SEMEN
AND URINE CULTURE IN THE DIAGNOSIS OF CHRONIC BACTERIAL PROSTATITIS
(
Download pdf )
L. R. ZEGARRA MONTES, A. A. SANCHEZ MEJIA, C. A. LOZA MUNARRIZ, E. CELIS GUTIERREZ
Department of Surgery, School of Medicine Alberto Hurtado, Peruvian University Cayetano Heredia, Lima, Peru
ABSTRACT
Objective:
To assess the diagnostic accuracy of semen and urine culture in the diagnosis
of chronic bacterial prostatitis (CBP).
Materials and Methods: In 70 consecutive
men suspected of having chronic bacterial prostatitis along with 17 asymptomatic
controls, we obtained urine and semen cultures followed 1 week later by
the Meares and Stamey test, our reference standard. The interpretation
of each of the cultures was blind to the results of other tests.
Results: 139 men were referred for evaluation
of chronic bacterial prostatitis and 70 received all tests. Additionally,
17 control men volunteered to participate. The Meares and Stamey Test
was positive in 69 (79%) patients. The semen culture had a sensitivity
of 45% and a specificity of 94%. The likelihood ratio associated with
a positive semen culture was 8.1 (95% confidence interval (CI) 1.2 to
55.3); the likelihood ratio associated with a negative semen culture was
0.6 (95% CI 0.5 to 0.7). The urine culture had a sensitivity of 4% and
a specificity of 100%. The likelihood ratio of a positive urine culture
was infinity and of a negative urine culture was 0.96 (95% CI 0.9 to 1).
Conclusions: While a positive semen culture
in a symptomatic patient may suffice to select and start antibiotic treatment
against chronic bacterial prostatitis, a negative culture does not rule
out the condition. Urine cultures alone are not useful for diagnosing
CBP. The Meares and Stamey test remains important for the diagnosis of
CBP in practice.
Key
words: urinary tract infections; prostate; prostatitis; diagnosis;
laboratory techniques and procedures
Int Braz J Urol. 2008; 34: 30-40
INTRODUCTION
Chronic
prostatitis (CP) is a very common urologic diagnosis in men (1, 2), with
50% of men having this condition at some point in their life (3). Men
with chronic prostatitis experience a similar loss in quality of life
that survivors of recent acute coronary syndromes do (4).
Chronic bacterial prostatitis (CBP) or chronic
prostatitis category II (5) is defined in men with documented recurrent
urinary tract infections (UTI) who may be asymptomatic between episodes,
or may present chronic genitourinary pain for more than 3 months in association
with bacterial isolation from the prostate (6-8). About 10% of cases of
CP have a bacterial etiology. Escherichia coli accounts for up to 80%
of cases of (CBP) (6).
In 1968, Meares and Stamey described the
four-glass test, which continues to be the reference standard test for
CBP. This test localizes the inflammatory and bacteriologic focus along
the lower urinary tract and prostate (9). The cost, inconvenience and
discomfort to patients, however, decrease its feasibility in practice:
a survey of U.S. urologists found that 80% hardly ever used the Meares
and Stamey test to diagnose CBP (10). Simpler tests including modifications
of the original technique such as the pre and post massage test (11),
expressed prostatic secretion culture, semen culture, and urine culture,
while more feasible, convenient, or inexpensive, seem unsatisfactory alternatives.
The reported sensitivity of semen culture to the diagnosis of CBP varies
between 10 and 100% (12 - 14), and that of urine culture is 10% (13).
In our urology referral service, we have
noticed that patients referred with the tentative diagnosis of chronic
bacterial prostatitis that were ultimately found to have a positive Meares
and Stamey test had a prior negative semen and urine culture. Therefore,
we sought to determine the diagnostic accuracy of the semen and urine
cultures compared to the Meares and Stamey test in the diagnosis of CBP.
MATERIALS AND METHODS
This
is a prospective test performance study. The institutional ethics committee
approved the protocol and consent procedures used in this study.
We enrolled consecutively 70 adult men attending
the Urology and Oncology Service of Cayetano Heredia National Hospital,
in Lima, Peru from September 2003 to October 2004 who had clinical suspicion
of CBP on the basis of recurrent episodes of UTI and/or symptoms of chronic
genitourinary pain within the last 3 months localized in the perineum,
suprapubic area, penis, testes, groin, low back, or pain during or after
ejaculation. Patients gave written informed consent to participate in
this study.
In order to asses the discriminatory capacity
of the semen and urine cultures, we additionally enrolled 17 asymptomatic
adult men who volunteered to participate in this study and who did not
have any history of previous UTI or chronic genitourinary pain.
Ineligible patients showed evidence of other
infections received antibiotic treatment within the month prior to the
study, used urinary catheters or other urological devices, had undergone
prostatectomy, or had prostate cancer. After a complete physical and urological
examination, the participants completed the National Institutes of Health
Chronic Prostatitis Symptom Index (NIH-CPSI) (15, 16).
In order to evaluate the diagnostic performance
of semen and urine cultures, we developed an Alternative test based on
the Meares and Stamey test. Table 1 describes how the samples were collected
for our alternative test.

The samples were examined directly and cultured.
For the Alternative test, we considered the test positive when: 1) there
were bacteria in the semen sample; and 2a) no bacteria was found in the
VB1 and VB2 samples or 2b) the bacterial colony count in the semen sample
was ≥ 10 times that in the VB1 and VB2 specimens. The VB3 sample
was not considered in the analysis of the results of the semen culture.
VB2 cultures were the urine cultures for this study. Positive urine cultures
had colony counts ≥ 105 UFC/mL.
The Meares and Stamey test was taken as
our standard. It was performed according to the standard procedure (Table-1)
(9) and one week after the Alternative test to avoid contamination of
the semen sample by the prostatic secretion. None of the patients received
any antibiotic therapy during this week. For the Meares and Stamey test,
we considered the test positive if there was a 10-fold increase in bacteria
in the expressed prostatic secretion (EPS) or in VB3 samples compared
with the VB1 and VB2 specimens.
All the samples of this study were centrifuged
and seeded on blood agar and McConkey media with standard biochemical
tests to characterize bacteria. Additionally, the presence of over 10
leukocytes per high-power field (X40) in the expressed prostatic secretion
sample indicated prostatitis (17, 18).
All the processing and reading of the samples
of this study were performed by the same expert microbiologist who was
blinded to the patient’s history and results of previous tests.
Samples from the Alternative test received a different codification from
those of the Meares and Stamey test in order to guarantee the independent
interpretation of the results.
Finally, we defined CBP as the presence
of 1) a positive result in the Meares and Stamey test and 2a) the presence
of recurrent episodes of UTI or 2b) symptoms of already described chronic
genitourinary pain.
Statistical data were gathered into a Microsoft
Excel XP database and transferred to the version 9STATA software. We use
descriptive statistics to characterize the study population, and we compare
the characteristics of those with and without documented chronic bacterial
prostatitis using either the chi square test (for proportions) or the
independent sample t-test (for continuous variables). We also estimated
the sensitivity and specificity of each test (and their corresponding
95% confidence intervals) compared with the reference standard, and calculated
the likelihood ratios associated with a positive and a negative semen
culture. The likelihood ratio of a test result is the ratio of the proportion
of patients with CBP who had the test result to the proportion of patients
without CBP who had the same result (19).
We used likelihood ratios because of their
advantages in the assessment of diagnostic tests, i.e., they are less
likely to change with the prevalence of the disease, they can be calculated
for several levels of symptoms, signs or tests, and they can be used to
calculate post-test probability for a target disorder (20).
RESULTS
We
assessed 139 consecutive men referred for symptoms consistent with chronic
prostatitis, but only 70 of them completed all the tests and were considered
in the study. We additionally enrolled 17 asymptomatic patients that volunteered
to participate. The mean age of the population was 37.5 years (±
9.7). Table-2 describes the population by their clinical presentation.
The means and standard deviations of the NIH-CPSI scores are also shown.

The Meares and Stamey test was positive
in 69 patients. Forty-four had gram-negative bacteria, and 25 had gram-positive
bacteria. The isolated bacteria were Escherichia coli in 32, Enterococcus
sp. in 13, Staphylococcus aureus in 10, Klebsiella sp. in 7, Enterobacter
sp. 4, Streptococcus Group D in 1, coagulase-negative Staphylococcus in
1 and Proteus vulgaris in 1. The results of the gold standard and semen
cultures in the symptomatic and asymptomatic patients are shown in Table-3.
There was coincidence in the bacteria isolated in the Alternative test
and their corresponding isolates in the reference standard, except for
one patient who presented S. aureus in the semen culture and E. coli in
the Meares and Stamey test. Although we performed a VB3 sample as part
of our Alternative test, we did not consider it in the analysis of the
semen cultures. It was found positive only in 9 of the 32 patients with
positive semen cultures, with a bacteriologic correlation of 100%.
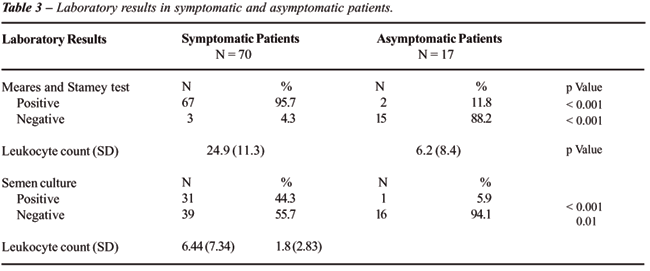
Table-4 describes the performance of the
semen culture in comparison to the reference standard. It shows a semen
culture sensitivity of 45% (95% CI 33.8% to 56.6%), specificity of 94%
(95% CI 74.2% to 99%), a likelihood ratio associated with a positive semen
culture of 8.1 (95% CI 1.2 to 55.3), and likelihood ratio associated with
a negative semen culture of 0.6 (95% CI 0.5 to 0.7).

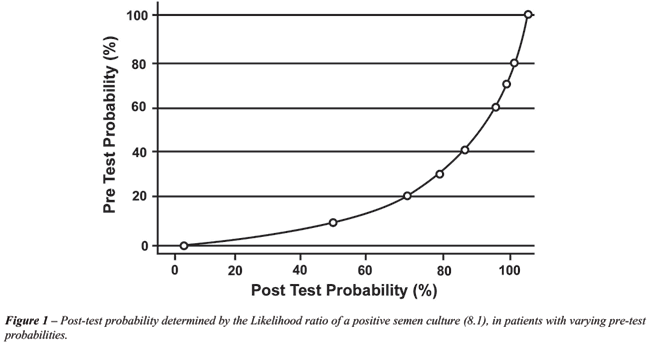
Figures-1 and 2 show how the post-test probability
of having CBP varies compared to the different pre-test probabilities
(clinical suspicion) according to the likelihood ratios.

The performance of the urine culture in
relation to the Meares and Stamey test is shown in Table -5. We found
a sensitivity of 4.3% (95%CI 1.5% to 12%), a specificity of 100% (CI 82.4%
to 100%), an infinite likelihood ratio of a positive urine culture and
a 0.96 (95% CI 0.9 to 1) likelihood of a negative urine culture.

Finally, the leukocyte count per high power
field in expressed prostatic secretions showed that 64 (91.4%) of the
symptomatic males and only 2 (11.7%) of the controls had prostatic inflammation
according to our criteria (p < 0.001). When we changed our parameters
to 5 or more leukocytes per high power field, the number of patients with
inflammation increased to 67 (95.7%) and 7 (41.1%) respectively (p <
0.001). The leukocyte count mean in the EPS sample from the symptomatic
patients was significantly higher than in asymptomatic males; 24.9 (±
11.3) vs. 6.2(± 8.4), p < 0.001. No other significant difference
was seen at the time we compared the leukocyte counts in the other samples
obtained in this study.
COMMENTS
In
our sample, 77% of patients had CBP according to their symptoms and results
in the Meares and Stamey test. The semen and urine cultures revealed limited
diagnostic properties. Our results argue that a negative semen culture
is not an adequate test to rule out CBP, particularly in patients with
high pre-test probability (i.e., men with classic symptoms). On the other
hand, a positive semen culture greatly increases the post-test probability
of CBP and may orient the choice of antibiotic therapy obviating the need
for the cumbersome reference standard test.
Figures-1 and 2 help the clinician determine
how the likelihood ratios associated with the semen culture results determine
the post-test probability of having CBP in patients with varying pre-test
probabilities.
The performance of the urine culture is
poor when used alone to diagnose CBP. In our opinion, its utility in CBP
is only in determining the presence of an active UTI, and then must have
negative or very low counts in order to correctly interpret the results
of the Meares and Stamey tests.
With respect to the presence of prostatic
inflammation, it was found that out of a total of 69 patients with positive
Meares and Stamey test, 66 patients suffered from prostate inflammation.
Of the 3 patients who did not present inflammation according to our definition,
2 had low growth cultures in the EPS samples (5000 and 10000 ufc/cc),
which reflects a good correlation between the microbiological results
and the presence of prostatitis (17, 18).
Our study applies to the urological referral
population in a South American country. The extent to which these results
apply to other patients depends on the extent to which they share similar
clinical presentations, referral patterns, and bacteriology. On the other
hand, our study is strengthened by the evaluation of both alternative
and reference standard tests in patients where there was suspicion of
CBP as well as in asymptomatic patients, with blind interpretation of
the results. Establishing the timing of the samples so that the Meares
and Stamey test always followed the alternative test prevented contamination
of the samples for the alternative test from expressed prostatic secretions.
The semen culture sensitivity in our study
was 44%, which is lower than the sensitivity described by Budia et al
of 100% (14). One explanation is that the semen culture in Budia et al
was obtained after the Meares and Stamey test and could have been “contaminated”
by expressed prostatic secretions. This would decrease the independence
between the test and the reference standard, a known cause of bias (21).
A previous study has shown no differences
in the rates of localization of bacterial cultures for men with chronic
prostatits/chronic pelvic pain syndrome or CP category III compared to
control men (22), but in our study, 95.7% of the symptomatic males and
only 11.8% of the controls had bacterial growth in the Meares and Stamey
test. The underlying explanations for these findings could be that we
included a different set of patients with a higher possibility of having
CBP due to their history of recurrent episodes of UTI and that our controls
were healthy asymptomatic men with no urological complaints or previous
UTI. Only two of our patients with CBP had EPS cultures with the so-called
non-uropathogens (Streptococcus group D in 1 and coagulase-negative Staphylococcus
in 1) (23), and could be labeled as contaminated samples – a category
that we would disagree with due to their corresponding leukocyte count
which was significant for inflammation.
Although one of our limitations is not having
followed the response of our symptomatic patients to their antibiotic
treatment, we consider that our results still reflect the pathogenic role
of these organisms in the development of CBP.
A significant difference was also found
in the leukocyte count of the EPS and semen samples between the symptomatic
and asymptomatic patients – results that differed from the ones
reported in association with CP category III (22), but otherwise consistent
with the isolation of bacteria in our patients with chronic bacterial
prostatitis. Using our definition of more than 10 leukocyte per high power
field as a parameter of prostatic inflammation, we found prostatitis in
91.4% of our symptomatic patients, and when we changed the cut-off to
more than 5 leukocytes per high power field, the frequency increased to
95.7% – a prevalence that differs from the 31% and 49% previously
reported by the NIH chronic prostatitis cohort study (23), and can be
related to the presence of pathogenic bacteria and CBP in almost all our
symptomatic patients.
CONCLUSIONS
While a negative semen culture does not rule out CBP, a positive test in a patient with high pre-test probability of CBP may be sufficient to select and start antibiotic treatment. Urine culture cannot be used alone in the diagnosis of CBP. The Meares and Stamey test, in spite of its difficulty and discomfort for the patient, remains the reference standard for diagnosing CBP in clinical practice.
ACKNOWLEDGEMENTS
Doctors Weymar Melgarejo Zevallos, Raul Medina Ninacondor, Alfonso Del Castillo Mory and Residents Jose Bellido Peti and Jose Medina Holguin from the Section of Urology, Cayetano Heredia National Hospital, Lima, Peru collaborated in the study.
CONFLICT OF INTEREST
None declared.
REFERENCES
- Collins MM, Stafford RS, O’Leary MP, Barry MJ: How common is prostatitis? A national survey of physician visits. J Urol. 1998; 159: 1224-8.
- Nickel JC, Nyberg LM, Hennenfent M: Research guidelines for chronic prostatitis: consensus report from the first National Institutes of Health International Prostatitis Collaborative Network. Urology. 1999; 54: 229-33.
- Naber KG, Weidner W: Chronic prostatitis - an infectious disease?. J Antimicrob Chemother 2000; 46: 157-61.
- Wenninger K, Heiman JR, Rothman I, Berghuis JP, Berger RE: Sickness impact of chronic nonbacterial prostatitis and its correlates. J Urol. 1996; 155: 965-8.
- Krieger JN, Nyberg L Jr, Nickel JC: NIH consensus definition and classification of prostatitis. JAMA. 1999; 282: 236-7.
- Nickel, JC: Prostatitis and related conditions. In: Walsh PC, Retik AB, Vaughan Ed Jr, Wein AJ. (ed.), Campbell’s Urology. 8th ed. Philadelphia, WB Saunders. 2002; pp. 603-26.
- Lummus W, Thompson I. Genitourinary Emergencies: Prostatitis. Emerg Med Clin North Am.2001:19(3).
- Krieger J, McGonagle L: Diagnostic considerations and interpretations of microbiological findings for evaluation of chronic prostatitis. J Clin Microbiol. 1989: 27: 2240-4.
- Meares EM, Stamey TA: Bacteriologic localization patterns in bacterial prostatitis and urethritis. Invest Urol. 1968; 5: 492-518.
- McNaughton Collins M, Fowler FJ Jr, Elliott DB, Albertsen PC, Barry MJ: Diagnosing and treating chronic prostatitis: do urologists use the four-glass test? Urology. 2000; 55: 403-7.
- Nickel JC, Shoskes D, Wang Y, Alexander RB, Fowler JE Jr, Zeitlin S, et al.: How does the pre-massage and post-massage 2-glass test compare to the Meares-Stamey 4-glass test in men with chronic prostatitis/chronic pelvic pain syndrome? J Urol. 2006; 176: 119-24.
- Mendoza DN, Aguirre CR, Del Castillo MA, Loza MC, Melgarejo ZW, Medina NR, et al.: Evaluación de la sensibilidad del espermocultivo en el diagnostico de prostatitis bacteriana crónica. Rev Med Hered. 2004; 15: 37-43.
- Lacquaniti S, Fulcoli V, Weir JM, Pisanti F, Servello C, Destito A: Bacterial prostatitis: urine and spermatic fluid culture. Arch Ital Urol Androl. 2000; 72: 21-3.
- Budia A, Luis Palmero J, Broseta E, Tejadillos S, Benedicto A, Queipo JA, et al.: Value of semen culture in the diagnosis of chronic bacterial prostatitis: a simplified method. Scand J Urol Nephrol. 2006; 40: 326-31.
- Litwin MS, McNaughton-Collins M, Fowler FJ Jr, Nickel JC, Calhoun EA, Pontari MA, et al.: The National Institutes of Health chronic prostatitis symptom index: development and validation of a new outcome measure. Chronic Prostatitis Collaborative Research Network. J Urol. 1999; 162: 369-75.
- Collins MM, O’Leary MP, Calhoun EA, Pontari MA, Adler A, Eremenco S, et al.: The Spanish National Institutes of Health-Chronic Prostatitis Symptom Index: translation and linguistic validation. J Urol. 2001; 166: 1800-3.
- Schaeffer AJ, Wendel EF, Dunn JK, Grayhack JT: Prevalence and significance of prostatic inflammation. J Urol. 1981; 125: 215-9.
- Thorburn-Wright E, Chmiel JS, Grayhack JT, Schaeffer AJ: Prostatic fluid inflammation in prostatitis. J Urol. 1994; 152: 2300-3.
- Bianchi MT, Alexander BM: Evidence based diagnosis: does the language reflect the theory? BMJ. 2006; 333: 442-5.
- Fletcher RH, Fletcher SW. Diagnosis. In: Clinical Epidemiology: The Essentials. Fourth Edition. Philadelphia, Lippincott Williams and Wilkins. 2005; pp. 35-58.
- Lijmer JG, Mol BW, Heisterkamp S, Bonsel GJ, Prins MH, van der Meulen JH, et al.: Empirical evidence of design-related bias in studies of diagnostic tests. JAMA. 1999; 282: 1061-6. Erratum in: JAMA 2000; 283: 1963.
- Nickel JC, Alexander RB, Schaeffer AJ, Landis JR, Knauss JS, Propert KJ, et al.: Leukocytes and bacteria in men with chronic prostatitis/chronic pelvic pain syndrome compared to asymptomatic controls. J Urol. 2003; 170: 818-22.
- Schaeffer AJ, Knauss JS, Landis JR, Propert KJ, Alexander RB, Litwin MS, et al.: Leukocyte and bacterial counts do not correlate with severity of symptoms in men with chronic prostatitis: the National Institutes of Health Chronic Prostatitis Cohort Study. J Urol. 2002; 168: 1048-53.
____________________
Accepted
after revision:
October 20, 2007
_______________________
Correspondence
address:
Dr. Luis Zegarra Montes
Departamento de Cirugía
Facultad de Medicina Alberto Hurtado
Av. Honorio Delgado 430, Urb. Ingeniería
San Martín de Porres, Lima, Peru
Fax: + 511 460-5499
E-mail: lzegarram@upch.edu.pe
EDITORIAL COMMENT
This
interesting study follows the traditional viewpoint that bacteria found
in semen have clinical relevance in the generation of symptoms in men.
The well conducted study from the NIH collaborative group showed that
both symptomatic and asymptomatic men had similar bacterial counts in
their semen, suggesting that these bacterial commensuals have nothing
to do with the generation of symptoms seen in these men. This study basically
attempts to reproduce the traditional belief, but adds the wrinkle of
comparing the Meares and Stamey test with their own modifications.
Although
the study is of interest, one weakness should be mentioned: i.e. the semen
bacteria may have nothing to do with the disease process.
Dr. C.
Lowell Parsons
Division of Urology, Department of Surgery
University of California, San Diego Med Ctr.
San Diego, California, 92103-8897, USA
E-mail: cparsons@ucsd.edu
EDITORIAL COMMENT
Prostatitis
is a very challenging disease. The causes are not known and diagnostic
methods are difficult to apply. The survey of U.S. urologist found that
80% hardly ever used the Meares & Stamey test to diagnose chronic
bacterial prostatitis. In this issue, the article by Zegarra Montes et
al. addresses an important issue regarding finding more feasible methods
to diagnose chronic bacterial inflammation. Semen sample was used instead
of expressed prostatic secretion sample. The alternative test did not
outdo the Meares & Stamey test in the sensitivity. While a positive
semen culture in a symptomatic patient may justify the treatment with
antibiotics, a negative culture does not rule out the condition. Relatively
small and regional study population and selection of the samples may account
for the differences with previous studies showing higher sensitivity for
the use of semen. It is obvious that carefully conducted large cohort
studies are required in order to asses the accuracy of traditional methods
vs. urine and semen cultures to establish the significance of acute or
chronic prostatitis. Careful localization cultures of urine, expressed
prostatic secretion, and semen along with the antimicrobial susceptibility
combined with clinical symptoms remain clinically important in management
of prostatic infections. Follow up of anti-microbial or anti-inflammatory
therapies would also be important when correlated with the diagnosis.
Only
a small percentage of all prostatitis cases involve proven bacterial infection.
Prostatitis is a much wider diagnostic and therapeutic problem. The classification
of National Institutes of Health (NIH) divides prostatitis into four categories.
The differentiation between the categories is based on the presence or
absence of bacteria, leukocytes, and clinical symptoms. Category I (acute
bacterial prostatitis) and Category II (chronic/recurrent bacterial prostatitis)
have infectious etiologies. Category III refers to chronic nonbacterial
prostatitis/chronic pelvic pain syndrome (CP/CPPS). The differentiation
between IIIa and IIIb is based on the finding of inflammatory cells in
EPS or voided specimen 3. Category IV refers to asymptomatic inflammatory
prostatitis that is diagnosed incidentally during evaluation of the patient
for other purposes.
Recent
studies have presented problems with this classification system (1,2).
Prostate biopsies from patients with non-inflammatory CPPS (category IIIb)
display a low-grade inflammation with diffuse distribution of lymphocytes
into the stroma and periglandular space. Inflammation appears to be common
in both forms of category III. The traditional marker of inflammation,
leukocytes in the prostatic fluid, does not correlate with the predominant
symptom of pelvic pain. Schaeffer et al. (3) assessed the relationship
between leukocytes and bacteria and symptom severity in men with CP/CPPS.
They observed no association between inflammation in any of the specimen
sites and symptoms. Further, no association between bacterial colonization
and symptoms were observed. Tsuboi et al. (4) found no correlation between
the aggressiveness and extent of inflammation and leukocyte count in prostatic
fluid. Neither a correlation was found between the number of leukocytes
in EPS and the histopathology of the prostate. The study by True et al.
(5) examined correlations between the symptoms and histology of prostatitis
and suggested that histologic inflammation may not be a significant factor
in the process of CP/CPPS. This findings extended by the large scale REDUCE
trial data which suggest that presence of chronic prostatitis-like symptoms
did not provide any discriminative value for a histologic diagnosis of
either acute or chronic inflammation (6). One has to conclude that leukocytes
and bacteria in the prostatic fluid do not distinguish between symptomatic
and asymptomatic individuals. Moreover, the lack of or weak correlation
between inflammation and infection with severity of symptoms implies that
factors other than inflammation and infection contribute to symptoms associated
with CP/CPPS (3).
Histologic
inflammation may not be important for the development of chronic pelvic
pain but it may bear other significances. There is emerging evidence that
inflammation in the prostate gland may be associated with BPH, voiding
dysfunctions and prostate cancer. Chronic inflammatory infiltrates have
been associated with human BPH nodules, and it is likely that gradual
infiltration of the prostate by lymphocytes leads to BPH (7). Results
from the REDUCE trial confirmed the important role of inflammation in
BPH (6). A statistically significant correlation was found between histological
grade of chronic prostatic inflammation and lower urinary tract symptoms
(8). The correlation was weak but did not preclude the possibility that
histopathological inflammation may be strongly correlated with changes
over time. Patients with chronic prostatitis may be more likely to develop
bladder dysfunction, bladder outlet obstruction or urinary retention than
men without inflammation. Finally, accumulating evidence indicates the
significance of inflammation in human prostate carcinogenesis. Chronic
or recurrent acute inflammation, a product of infectious agents or other
sources, has potential promotional roles in the development of prostate
cancer (9).
There
is evidence predominantly from animal studies that the nonbacterial prostatic
inflammation results from an autoimmune process (10). The onset of autoimmune
reaction may be triggered by an infection through antigen mimicry. Although
no microorganisms are detectable, it has been suspected that an infection
(occult, unculturable, or regarded as non-pathogen) may be responsible
for the changes in immunological parameters (11). The infectious and autoimmune
etiologies would thus be compatible. Further, the possible autoimmune
process may be under the hormonal control. Findings in preclinical models
indicate that the balance between immunosuppressive androgens and pro-inflammatory
estrogens may be of particular importance (12,13). Finally, intraprostatic
reflux of urine or semen may produce a chemical injury to the epithelium
that initiates the immunological reaction. There may be several triggers
of inflammation, which act in concert simultaneously or sequentially.
If
inflammation is indeed in causal relationship with BPH and prostate cancer,
anti-inflammatory agents should be investigated as drug candidates for
the treatment and prevention of BPH and prostate cancer (6,9).
REFERENCES
- Maake C, John H: Prostatitis versus pelvic pain syndrome: immunologic studies. Curr Urol Rep. 2003;4 : 327-34.
- Pontari MA, Ruggieri MR: Mechanisms in prostatitis/chronic pelvic pain syndrome. J Urol. 2004; 172: 839-45.
- Schaeffer AJ, Knauss JS, Landis JR: Leukocyte and bacterial counts do not correlate with severity of symptoms in men with chronic prostatitis: the National Institutes of Health Chronic Prostatitis Cohort Study. J Urol. 2002; 168: 1048-53.
- Tsuboi N, Nishimura T, Chen H, Norose Y, Shimizu M, Kondo Y, at al.: Relation of leukocytosis in prostatic fluid and inflamed prostatic tissue. J Nippon Med Sch. 2007; 74: 210-216.
- True LD, Berger RE, Rothman I, Ross SO, Krieger JN: Prostate histopathology and the chronic prostatitis/chronic pelvic pain syndrome: a prospective biopsy study. J Urol. 1999; 162: 2014-2018.
- Nickel JC: Inflammation and benign prostatic hyperplasia. Urol Clin North Am. 2008; 35: 109-15.
- Eaton CL: Aetiology and pathogenesis of benign prostatic hyperplasia. Curr Opin Urol. 2003, 13: 7-10.
- Nickel JC, Roehrborn CG, O’Leary MP, Bostwick DG, Somerville MC, Rittmaster RS: The relationship between prostate inflammation and lower urinary tract symptoms: examination of baseline data from the REDUCE trial. Eur Urol. 2007; PMID: 18036719 [Epub ahead of print].
- Stock D, Groome PA, Siemens DR: Inflammation and prostate cancer: a future target for prevention and therapy? Urol Clin North Am. 2008; 35: 117-30.
- Motrich RD, Maccioni M, Riera CM, Rivero VE: Autoimmune prostatitis: state of the art. Scand J Immunol. 2007; 66: 217-27.
- Krieger JN, Ross SO, Riley DE: Chronic prostatitis: epidemiology and role of infection. Urology. 2002; 60: 8-12.
- Bernoulli J, Yatkin E, Talvitie EM, Santti R, Streng T: Urodynamic changes in a Noble rat model for nonbacterial prostatic inflammation. Prostate. 2007; 67: 888-899.
- Bernoulli J, Yatkin E, Laakso A, Anttinen M, Bosland MC, Vega K, et al.: Histopathological evidence for an association of inflammation with ductal PIN-like lesions but not with ductal adenocarcinoma in the prostate of the Noble rat. Prostate. 2008 (in press).
Dr. Emrah Yatkin &
Dr. Risto Santti
Dept of Anatomy, Institute of Biomedicine
University of Turku
Turku, Finland
E-mail: emrah.yatkin@utu.fi