SARCOMATOID
DIFFERENTIATION IN RENAL CELL CARCINOMA: PROGNOSTIC IMPLICATIONS
(
Download pdf )
MARCOS F. DALL’OGLIO, MARCO LIEBERKNECHT, VALTER GOUVEIA, ALEXANDRE C. SANT’ANNA, KATIA R. LEITE, MIGUEL SROUGI
Division
of Urology, Paulista School of Medicine, Federal University of Sao Paulo,
UNIFESP,
Sao Paulo, SP, Brazil
ABSTRACT
Introduction:
Renal cell carcinoma with sarcomatoid differentiation is a tumor with
aggressive behavior that is poorly responsive to immunotherapy. The objective
of this study is to report our experience in the treatment of 15 patients
with this tumor.
Materials and Methods: We retrospectively
analyzed 15 consecutive cases of renal cell carcinoma with sarcomatoid
differentiation diagnosed between 1991 and 2003. The clinical presentation
and the pathological stage were assessed, as were the tumor’s pathological
features, use of adjuvant immunotherapy and survival. The study’s
primary end-point was to assess survival of these individuals.
Results: The sample included 8 women and
7 men with mean age of 63 years (44 - 80); follow-up ranged from 1 to
100 months (mean 34). Upon presentation, 87% were symptomatic and 4 individuals
had metastatic disease. Mean tumor size was 9.5 cm (4 - 24) with the following
pathological stages: 7% pT1, 7% pT2, 33% pT3, and 53% pT4. The pathological
features showed high-grade tumors with tumoral necrosis in 87% of the
lesions and 80% of intratumoral microvascular invasion. Disease-free and
cancer-specific survival rates were 40 and 46% respectively, with 2 cases
responding to adjuvant immunotherapy.
Conclusions: Patients with sarcomatoid tumors
of the kidney have a low life expectancy, and sometimes surgical resection
associated with immunotherapy can lead to a long-lasting therapeutic response.
Key
words: kidney neoplasms; carcinoma; renal cell; neoplasms metastasis
Int Braz J Urol. 2005; 31: 10-16
INTRODUCTION
Every
year, renal cell carcinoma (RCC) is responsible for approximately 2% of
new cases of cancer in the United States and for the highest proportion
of cancer-associated deaths in relation to all other malignant urological
diseases (1).
Initially called carcinosarcoma (2), mixed
renal tumor (3), and subsequently renal sarcomatoid tumor according to
Farrow et al. (4), the sarcomatoid differentiation is not a distinct histological
entity and confers higher aggressiveness on any of the different subtypes
of RCC, with a frequency ranging from 1 to 8% in the series (4-6). Studies
indicate that the presence of a sarcomatoid component makes the disease
locally aggressive, which typically presents an advanced grade that is
associated with fast progression and fatal outcome in a vast proportion
of cases, with median survival lower than 1 year in many series (5-11).
This is important for predicting the outcome for patients undergoing nephrectomy
due to RCC, since adjuvant therapy in a certain group of patients with
higher risk of progressive disease can be a reasonable alternative.
We report our experience with sarcomatoid
RCC after assessing its presentation form, the role of the pathology,
the tumor’s pathological features, the role of adjuvant immunotherapy
and the patient survival rate.
MATERIALS AND METHODS
We
retrospectively assessed 231 patients diagnosed with RCC who underwent
surgical treatment at our institution from March 1991 to June 2003. The
study included 15 patients aged from 44 to 80 years old (mean 63 years)
whose pathological examination of the surgical specimen revealed a sarcomatoid
component.
For pathological analysis, the specimen
is fixed in formalin, embedded in paraffin, sectioned and fixed as usual
by hematoxylin-eosin (HE). In addition to sarcomatoid differentiation
characterized by elongation of the tumor cells (Figure-1), we analyzed
the nuclear grade, pleomorphism, mitotic index and tumoral necrosis. The
immunohistochemical analysis was performed in 8 patients with exclusive
sarcomatoid differentiation, confirming the presence of an epithelial
component.

Age, gender and presentation symptoms were
assessed, as well as the stage of the disease, the pathological features,
the use of adjuvant immunotherapy, the time to recurrence of the disease
and the survival of this group of individuals. Postoperative follow-up
was bi-annual through hematological and imaging exams with ultrasound
interpolated with tomography and chest radiography.
The statistical analysis through survival
curves was performed by the Kaplan-Meier method, with the log Rank test
being used for comparison.
RESULTS
Of
the 15 patients under study, the mean age was 63 years (44 - 80), comprising
8 women and 7 men. Upon presentation, 87% were symptomatic, with pain,
weight loss and hematuria being the most frequent complaints. Additionally,
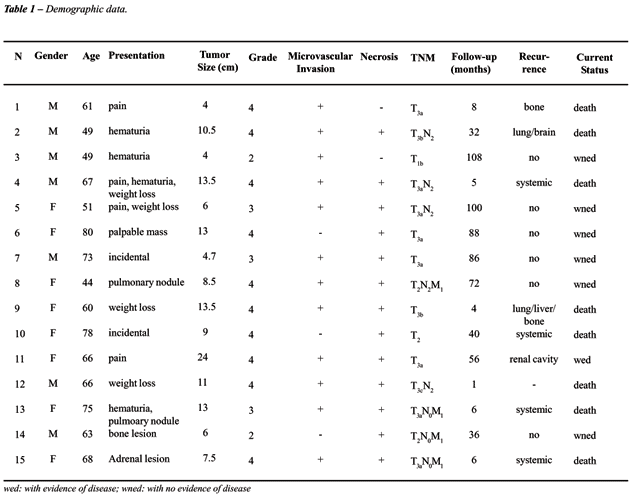
in 4 patients the initial presentation was metastatic lesion. Table-1
lists the demographic data of patients under study.
Eight patients had exclusive sarcomatoid
differentiation, and 7 patients presented association with clear cell
carcinoma (Figure-1).
All patients underwent radical nephrectomy.
One woman who presented recurrence in retroperitoneum on 3 occasions underwent
resection of the lesion at all times, and has been followed for 6 months
now.
Systemic adjuvant treatment was instituted
in 2 patients (13.3%). Patient #8, whose initial presentation was pulmonary
metastatic disease, received adjuvant immunotherapy with interferon and
interleukin with full response and disappearance of the pulmonary nodule,
remaining free of the disease in 72 months of follow-up. Patient #11 received
adjuvant dendritic cell vaccine and is presenting a good performance status
after 56 months of follow-up despite having undergone 3 resections of
retroperitoneal masses with pathological diagnosis of sarcomatoid RCC
and being submitted to new vaccine applications.
Cancer-specific and disease-free survival
curves are represented in Figures-2 and 3, respectively. Mean time to
recurrence of the disease was 30 months. Among the total of 8 deaths,
7 were due to progression of the disease within a mean period of 17.2
months (4 - 40). A 66-year man (#12) with a stage T3cN2M0 tumor who underwent
radical nephrectomy by thoracophrenolaparotomy, died as a consequence
of pulmonary embolism on the 30th postoperative day.


We observed that 80% of the surgical specimens
had microvascular invasion, 87% were tumors with high Fuhrman’s
nuclear grade. Two individuals (14%) had localized disease (stage I and
II), 33% stage III, and 53% stage IV.
COMMENTS
Our
work showed that RCC with sarcomatoid differentiation presents an advanced
stage and is symptomatic in 87% of cases, with a mean tumor size of 9.5
cm, with high Fuhrman’s nuclear grade, and life expectancy of 40%
within 3 years.
There is controversy as to whether the amount
of sarcomatoid tumor is relevant when analyzing the disease’s potential
for recurrence. In the present series, there is similarity concerning
the clinical presentation and the outcome of individuals with pure sarcomatoid
tumors compared with tumors associated with clear cell carcinoma. However,
when there is more than 50% of this component, survival is worse (6,9),
which is contested by Bertoni et al. (8). Nevertheless, the same author
states that only individuals with low Fuhrman’s nuclear grade tumors
and less than 5% of sarcomatoid component have real chances of cure. The
frequency of sarcomatoid tumors with high Fuhrman’s nuclear grade
is 64 - 100% (6,9,12), and in our series, 87 % of tumors presented this
characteristic.
Some evidence suggests that the sarcomatoid
tumor is most often associated with chromophobe RCC (6); however, the
histological type does not influence the disease’s outcome (6,9,11).
In our sample, we did not identify any chromophobe RCC, and the clear
cell subtype was identified in half the cases.
Cheville et al. (12) explain the median
survival of 8 months in their series as resulting from advanced stage,
tumor size and presence of necrosis, with the same significance attributed
by Frank et al. (13). Additionally, the presence of necrosis promotes
a relative risk of 3.35 (12). For the studied group, in 87% of cases there
was tumoral necrosis.
The fact that these tumors are symptomatic
in 86 to 89% of cases (11,12) and the identification of advanced stage
in two-thirds of the cases presenting voluminous tumors explains their
aggressiveness potential. Table-2 displays several studies on sarcomatoid
RCC, facilitating a comparative analysis with the present series and confirming
the invasive profile of the disease. In our sample, the predominant symptoms
were hematuria, pain and emaciation in 75% of patients, with diagnosis
being incidental in only 2 individuals. On the other hand, 4 patients
(26,6%) had metastatic disease at the time of surgery.

Patients with sarcomatoid RCC have a mean
survival of 49.7 months for pT1 stage and 6.8 months for pT2 - pT4 stages
(9,14). Other series present global survival lower than 1 year (5-11).
Surgical resection only, does not significantly
change the disease’s clinical outcome since the majority of these
tumors is metastatic or locally advanced at the moment of diagnosis, with
a very short survival rates for these patients regardless of the type
of treatment (3-5,7,11,15). If left untreated, the sarcomatoid RCC leads
to death within a period of 3.8 to 6.8 months (4,7). The potential for
aggressiveness of such tumors is highlighted by Cangiano et al. (10),
who identified in their series 84% of metastases at the moment of surgery.
Despite controversies concerning the responsiveness of sarcomatoid RCC
to chemotherapy and immunotherapy, (7,10) we had satisfactory responses
with dendritic cell vaccine in 1 case – a woman who presented a
recurrence in the renal cavity and underwent resection of the retroperitoneal
mass at 3 occasions with adjuvant vaccine – and who currently has
an optimal performance status with 56 months of follow-up. In another
patient who received interleukin and interferon adjuvant to nephrectomy,
the pulmonary metastatic lesion disappeared and she is alive and disease-free
72 months after surgery. One individual who underwent resection of a trochanteric
bone lesion following a pathological fracture and whose specimen revealed
metastatic RCC, underwent radical nephrectomy and subsequently femoral
radiotherapy, and has been free of disease for 36 months now. The best
results for regression of metastases with immunotherapy reach 31% in 3
years (16); moreover, the rate of full remission with interferon and interleukin-2
as monotherapy is 15 to 20% (17), which are discouraging responses.
As a future perspective, we should consider
the possibility of adjuvant therapy for high-risk diseases such as renal
sarcomatoid tumors since this treatment is recommended even for localized
disease due to the extremely low morbidity of the autologous renal tumor
cell vaccine (18).
Individuals diagnosed with RCC with sarcomatoid differentiation present
a serious disease with reserved prognosis; however, adjuvant immunotherapy
can improve the outcome of some individuals.
________________________________________
Adriana Sañudo performed the statistical analysis.
REFERENCES
- Greenlee RT, Murray T, Bolden S, Wingo PA: Cancer statistics, 2000. CA Cancer J Clin. 2000; 50: 7-33.
- Hou LT, Willis RA: Renal carcino-sarcoma, true and false. J Pathol Bacteriol. 1963; 85: 139-44.
- Juhasz J, Sebok J, Galambos J, Kiss P: Renal carcinosarcoma (mixed tumours) of the kidney. Int Urol Nephrol. 1980; 12: 103-8.
- Farrow GM, Harrison EG Jr, Utz DC: Sarcomas and sarcomatoid and mixed malignant tumors of the kidney in adults. 3. Cancer. 1968; 22: 556-63.
- Tomera KM, Farrow GM, Lieber MM: Sarcomatoid renal carcinoma. J Urol. 1983; 130: 657-9.
- de Peralta-Venturina M, Moch H, Amin M, Tamboli P, Hailemariam S, Mihatsch M, et al.: Sarcomatoid differentiation in renal cell carcinoma: a study of 101 cases. Am J Surg Pathol. 2001; 25: 275-84.
- Sella A, Logothetis CJ, Ro JY, Swanson DA, Samuels ML: Sarcomatoid renal cell carcinoma. A treatable entity. Cancer. 1987; 60: 1313-8.
- Bertoni F, Ferri C, Benati A, Bacchini P, Corrado F: Sarcomatoid carcinoma of the kidney. J Urol. 1987; 137: 25-8.
- Ro JY, Ayala AG, Sella A, Samuels ML, Swanson DA: Sarcomatoid renal cell carcinoma: clinicopathological. A study of 42 cases. Cancer. 1987; 59: 516-26.
- Cangiano T, Liao J, Naitoh J, Dorey F, Figlin R, Belldegrun A: Sarcomatoid renal cell carcinoma: biologic behavior, prognosis, and response to combined surgical re-section and immunotherapy. J Clin Oncol. 1999; 17: 523-8.
- Mian BM, Bhadkamkar N, Slaton JW, Pisters PW, Daliani D, Swanson DA, et al.: Prognostic factors and survival of patients with sarcomatoid renal cell carcinoma. J Urol. 2002; 167: 65-70.
- Cheville JC, Lohse CM, Zincke H, Weaver AL, Leibovich BC, Frank I, et al.: Sarcomatoid renal cell carcinoma: an examination of underlying histological subtype and an analysis of associations with patient outcome. Am J Surg Pathol. 2004; 28: 435-41.
- Frank I, Blute ML, Cheville JC, Lohse CM, Weaver AL, Zincke H: An outcome prediction model for patients with clear cell renal cell carcinoma treated with radical nephrectomy based on tumor stage, size, grade and necrosis: the SSIGN score. J Urol. 2002; 168: 2395-400.
- Selli C, Hinshaw WM, Woodard BH, Paulson DF: Stratification of risk factors in renal cell carcinoma. Cancer. 1983; 52: 899-903.
- Pantuck AJ, Zisman A, Belldegrun AS: The changing natural history of renal cell carcinoma. J Urol. 2001; 166: 1611-23.
- Figlin RA, Pierce WC, Kaboo R, Tso CL, Moldawer N, Gitlitz B, et al.: Treatment of metastatic renal cell carcinoma with nephrectomy, interleukin-2 and cytokine-primed or CD8(+) selected tumor infiltrating lymphocytes from primary tumor. J Urol. 1997; 158: 740-5.
- Figlin RA: Renal cell carcinoma: management of advanced disease. J Urol. 1999; 161: 381-6.
- Jocham D, Richter A, Hoffmann L, Iwig K, Fahlenkamp D, Zakrzewski G, et al.: Adjuvant autologous renal tumour cell vaccine and risk of tumour progression in patients with renal-cell carcinoma after radical nephrectomy: phase III, randomised controlled trial. Lancet. 2004; 363: 594-9.
_______________________
Received: October 13, 2004
Accepted after revision: January 01, 2005
_______________________
Correspondence address:
Dr. Marcos F. Dall’Oglio
Rua Barata Ribeiro, no. 398, 5o. andar
01308 - 000, São Paulo, SP, Brazil
Fax: + 55 11 3159-3618
E-mail: marcosdallogliouro@terra.com.br