Prostatic
Atrophy. Clinicopathological Significance
(
Download pdf )
Review Article
doi: 10.1590/S1677-55382010000400003
Athanase Billis
Department of Anatomic Pathology, School of Medicine, University of Campinas (Unicamp), Campinas, Sao Paulo, Brazil
ABSTRACT
Prostatic atrophy is a benign lesion that may mimic adenocarcinoma histologically and on imaging. It is more frequent in the peripheral zone and has gained importance with the increasing use of needle biopsies. Diffuse atrophy occurs secondarily to radiotherapy and/or endocrine therapy. Inflammation and/or chronic local ischemia may cause focal atrophy with an increasing frequency in age. Atrophy may be classified morphologically into diffuse and focal. The latter may be partial, complete or combined. Partial focal atrophy is the most frequent mimicker of adenocarcinoma on needle biopsies. Complete focal atrophy may be subtyped into simple, sclerotic and hyperplastic (or postatrophic hyperplasia). Combined lesions are frequent and partial atrophy may precede complete atrophy. The several morphologic types of focal atrophy may represent a morphologic continuum and the hyperplastic (or postatrophic hyperplasia) subtype seems to be at the extreme end of this continuum. Chronic inflammation associated to focal atrophy (proliferative inflammatory atrophy) has been linked to high-grade prostatic intraepithelial neoplasia and/or carcinoma. This link, however, remains controversial in the literature. The question whether inflammation directly produces tissue damage and atrophy or some other insult induces atrophy directly, with inflammation occurring secondarily, is still unresolved. An intriguing finding that needs further studies is a possible association of extent of atrophy to serum PSA elevation.
Key
words: prostate; atrophy; prostatic neoplasms; carcinoma; biopsy;
needle
Int Braz J Urol. 2010; 36: 401-9
INTRODUCTION
Prostatic atrophy (PA) is one of the most frequent histologic mimics of prostatic adenocarcinoma (1). On conventional and color Doppler transrectal ultrasound and on magnetic resonance spectroscopic imaging studies, PA may also simulate prostate cancer (2,3). Thus, PA should be considered together with prostatitis as an important cause of false-positive results on imaging of the prostate (2). Of further interest as regards this lesion relates to a possible link to high-grade intraepithelial neoplasia (HGPIN) and/or carcinoma (4). PA occurs most frequently in the peripheral zone (5-7) and gained importance with the increasing use of needle biopsies for the detection of prostatic carcinoma (8). It is a very frequent lesion: 83.7% on needle biopsies (9) and 85% in autopsies (10).
HISTORICAL BACKGROUND
AND NOMENCLATURE
The
nomenclature of PA is cumbersome and confusing. Moore (5), in 1936, was
one of the first authors to describe prostatic atrophy in a systematic
autopsy study. He found that there was a strong correlation with age and,
according to his study, prostatic atrophy is initiated during the 5th
decade and continues as a progressive process into the 8th decade. Moore
classified PA into simple acinar atrophy and sclerotic atrophy.
In 1954, Franks (6) added to simple acinar
atrophy and sclerotic atrophy a lesion that he called postatrophic hyperplasia.
When hyperplasia followed simple acinar atrophy, it was called lobular
hyperplasia. Sclerotic atrophy with hyperplasia was called postsclerotic
hyperplasia. According to the author, lobular hyperplasia and postsclerotic
hyperplasia are closely related existing conditions in which it is impossible
to decide to which group a particular area of hyperplasia belongs.
Since these first studies, authors have
described similar morphologic features with different names. For example,
despite not using the term hyperplasia, Moore’s Figure-14 called
by the author as simple acinar atrophy (5) is identical to the lesion
shown in Figure-8 and called lobular hyperplasia in Franks’ article
(6).
PATHOGENESIS
Radiotherapy and/or endocrine therapy are established causes of diffuse atrophy. Inactive or active inflammation is a frequent cause of focal atrophy. Based on a study of autopsies there is evidence that chronic local ischemia may be a cause of focal atrophy, which was clearly more frequent in advanced age (10). A further evidence for a possible role of chronic local ischemia in the pathogenesis of prostatic atrophy was the finding that blood flow, assessed by color Doppler transrectal ultrasound, was absent in 60% of hypoechoic nodules due to prostatic atrophy (3). However, many examples of atrophy are still considered idiopathic in nature (11).
MORPHOLOGIC CLASSIFICATION
A
morphologic classification of prostatic atrophy is shown in Table-1. In
diffuse atrophy, all acini of the gland are atrophic. This occurs secondarily
to radiotherapy and/or hormonal therapy. Focal atrophy is the usual lesion
seen by the pathologist on routine practice. It occurs in patches and
preserves the lobular architecture of the gland. The latter feature helps
in the histological differential diagnosis of adenocarcinoma.
Partial atrophy. This is the most common
benign variant of focal atrophy that causes difficulty in the differential
diagnosis of adenocarcinoma (1,12). The lesion was reported in the literature
in 1998 (13). Architecturally, partial atrophy consists of crowded glands
often with a disorganized growth pattern. In contrast to complete atrophy,
which can typically be diagnosed at scanning magnification owing to the
presence of well-formed glands with a very basophilic appearance, partial
atrophy has a pale cytoplasm lateral to the nuclei giving rise to pale
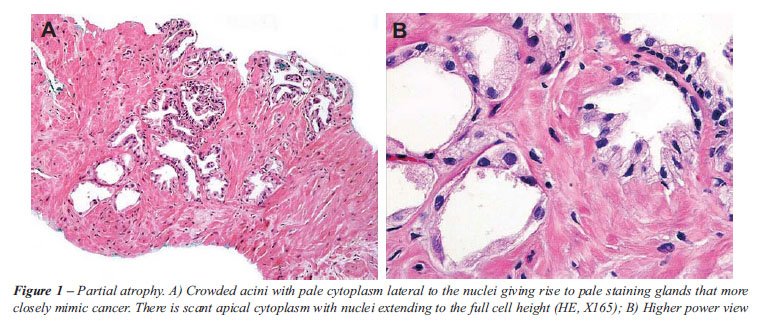
staining glands that more closely mimic cancer (Figure-1). An additional
difficulty in distinguishing cancer from partial atrophy is the positivity
for alpha-methylacyl coenzyme A racemase (AMACR) in some acini (1,12,14).

Complete atrophy. The glands show a basophilic
appearance due to the scant cytoplasm and crowding of nuclei. The hyperplastic
(or postatrophic hyperplasia) subtype most frequently mimics adenocarcinoma.
In a working group classification (15), complete focal atrophy was subtyped
into simple atrophy, simple atrophy with cyst formation, and postatrophic
hyperplasia. Based on an autopsy study, complete focal atrophy was subtyped
into simple atrophy, sclerotic atrophy, and hyperplastic atrophy (or postatrophic
hyperplasia) (10). Sclerotic atrophy in the morphologic classification
shown in Table-1 corresponds to simple atrophy with cyst formation in
the working group classification.
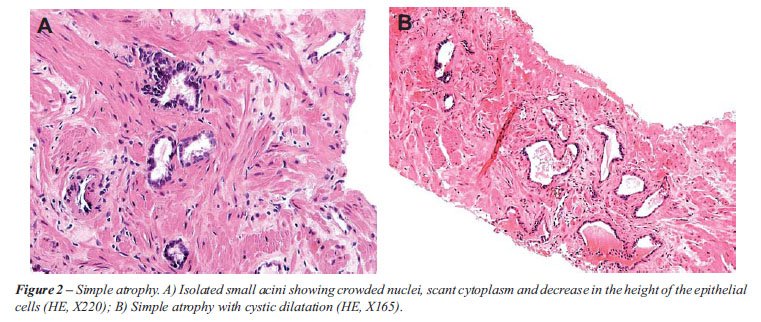
Simple atrophy. Usually involves an entire
lobule, although isolated acini may be affected. The acini are small showing
scant cytoplasm and decrease in the height of the epithelial cells (Figure-2A).
They may be cystic with flattened epithelium (Figure-2B). The surrounding
stroma may or may not show any fibrosis.
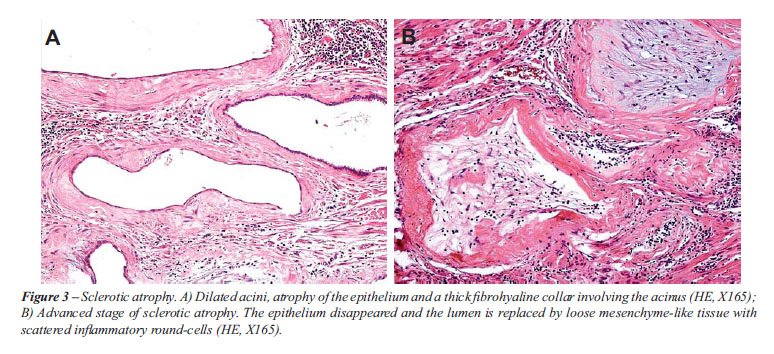
Sclerotic atrophy. This is a very peculiar
and distinctive lesion described in detail by Moore in 1936 (5). There
is simultaneous atrophy of the epithelium and a proliferation of the fibroblasts
around the acinus. Continued proliferation results in a fibrohyaline collar
frequently associated to elastosis that involves the acinus. With higher
degrees of hyalinization the acini dilate, sometimes prominently, and
the epithelium becomes extremely flattened and eventually no longer can
be identified (Figure-3A). In advanced stages, the lumen is replaced by
loose mesenchyme-like tissue with scattered inflammatory round-cells (Figure-3B).
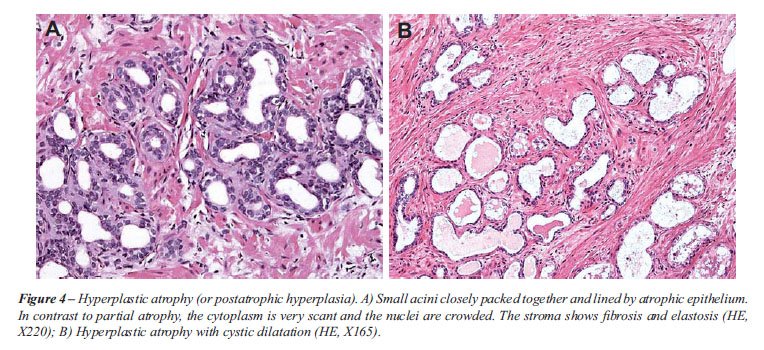
Hyperplastic atrophy (or postatrophic hyperplasia).
Shows small acini closely packed together and lined by atrophic epithelium
(Figure-4A). The acini may also show cystic dilatation (Figure-4B). In
contrast to partial atrophy, the cytoplasm is very scant and the nuclei
are crowded conferring a basophilic tinge to the lesion. When fibrosis
is present in the stroma, the proliferation is irregular and can result
in distortion of the acini mimicking infiltrative adenocarcinoma. Elastosis
of the stroma (Figure-4A) may be seen in all subtypes of complete atrophy
and is a microscopic feature useful for the differential diagnosis of
adenocarcinoma (16). The cysts of simple or hyperplastic atrophy must
be differentiated from other simple or multiple parenchymal cysts due
to retention of prostatic secretions, difficult drainage of secretions
due to benign prostatic hyperplasia (BPH) nodule, inflammation of ducts
that leads to obstruction, and cystic degeneration of BPH (17).
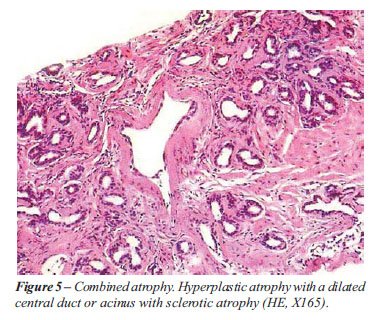
Combined. Subtypes of focal complete atrophy
are frequently combined (10). They may occur in adjacent but separate
foci or merging in the same focus. Hyperplastic atrophy with a central
duct or acinus showing sclerotic atrophy is a frequent combined lesion
in the same focus and very characteristic of prostatic atrophy being a
feature useful for the differential diagnosis with adenocarcinoma (Figure-5).
The existence of combined subtypes supports the hypothesis that complete
prostatic atrophy is a morphologic continuum and that hyperplastic type
(or postatrophic hyperplasia) seems to be at the extreme end of this morphologic
continuum (8). The hyperplastic small acini seem to represent a regenerative
process. A high cell proliferation using immunohistochemistry seems to
support this hypothesis (18).
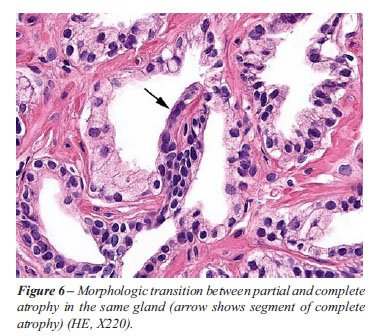
Partial and complete atrophy may be also
combined. Studying needle prostatic biopsies we found that partial atrophy
and complete atrophy were present concomitantly in 47/75 (63%) biopsies.
In 20/75 (27%) biopsies, we found topographical mergence of partial and
complete atrophy. In these areas, transitions between partial and complete
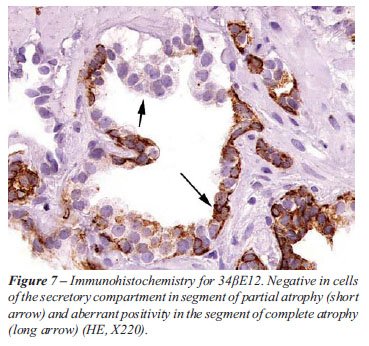
atrophy could be appreciated in the same gland (Figure-6). Based on the
aberrant phenotypic expression of the secretory compartment in complete
atrophy (19) but not in partial atrophy, immunohistochemistry may highlight
these transitions in the same gland (Figure-7).
In a study by Oppenheimer et al. (13) partial
atrophy was present simultaneously with more fully developed atrophy (complete
atrophy) in 35.35% of biopsies. In the study by Wang et al. (12), 48 of
278 (17.3%) partial atrophy cases were mixed with postatrophic hyperplasia.
Przybycin et al. (20) found a much higher frequency. The authors described
that complete atrophy was present but distinct from partial atrophy foci
in 41/45 (91%) needle biopsies.
What is the significance of the mergence
of partial atrophy and complete atrophy and the transitions in the same
gland? According to Oppenheimer et al. (13) the transition from partial
to more established atrophy, is accompanied by a parallel increase in
the nuclear/cytoplasmic ratio, suggesting gradual evolution from the partial
form of atrophy to the more complete variety. This suggests that partial
atrophy may be also part of a morphologic continuum in focal prostatic
atrophy.
An intriguing finding in partial atrophy
foci is the very rare presence of chronic unspecific inflammation. Przybycin
et al. (20), found inflammation in an insignificant 1% of partial atrophy
foci. In a study on 75 biopsies, we did not find chronic unspecific inflammation
in partial atrophy foci as well as in areas of topographic mergence between
these lesions. On the other hand, inflammation was frequently seen in
complete atrophy foci: 56.2%, 48%, and 54.3%, in simple, sclerotic, and
hyperplastic atrophy, respectively.
PRECANCEROUS LESION?
The
term “proliferative inflammatory atrophy” (PIA) was proposed
by De Marzo et al. (4) to designate focal simple or postatrophic hyperplasia
occurring in association with inflammation. Several studies have postulated
that PIA may represent a precursor lesion to high-grade prostatic intraepithelial
neoplasia (HGPIN) and, therefore, prostatic carcinoma (4,19,21-23). Chronic
inflammation of longstanding duration has been linked to the development
of carcinoma in several organ systems and HGPIN is considered the most
likely precursor of prostate carcinoma (4,24).
Several separate findings provide supportive evidence for this novel hypothesis:
1) There is a shift in the topographic fidelity of proliferation in PIA
similar to HGPIN and carcinoma (4). Most cell division in the normal human
prostate epithelium occurs in the basal cell compartment, yet HGPIN and
adenocarcinoma cells possess phenotypic and morphologic features of secretory
cells. Thus, cell proliferation has been shifted up from the basal into
the secretory compartment in HGPIN and carcinoma;
2) The phenotype of many of the cells in PIA is most consistent with that
of an immature secretory-type cell similar to that for the cells of HGPIN
and carcinoma (4,19,22). Atrophic luminal cells show an intermediate phenotype
in that many cells express bcl-2 (normally a basal cell marker in the
prostate), and virtually all of the cells express high levels of keratins
8/18. Intermediate cell population has been suggested to represent amplifying
cells modulating the expansion and development of the prostate epithelium.
Increased proliferation has been observed in atrophic glandular epithelium
(18);
3) PIA, HGPIN, and carcinoma all occur with high prevalence in the peripheral
zone and low prevalence in the central zone of the human prostate (21);
4) Topographic relation of PIA to HGPIN, i.e. areas of atrophy merging
directly with areas of HGPIN within the same glands. In radical prostatectomy
specimens, Putzi and De Marzo (21) identified morphologic merging between
PIA and HGPIN in 34% of the PIA lesions. They also found frequent occurrences
of small carcinoma lesions in the vicinity of focal atrophy. In a study
by Wang et al. (22), a total of 1,188 HGPIN lesions were identified, of
which 17% (198) were in the morphological process of merging with PIA.
Thirty-six PIA-merging prostatic carcinoma lesions were also detected.
The atrophic epithelial cells in such merging lesions had increased Ki-67
proliferative index and an intermediate phenotype: increased expression
for cytokeratin 5, GSTP1, c-MET, and C/EBPß.
The link of PIA to prostatic carcinogenesis
and the morphological transition of PIA, HGPI, and invasive carcinoma,
however, are not favored in other studies. In autopsies, prostates with
atrophy showed no association with histologic carcinoma and/or HGPIN (10).
The only significant association found was with arteriosclerosis. In this
study ischemia caused by local intense arteriosclerosis seemed to be a
potential factor for the pathogenesis of atrophy. In a subsequent study,
the same lack of association was found to HGPIN and/or histologic carcinoma
comparing atrophy with and without inflammation (9).
In 272 radical prostatectomies, Anton et
al. (25) analyzed the presence, location, and number of foci of postatrophic
hyperplasia. They found the lesion in 32% of radical prostatectomy specimens
and in 12% of cystoprostatectomy specimens, and concluded that is a relatively
common lesion but without any topographical association with prostatic
carcinoma. In 172 needle prostatic biopsies, from a total of 481 cores
with cancer, 184/481 (38.25%) cores showed no atrophy; 166/481 (34.51%)
cores showed atrophy and no inflammation; 111/481 (23.08%) cores showed
both PIA and atrophy without inflammation; and 20/481 (4.16%) cores showed
only PIA (26). Postma et al. (27) evaluated whether the incidence of atrophy
reported on sextant biopsies was associated with subsequent prostate cancer
detection. The authors concluded that atrophy, especially in its simple
form, is a very common lesion in prostate biopsy cores (94%). Atrophy
in an asymptomatic population undergoing screening was not associated
with a greater prostate cancer or HGPIN incidence during subsequent screening
rounds.
A question to be raised is whether atrophy
by itself is implicated in carcinogenesis or the key event is chronic
inflammation leading to atrophy, HGPIN and cancer. This question is still
unresolved (28,29). Inflammation directly may produce tissue damage and
atrophy or, alternatively, some other insult like ischemia induces the
atrophy directly, with inflammation occurring secondarily. Hypothesizing
that partial atrophy precedes complete atrophy, the absence of inflammation
in the former as well as in areas of mergence between these two lesions
seems to favor that chronic inflammation in complete focal atrophy may
be a secondary phenomenon.
PROSTATIC ATROPHY AND
PSA
An
intriguing finding was a positive and significant association between
extent of atrophy and total or free serum prostate-specific antigen (PSA)
elevation (30). The study was based on 131 needle prostatic biopsies corresponding
to 107 patients. The only diagnosis in all biopsies was focal prostatic
atrophy without presence of cancer, HGPIN or suspicious for cancer (ASAP).
In a subsequent study it was shown that this association is not related
to the type of atrophy (31).
What would be a possible pathogenesis for
the serum PSA elevation associated with focal prostatic atrophy? It is
intriguing that cells of the secretory compartment of atrophic acini may
produce higher levels of PSA. The authors hypothesize that injurious stimuli
causing focal prostatic atrophy may interfere in the physiologic barrier
that prevents the escape of any significant amounts of PSA to the general
circulation.
PSA is a single chain glycoprotein with
proteolytic enzyme activity mainly directed against the major gel-forming
protein of the ejaculate (semenogelin). PSA induces liquefaction of semen
with release of progressively motile spermatozoa (32). There are several
efficient physiologic barriers to prevent the escape of any significant
amounts of PSA from the prostatic ductal system: basement membrane of
the acini, basal cells lining the acini, prostatic stroma, basement membrane
of capillary endothelial cells, and endothelial cells. These barriers
normally prevent PSA from entering the general circulation at concentrations
of more than 3 ng/mL (32).
Focal prostatic atrophy represents a form
of adaptive response to injury most commonly to inflammation and/or local
ischemia. Inflammation and/or ischemia are injurious stimuli resulting
in diminished oxidative phosphorilation, membrane damage, influx of intracellular
calcium, and accumulation of oxygen-derived free radicals (oxidative stress)
(4). Studies showing elevated levels of glutathione S-transferase P1,
glutathione S-transferase A1, and Cox-2 in prostatic atrophic epithelial
cells suggest a stress-induced response (33-35). We do not know which
mechanisms are involved in the physiologic barrier that prevents the escape
of any significant amounts of PSA to the general circulation, however,
all these stress-induced responses may affect this barrier. Inflammation
and particularly ischemia may have also a field effect affecting the physiologic
barrier of normal acini close to atrophic acini.
CONCLUSION
Prostatic atrophy is a benign lesion frequently found on needle biopsies, which may mimic histologically and on imaging adenocarcinoma. Diffuse atrophy may be secondary to radiation and/or endocrine therapy. Inflammation and chronic local ischemia are the main causes involved in the pathogenesis of focal atrophy, which increases in frequency with age. Atrophy may be morphologically classified into diffuse and focal. The latter may be partial, complete and combined. Partial atrophy is the most frequent mimicker of adenocarcinoma on needle biopsies. Probably the several morphologic variants of focal atrophy represent a morphologic continuum and the hyperplastic (or postatrophic hyperplasia) subtype seem to be at the extreme end of this continuum. A possible link of prostatic atrophy to HGPIN and/or carcinoma remains controversial in the literature. The possible association of extent of prostatic atrophy to serum PSA elevation needs further studies.
CONFLICT OF INTEREST
None declared.
REFERENCES
- Herawi M, Parwani AV, Irie J, Epstein JI: Small glandular proliferations on needle biopsies: most common benign mimickers of prostatic adenocarcinoma sent in for expert second opinion. Am J Surg Pathol. 2005; 29: 874-80.
- Prando A, Billis A: Focal prostatic atrophy: mimicry of prostatic cancer on TRUS and 3D-MRSI studies. Abdom Imaging. 2009; 34: 271-5.
- Meirelles LR, Billis A, Cotta AC, Nakamura RT, Caserta NM, Prando A: Prostatic atrophy: evidence for a possible role of local ischemia in its pathogenesis. Int Urol Nephrol. 2002; 34: 345-50.
- De Marzo AM, Marchi VL, Epstein JI, Nelson WG: Proliferative inflammatory atrophy of the prostate: implications for prostatic carcinogenesis. Am J Pathol. 1999; 155: 1985-92.
- Moore RA: The Evolution and Involution of the Prostate Gland. Am J Pathol. 1936; 12: 599-624.
- Franks LM: Atrophy and hyperplasia in the prostate proper. J Pathol Bacteriol. 1954; 68: 617-21.
- Liavåg I: Atrophy and regeneration in the pathogenesis of prostatic carcinoma. Acta Pathol Microbiol Scand. 1968; 73: 338-50.
- Cheville JC, Bostwick DG: Postatrophic hyperplasia of the prostate. A histologic mimic of prostatic adenocarcinoma. Am J Surg Pathol. 1995; 19: 1068-76.
- Billis A, Magna LA: Inflammatory atrophy of the prostate. Prevalence and significance. Arch Pathol Lab Med. 2003; 127: 840-4.
- Billis A: Prostatic atrophy: an autopsy study of a histologic mimic of adenocarcinoma. Mod Pathol. 1998; 11: 47-54.
- Srigley JR: Benign mimickers of prostatic adenocarcinoma. Mod Pathol. 2004; 17: 328-48.
- Wang W, Sun X, Epstein JI: Partial atrophy on prostate needle biopsy cores: a morphologic and immunohistochemical study. Am J Surg Pathol. 2008; 32: 851-7.
- Oppenheimer JR, Wills ML, Epstein JI: Partial atrophy in prostate needle cores: another diagnostic pitfall for the surgical pathologist. Am J Surg Pathol. 1998; 22: 440-5.
- Worschech A, Meirelles L, Billis A: Expression of AMACR (alpha-methylacyl coenzyme A racemase) in partial and complete focal atrophy on prostate needle biopsies. Anal Quant Cytol Histol. 2010; (in press).
- De Marzo AM, Platz EA, Epstein JI, Ali T, Billis A, Chan TY, et al.: A working group classification of focal prostate atrophy lesions. Am J Surg Pathol. 2006; 30: 1281-91. Erratum in: Am J Surg Pathol. 2006; 30: 1489.
- Billis A, Magna LA: Prostate elastosis: a microscopic feature useful for the diagnosis of postatrophic hyperplasia. Arch Pathol Lab Med. 2000; 124: 1306-9.
- Galosi AB, Montironi R, Fabiani A, Lacetera V, Gallé G, Muzzonigro G: Cystic lesions of the prostate gland: an ultrasound classification with pathological correlation. J Urol. 2009; 181: 647-57.
- Ruska KM, Sauvageot J, Epstein JI: Histology and cellular kinetics of prostatic atrophy. Am J Surg Pathol. 1998; 22: 1073-7.
- van Leenders GJ, Gage WR, Hicks JL, van Balken B, Aalders TW, Schalken JA, et al.: Intermediate cells in human prostate epithelium are enriched in proliferative inflammatory atrophy. Am J Pathol. 2003; 162: 1529-37.
- Przybycin CG, Kunju LP, Wu AJ, Shah RB: Partial atrophy in prostate needle biopsies: a detailed analysis of its morphology, immunophenotype, and cellular kinetics. Am J Surg Pathol. 2008; 32: 58-64.
- Putzi MJ, De Marzo AM: Morphologic transitions between proliferative inflammatory atrophy and high-grade prostatic intraepithelial neoplasia. Urology. 2000; 56: 828-32.
- Wang W, Bergh A, Damber JE: Morphological transition of proliferative inflammatory atrophy to high-grade intraepithelial neoplasia and cancer in human prostate. Prostate. 2009; 69: 1378-86.
- De Marzo AM, Meeker AK, Zha S, Luo J, Nakayama M, Platz EA, et al.: Human prostate cancer precursors and pathobiology. Urology. 2003; 62(5 Suppl 1): 55-62.
- Montironi R, Mazzucchelli R, Scarpelli M: Precancerous lesions and conditions of the prostate: from morphological and biological characterization to chemoprevention. Ann N Y Acad Sci. 2002; 963: 169-84.
- Anton RC, Kattan MW, Chakraborty S, Wheeler TM: Postatrophic hyperplasia of the prostate: lack of association with prostate cancer. Am J Surg Pathol. 1999; 23: 932-6.
- Billis A, Freitas LL, Magna LA, Ferreira U: Inflammatory atrophy on prostate needle biopsies: is there topographic relationship to cancer? Int Braz J Urol. 2007; 33: 355-60; discussion 361-3.
- Postma R, Schröder FH, van der Kwast TH: Atrophy in prostate needle biopsy cores and its relationship to prostate cancer incidence in screened men. Urology. 2005; 65: 745-9.
- Mikuz G, Algaba F, Beltran AL, Montironi R: Prostate carcinoma: atrophy or not atrophy that is the question. Eur Urol. 2007; 52: 1293-6.
- Tomas D, Kruslin B, Rogatsch H, Schäfer G, Belicza M, Mikuz G: Different types of atrophy in the prostate with and without adenocarcinoma. Eur Urol. 2007; 51: 98-103; discussion 103-4.
- Billis A, Meirelles LR, Magna LA, Baracat J, Prando A, Ferreira U: Extent of prostatic atrophy in needle biopsies and serum PSA levels: is there an association? Urology. 2007; 69: 927-30.
- Billis A, Meirelles L, Freitas LL, Magna LA, Ferreira U: Does the type of prostatic atrophy influence the association of extent of atrophy in needle biopsies and serum prostate-specific antigen levels? Urology. 2009; 74: 1111-5.
- Oesterling JE, Lilja H: Prostate-specific antigen. The value of molecular forms and age-specific reference ranges. In: Vogelzang NJ, Scardino PT, Shipley WU et al. (ed.), Comprehensive Textbook of Genitourinary Oncology. Baltimore, Williams & Wilkins. 1996; pp. 668-80.
- Kumar V, Abbas AK, Fausto N: Robbins and Cotran Pathologic Basis of Disease, 7th ed. Philadelphia, Elsevier Sanders. 2005; pp. 3-46.
- Parsons JK, Nelson CP, Gage WR, Nelson WG, Kensler TW, De Marzo AM: GSTA1 expression in normal, preneoplastic, and neoplastic human prostate tissue. Prostate. 2001; 49: 30-7.
- Zha S, Gage WR, Sauvageot J, Saria EA, Putzi MJ, Ewing CM, et al.: Cyclooxygenase-2 is up-regulated in proliferative inflammatory atrophy of the prostate, but not in prostate carcinoma. Cancer Res. 2001; 61: 8617-23.
________
Accepted:
January 21, 2010
_______________________
Correspondence
address:
Dr. Athanase Billis
Anatomia Patológica, FCM, Unicamp
Caixa Postal 6111
Campinas, SP, 13084-971, Brazil
Fax: + 55 19 3289-3897
E-mail: athanase@fcm.unicamp.br