THE
ROLE OF ENDOLUMINAL ULTRASONOGRAPHY IN UROLOGY: CURRENT PERSPECTIVES
(
Download pdf )
SIRISH KONDABOLU (2), SARDAR A. KHAN (1), JENNIFER WHYARD (2), CHRISTOPHER DIBLASIO (1), MANASA AYYALA (2), SRINIVAS PENTYALA (1,2)
Departments
of Urology (1) and Anesthesiology (2), School of Medicine, State University
of New York,
Stony Brook, New York, USA
ABSTRACT
Endoluminal ultrasonography (ELUS) is a noninvasive diagnostic technique used in urology to image tubular structures of the urinary tract. Through advancements in technology, modern ELUS is able to create three-dimensional images, which provide valuable clinical information for the diagnosis and treatment of urologic disorders. The efficiency and accuracy of this technology is confirmed through validation studies using human and animal models. Although a relatively new method, the clinical application of this technique holds great promise in the field of endourology. The technology, advantages, limitations, validation studies, clinical applications, and future of ELUS are explored through this comprehensive review of current urologic literature.
Key
words: ultrasound; urinary tract; diagnostic imaging; ureteral
cancer
Int Braz J Urol. 2004; 30: 96-101
INTRODUCTION
The role of endoluminal transrectal ultrasound in the diagnosis and biopsy of prostatic diseases is well established. The pathology of infra-renal tubular and hollow structures such as the ureters, bladder and urethra is heavily reliant on contrast studies such as voiding cysto-urethrograms, retrograde urethrograms and retrograde pyelograms. Other radiological studies such as helical computed tomography (CT) and magnetic resonance imaging (MRI) studies may be additive to diagnostic urologic instrumentation. Advances in endoscopic instruments such as flexible cystoscopies and ureteroscopes provide complimentary visualization of intraluminal pathology. Three-dimensional diagnostic evaluations of urologic structures to study the pathology in the wall and the bed surrounding the tubular and hollow urologic anatomical structures are currently explored by advances in endoluminal ultrasonography (ELUS) techniques. This report reviews current advances in ELUS to diagnose urologic lesions in the lumen and extraluminal anatomical structures.
TECHNOLOGY OF ELUS
Endoluminal
ultrasonography is an imaging modality where flexible catheters with high
frequency transducers are inserted into a lumen endoscopically to image
the urethra, bladder, ureter, and renal pelvis (1-3). These high frequency
ultrasound transducers, which create very detailed images by providing
a greater axial resolution, are usually located at the tip of the ELUS
catheters and are used to image lesions in tubular and hollow structures
(4,5). A common frequency used in this procedure is 20 MHz as it distinguishes
between anatomical and pathological structures more accurately than lower
frequencies. However, in some superficial carcinomas, frequencies higher
than 20 MHz are more efficient as they provide a more exact staging (4,6).
Conventional ELUS is a catheter-based system
that produces two-dimensional (2D) cross sections of tubular structures
and useful visualization of tissues (5,7). However since anatomy is three-dimensional
(3D), 2D imaging is not an efficient method for diagnosticians to correctly
identify intraluminal pathology. In addition, 2D images represent a thin
plane at some arbitrary angle in the body and thus the image plane is
difficult to localize and reproduce for follow-up studies (8). For this
reason, it was essential to develop a system whereby these 2D images could
be reconstructed into longitudinal 3D or volume reconstructions, which
could be viewed and manipulated interactively by image-rendering techniques
(2,9). 2D images can be processed using PC-based image-analysis systems,
many of which make use of B mode, color Doppler and power Doppler imaging
(5,10). Specific software for 3D reconstruction of 2D images has become
feasible since the early 1990s. In 3D reconstruction, by adjusting the
image density threshold and changing the viewing angle by rotating the
image longitudinally, the lumen and wall morphology can be examined in
any projection (5,9). Unlike 2D images, these images can be rotated on
any spatial plane to demonstrate the volume dependent detail of the internal
lumen (9). This volume data allows evaluation of a specific point in space
from various orientations by rotating, slicing, and referencing the slice
to other orthogonal slices. The data also allows for new volume-rendering
displays that show depth, curvature, and surface images, which are not
available in conventional methods (11).
3D reconstruction is a new technique applicable
to endoluminal imaging. It provides many advantages over 2D imaging by
highlighting the spatial relation of anatomic structures, which cannot
be appreciated in conventional imaging (12). It is a valuable diagnostic
and research tool that is continually evolving. Although still in its
infancy, 3D ELUS is growing in popularity and accuracy as further technological
developments are being made to augment its clinical application and efficiency.
With the miniaturization of catheters, tranducers and advancement of imaging
systems, 3D ELUS might soon adjunct conventional imaging systems, such
as CT and MRI.
VALIDATION STUDIES
In vitro and in vivo studies that compare ELUS measurements with actual anatomic specimens of tubular and hollow structures have generally shown close correlations (4,13). To insure the success and accuracy of this technology, numerous validation studies were conducted with both animals and humans.
Animal
Experimentation
In one reported study, ELUS was performed
to image the wall of a pig urethra to compare the cross-sectional data
obtained by ELUS images and the actual anatomic cross sections. The results
showed high quality images detailing different anatomic layers of the
urethra. The accuracy of the study was later confirmed by histological
cross-sectional studies (4). In studies conducted by Goldberg et al.,
artificial stones and surgically created pseudopolyps were inserted into
nonvascular lumina and were successfully imaged and identified in animal
models using ELUS (13,14).
The efficiency of ELUS as a technique to
guide surgical procedures was also tested through animal validation studies.
Rivas et al., injected glutaraldehyde cross-linked collagen in an animal
model was used to test the therapeutic efficacy of collagen in the treatment
of urinary incontinence. Submucosal, intramucosal, and periadventitial
depths of collagen into the urethra and bladder walls in 2 pigs were studied
using ELUS. It was shown that ELUS aids in accurately identifying the
submucosal location of collagen injection and avoids dispersion of the
material (15). Preliminary ELUS validation studies in animal models demonstrate
the efficiency, accuracy, and feasibility of this technique for intraluminal
use in humans (13).
Human
Experimentation
Human validation studies revolve around
correlation of ELUS measurements with anatomical data and accurate differentiation
between normal and abnormal structures, which include embedded stones
and aberrant vessels (6). ELUS detected stones embedded in the renal parenchyma
and the mucosa of the distal ureter, which was confirmed postoperatively.
ELUS identified a tumor in the distal ureter and its depth, which was
confirmed by a biopsy. ELUS demonstrated a crossing vessel as the cause
for narrowing of the proximal ureter and an idiopathic ureteral stricture
(14). These validation studies in humans encourage potential of this technique
as a powerful diagnostic tool in urology.
ADVANTAGES OF ENDOLUMINAL ULTRASONOGRAPHY
ELUS
is a noninvasive diagnostic technique that has evolved rapidly over the
past 15 years and is now used in urology to image tubular and hollow structures,
diagnose urological disorders, and aid surgical procedures. ELUS may readily
provide useful imaging because of its safety, minimal invasiveness, and
relatively low cost (16). ELUS images are created in real time and avoid
radiation, which is especially important for pregnant patients (1,17).
With advances in ELUS instruments, this technology may replace or improve
current diagnostic techniques in urology.
There are many advantages to using ELUS.
It is used to image lesions in the urethra, bladder, ureter, and renal
pelvis (1). The high quality of ultrasound images allows clear visualization
of distinct anatomic layers and provides accurate representation of tubular
and hollow structures with volumetric and geometric validation (4,18).
A major advantage of this technique is its ability to image structures
beyond the lumen of the tubular structure or hollow viscus (19). In a
study conducted by Goldberg et al., ELUS was able to measure the wall
thickness and echotexture of the urethra, urinary bladder, ureter, renal
pelvis, bile ducts, small bowel, fallopian tubes, and the uterus (13).
Evaluation of the pathology and morphology of such tubular structures
with multiplanar and surface-rendered images allow physicians to obtain
valuable clinical information promptly (11).
ELUS is currently employed in the evaluation,
diagnosis, and staging of a wide range of urological abnormalities, such
as urinary incontinence. ELUS can be used to directly visualize the sphincter
mechanism and identify this condition (20). This technique is also used
to guide collagen injection, which is used to treat urinary incontinence.
ELUS accurately identifies the submucosal location of collagen injection,
avoids dispersion of the material, and augments the therapeutic efficacy
in the treatment of urinary incontinence (15). Through its technological
evolution, ELUS may be used to image the depth of tumors, provide 360°
cross-sections of structures and tissues, and convey more information
about the spatial relationships of anatomic structures (6,14,21). A novel
dimension in ELUS creates new clinical advantages of this technique. For
example, ELUS is used to diagnose stones in the ureter and renal pelvis,
locate crossing blood vessels that produce compression of the ureter,
diagnose tumors in the urinary bladder and ureter, diagnose lumen encroaching
pathology, assist in tumor staging biopsy guidance, distinguish between
embedded stones and aberrant vessels, guide intraluminal instruments,
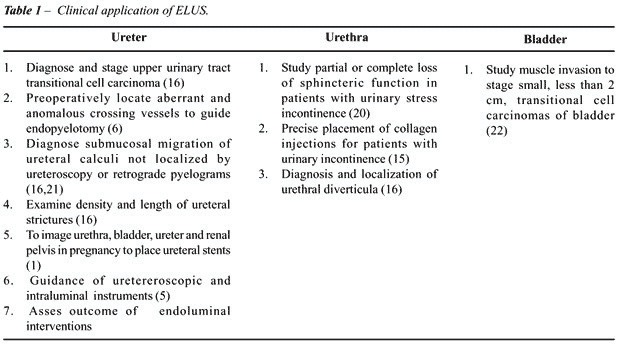
and distinguish superficial tumors from those with muscle invasion (2,6,15,22)
(Table-1).
ELUS
FOR FEMALE PATIENTS
Diagnosis and localization of urethral diverticula in females may pose a problem, as the urethral diverticulum can be easily misdiagnosed to be a redundant vaginal mucous (23). Female urethral diverticulum or its contents such as stones, endometriosis or tumors may present with diverse symptoms such as voiding dysfunction, pelvic pain, dyspareunia, incontinence, dribbling, and urethral discharge. Diverticula in females may be delayed or misdiagnosed, “mean interval between onset of symptoms to diagnosis was 5.2 years” (23). From the diagnosis point, urethral diverticula can be classified into 2 main groups. Female urethral diverticula whose ostia are open “open or communicating diverticula” where in the opening of the urethral diverticulum is wide and communicates with the main urethral lumen. Female urethral diverticulum whose ostia are closed, “closed or non-communicating diverticula” whose opening of the urethral diverticula is narrow or closed and is non-communicating with the main urethral lumen may present as cystic palpable mass on urethral examination. Pressure dependent urethral contrast studies may fail to diagnose and locate the closed urethral diverticulum, in these difficult cases ELUS may become diagnostic technique of choice and play a key role in the future (2). Currently MRI, transvaginal ultrasound have been reported to help diagnose, locate and study the contents of the suspected “non-communicating or closed” urethral diverticulum (24-26).
LIMITATIONS OF ENDOLUMINAL ULTRASOUND
Although miniaturized transducers guided ultrasound catheters are available, limitations of ELUS include restricted catheters flexibility, which may lead to noncoaxial or eccentric positioning of the intraluminal ultrasound probe, which may result in image artifacts, or loss of echo signals known as “echo drop outs”. In small or tortuous tubular lumens, malpositions of the ultrasound probes may preclude quantifications of lumen dimensions. Other limitations include invasiveness of the procedures with attendant risks of infection, luminal trauma, perforation, migration or ischemia due to lumen obstruction. Presence of gas in the diagnostic fields of bladder and ureter pose a hindrance to accurately visualize the lesions (16). In addition, the lack of penetration of sonographic beams in ELUS places major limitations on the evaluation of the depth of the invasion in large greater than 2 CMS bladder tumors with a broad base (22).
SUMMARY
Validation
studies consistently suggest that future advances in endoluminal ultrasound
technology are bound to improve and better this technique. In an attempt
to stretch high index of clinical suspicion of urologic diseases in the
realm of pathology and clinical urology, ELUS provides an opportunity
to exploit and enhance complimentary radiologic disparate disciplines
for diagnosis of difficult upper and lower urinary tract lesions.
ELUS may be useful to screen for early tumors
of tubular and hollow anatomical structures of the urinary tract to diagnose
screening of the upper and lower urinary tract in patients with refractory
positive urine cytology with negative results obtained by currently available
urologic instrumentation, contrast dependent studies or by other radiologic
imaging such as MRI or computed axial tomography (CAT) scan. Thickening
of the luminal wall or hollow viscus may provide an early answer to the
refractory positive cytology. Future advances in ELUS may compliment emerging
robotic techniques that are currently used in laparoscopic urology (27-30).
FUTURE OF ELUS
Endoluminal diagnostic ultrasound should be designed to provide urologists with an opportunity to integrate other diagnostic disciplines. Ideal diagnostic instrumentation should include fast, efficient, non-complicated instruments with short learning curve: 1) The diagnosis should be easily duplicated; 2) Outcome should carry an increased sensitivity and specificity when compared to the present technologic advances; 3) Early detection diagnosis of malignant lesions of the tubular and hollow structures of the urinary tract and clarity of imaging should be easily achieved; 4) Miniature catheter based ultrasound technique and effective instrumentation with excellent diagnostic yield should be readily available at a small cost; 5) The technique should carry minimal morbidity and mortality; 6) The technology can be safely used in patients with contrast allergies and in pregnant patients. 7) Accurately study small retroperitoneal vessel variations that cannot be imaged by current CAT scan or MRI 8. The tubular lumen of the ureter can be utilized to study and explore vessel morphology combined with duplex Doppler technology. Endoluminal ultrasound is still in its infancy, advances in transducer-loaded flexible miniaturized catheters and computer software holds great promise. Long-term multi-institutional studies to examine lesions of the upper and lower urinary tract may provide a reliable answer to difficult diagnostic lesions in the ureters, bladder and urethra.
REFERENCES
- Wolf MC, Hollander JB, Salisz JA, Kearney DJ: A new technique for ureteral stent placement during pregnancy using endoluminal ultrasound. Surg Gynecol Obstet. 1992; 175: 575-6.
- Goldberg BB, Liu JB: Endoluminal urologic ultrasound. Scand J Urol Nephrol.. 1991; 137 (suppl.): 147-54.
- Odegaard S: High-resolution endoluminal sonography in gastroenterology. Eur J Ultrasound. 1999; 10: 85-91.
- Messelink EJ, Dabhoiwala NF, Vrij V, Verbeek FJ, Kurth K: Endoluminal ultrasound of the urethra: a new modality for cross-sectional imaging of the urethra? Tech Urol. 1996; 2: 159-64.
- Cavaye DM, Tabbara MR, Kopchok GE, Laas TE, Cormier F, White RA: A new technique for intraluminal hollow organ imaging: three-dimensional ultrasound. J Laparoendosc Surg. 1991; 1: 259-68.
- Klotz T, Homann H, Mathers MJ, Vorreuther R, Engelmann U: (Value of endoluminal high-resolution ultrasound in urology). Ultraschall Med. 1994; 15: 198-201.
- Holmes DR, 3rd, Robb R: Trans-urethral ultrasound (TUUS) imaging for visualization and analysis of the prostate and associated tissues. Stud Health Technol Inform. 2000; 70: 126-32.
- Fenster A, Downey DB, Cardinal HN: Three-dimensional ultrasound imaging. Phys Med Biol. 2001; 46: 67-99.
- Campani R, Bottinelli O, Calliada F, Coscia D: The latest in ultrasound: three-dimensional imaging. Part II. Eur J Radiol. 1998; 27 (suppl 2): S183-7.
- Fenster A, Downey DB: Three-dimensional ultrasound imaging. Annu Rev Biomed Eng. 2000; 2: 457-75.
- Pretorius DH, Borok NN, Coffler MS, Nelson TR: Three-dimensional ultrasound in obstetrics and gynecology. Radiol Clin North Amer. 2001; 39: 499-521.
- Grotas A, Grasso M: Endoluminal sonographic imaging of upper urinary tract: three-dimensional reconstruction. J Endourol. 2001; 15: 485-8.
- Goldberg BB, Liu JB, Merton DA, Kurtz AB: Endoluminal US: experiments with nonvascular uses in animals. Radiology. 1990; 175: 39-43.
- Goldberg BB, Bagley D, Liu JB, Merton DA, Alexander A, Kurtz AB: Endoluminal sonography of the urinary tract: preliminary observations. AJR Am J Roentgenol. 1991; 156: 99-103.
- Rivas DA, Chancellor MB, Liu JB, Hanau C, Bagley DH, Goldberg B: Endoluminal ultrasonographic and histologic evaluation of periurethral collagen injection. J Endourol. 1996; 10: 61-6.
- Lee DI, Bagley DH, Liu JB: Experience with endoluminal ultrasonography in the urinary tract. J Endourol. 2001; 15: 67-74.
- Trinkler FB: Ultrasound examination of the urogenital tract: indications and limits. Schweiz Rundsch Med Prax. 2001; 90: 796-802.
- Chin JL, Downey DB, Elliot TL, Tong S, McLean CA, Fortier M, et al.: Dimensional transrectal ultrasound imaging of the prostate: initial experience with an emerging technology. Can J Urol. 1999; 6: 720-6.
- Liu JB, Miller LS, Bagley DH, Goldberg BB: Endoluminal sonography of the genitourinary and gastrointestinal tracts. J Ultrasound Med. 2002; 21: 323-37.
- Frauscher F, Helweg G, Strasser H, Enna B, Klauser A, Knapp R, et al.: Intraurethral ultrasound: diagnostic evaluation of the striated urethral sphincter in incontinent females. Eur Radiol. 1998; 8: 50-3.
- Liu JB, Goldberg BB: 2-D and 3-D endoluminal ultrasound: vascular and nonvascular applications. Ultrasound Med Biol. 1999; 25: 159-73.
- Horiuchi K, Tsuboi N, Shimizu H, Matsuzawa I, Kimura G, Yoshida K, et al.: High-frequency endoluminal ultrasonography for staging transitional cell carcinoma of the bladder. Urology. 2000; 56: 404-7.
- Romanzi LJ, Groutz A, Blaivas JG: Urethral diverticulum in women: diverse presentations resulting in diagnostic delay and mismanagement. J Urol. 2000; 164: 428-33.
- Hickey N, Murphy J, Herschorn S: Carcinoma in a urethral diverticulum: magnetic resonance imaging and sonographic appearance. Urology. 2000; 55: 588-9.
- Siegel CL, Middleton WD, Teefey SA, Wainstein MA, McDougall EM, Klutke CG: Sonography of the female urethra. AJR Am J Roentgenol. 1998; 170: 1269-74.
- Mouritsen L, Bernstein I: Vaginal ultrasonography: a diagnostic tool for urethral diverticulum. Acta Obstet Gynecol Scand. 1996; 75: 188-90.
- Stoianovici D: URobotics-Urology Robotics at Johns Hopkins. Comput Aided Surg. 2001; 6: 360-9.
- Rassweiler J, Binder J, Frede T: Robotic and telesurgery: will they change our future? Curr Opin Urol. 2001; 11: 309-20.
- Cadeddu JA, Stoianovici D, Kavoussi LR: Robotic surgery in urology. Urol Clin North Amer. 1998; 25: 75-85.
- Partin AW, Adams JB, Moore RG, Kavoussi LR: Complete robot-assisted laparoscopic urologic surgery: a preliminary report. J Am Coll Surg. 1995; 181: 552-7.
______________________
Received: March 5, 2004
Accepted: April 12, 2004
_______________________
Correspondence address:
Dr. Srinivas Pentyala
Department of Urology and Anesthesiology
HSC Level 4 Room 070
SUNY at Stony Brook,
New York, NY 11794-8480
Fax: 1 631 444-2907
E-mail: spentyala@anesthes.sunysb.edu