fuller TF, Kang SM, Hirose R, Feng S, Stock PG, Freise CE: Management of lymphoceles after renal transplantation: laparoscopic versus open drainage. J Urol. 2003; 169: 2022-5.
________________________
Received: October 20, 2003
Accepted after revision: February 16, 2004
_______________________
Correspondence address:
Dr. Victor T. Dubeaux
Est da Gávea, 611, bl. 1, 403
Rio de Janeiro, RJ, 22610-000, Brazil
E-mail: vdubeaux@bol.com.br