EPIDEMIOLOGICAL
ASPECTS OF PENILE CANCER IN RIO DE JANEIRO: EVALUATION OF 230 CASES
(
Download pdf )
LEANDRO KOIFMAN, ANTONIO J. VIDES, NELSON KOIFMAN, JOÃO P. CARVALHO, ANTONIO A. ORNELLAS
Department of Urology, Mario Kroeff Hospital (LK, AJV, NK, JPC, AAO), Department of Urology, Souza Aguiar Municipal Hospital (LK) and Department of Urology, National Cancer Institute (NK, AAO), Rio de Janeiro, RJ, Brazil
Clinical Urology
Vol. 37 (2):
231-243, March - April, 2011
doi: 10.1590/S1677-55382011000200010
ABSTRACT
Purpose:
To determine epidemiological characteristics of penile cancer in Rio de
Janeiro, its associated risk factors and clinical manifestations.
Patients
and Methods: Between 2002 and 2008 we evaluated 230 patients at three
public institutions, considering age, ethnicity, birthplace, marital status,
educational level, religion, tobacco smoking, presence of phimosis and
practice of circumcision.
Results:
The ages ranged from 25 to 98 years, with an average of 58.35 years. Of
the 230 patients, 167 (72.7%) were from the southeast region of Brazil
(which includes Rio de Janeiro) and 45 (19.5%) were from the northeast
of the country. Most patients were white (67.3%), married (58.6%), smokers
(56.5%) and had not completed primary school (71.3%). The predominant
religion was Catholic (74.8%). Of the 46 (20%) circumcised patients, only
1 (2.2%) had undergone neonatal circumcision. Grade I tumors were present
in 87 (37.8%) of the patients, grade II in 131 (56.9%) and grade III in
12 (5.3%). Lymphovascular embolization was observed in 63 (27.3%) and
koilocytosis in 124 (53.9%) patients. Of the total, 41.3% had corpora
cavernosa or corpus spongiosum infiltration, and 40 (17.4%) had urethral
invasion. Prophylactic lymphadenectomy was performed on 56 (36.1%), therapeutic
lymphadenectomy on 84 (54.2%) and hygienic lymphadenectomy for advanced
disease on 15 (9.7%) patients. The median time between the lesion onset
and clinical diagnosis was 13.2 months. The mean follow up was 28.8 months.
Conclusion:
Most of our patients were born in this state and had low socioeconomic
status. Most of them were white men, married, smokers, uncircumcised,
of the Catholic faith and in their sixties or older. Their disease was
in most cases diagnosed only in the advanced stages.
Key
words: penis; penile cancer; epidemiology
Int Braz J Urol. 2011; 37: 231-43
INTRODUCTION
Cancer
of the penis is a rare neoplasm whose treatment causes devastating effects
on patients’ physical and mental health. The low incidence of this
disease in developed countries in contrast with the high incidence in
developing countries clearly indicates the disease’s association
with local economic conditions (1). Some areas of Brazil have high incidences
of penile cancer, reaching about 17% of all malignant neoplasms in men,
thus constituting a serious public health problem (2).
The etiology of penile cancer has not been fully elucidated. However,
its incidence varies according to the practice of circumcision, personal
hygiene, presence of phimosis, human papilloma virus infection and tobacco
use (3-5).
Squamous
cell carcinoma represents approximately 95% of penile cancers. The remaining
5% of cases result from metastases from tumors in other organs or less
frequent tumor types, such as sarcomas, melanomas and lymphomas (6).
The
aim of this study was to assess the epidemiological characteristics of
penile cancer in the city of Rio de Janeiro, its associated risk factors
and clinical manifestations.
MATERIALS AND METHODS
Between
January 2002 and October 2008, 240 patients with malignant neoplasm of
the penis were evaluated at three public institutions in the city of Rio
de Janeiro. Of the 240 patients studied, 10 (3.9%) were excluded for lack
of histopathological data or clinical or epidemiological studies. Thus,
230 patients remained for analysis. All patients were evaluated using
the following epidemiological variables: age, ethnicity, birthplace, marital
status, educational level, religion, smoking, presence of phimosis, practice
of circumcision and clinical history of sexually transmitted diseases.
The clinical and pathological staging was done according to the latest
TNM classification system (2002). All patients underwent biopsy of the
primary lesion for diagnostic confirmation. Patients were clinically evaluated
for the presence of metastases by CT scan of the abdomen, pelvis and chest.
All patients were evaluated prospectively and gave their informed consent
to participate in the study. Our Institutional Review Board also approved
the study. The mean follow up was 28.8 months.
Pathological
material was reviewed and all tumors histologically classified based on
Broders system. Only two pathologists were responsible for reviewing the
specimens. The pathological variables studied were histological type,
grade, size of the lesion, corpus spongiosum and/or corpora cavernosa
infiltration, urethral infiltration, lymphovascular involvement, presence
or absence of koilocytosis (uni or binucleated cells and chromatin surrounded
by dark vacuolated cytoplasm).
We also evaluated the time between the onset of clinical symptoms and
diagnosis. The type of treatment for each patient was included in the
assessment. All patients who were indicated for adjunctive treatment of
inguinal lymphatic basins underwent radical bilateral inguinal lymphadenectomy.
We considered lymphadenectomy to be prophylactic when performed on patients
with clinically negative lymph nodes and high risk of inguinal dissemination
(PT2 and/or lymphovascular invasion and/or Broders histological classification
greater than or equal to II). We considered it to be therapeutic when
performed on patients with clinically positive inguinal lymph nodes. Finally,
we considered it to be palliative for patients with large ulcerated tumor
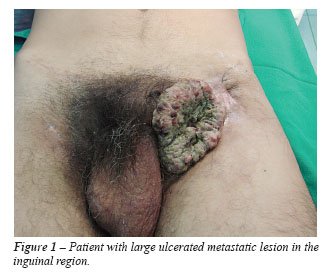
masses and/or masses fixed in the inguinal region (Figure-1).
Statistical
analysis was performed using One Way Analysis of Variance (ANOVA) with
Bonferroni post test for comparison between data. A p value < 0.05
was considered statistically significant. Linear regression was performed
when studying the stratification of tumor grade in comparison with tobacco
use and nonsmoking patients.
RESULTS
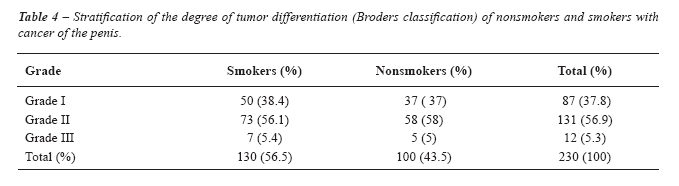
The patients’ ages ranged from 25 to 98 years, with a mean of 58.35 years (Table-1). Of the 230 patients evaluated, 155 (67.3%) were white, 55 (23.9%) mulatto and 20 (8.8%) black. The distribution of patients in relation to the birthplace is shown in Figure-2. Of the 230 patients, 167 (72.7%) were from the southeast region, 45 (19.5%) from the northeast, 6 (2.6%) from the north, 6 (2.6%) from the Midwest and 2 (0.9%) from the south of the country. Four (1.7%) patients were foreigners. Of the four foreign patients evaluated one came from Israel and other three from Portugal. In this series, 135 (58.6%) patients were married, 57 (24.7%) were single, 23 (10%) divorced and 15 (6.5%) widowed. The level of education ranged from illiterate, with 35 (15.2%) patients, to college graduates, with 8 (3.4%) patients. Of the remaining patients, 164 (71.3%) had not finished primary school and 23 (10%) were high-school graduates. The predominant religion was Catholic, with 172 (74.8%) patients, followed by various Protestant denominations, with 31 patients (13.5%). Only one patient (0.4%) was Jewish in this series and another 26 (11.3%) had various other religious beliefs.
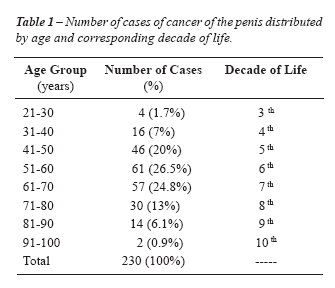

In
this series 130 (56.5%) patients were tobacco smokers and only 46 (20%)
patients had been circumcised. Among circumcised patients, 1 (2.2%) had
undergone neonatal circumcision, while 10 patients (21.7%) had been circumcised
in adolescence and 35 (76.1%) in adulthood. Of the circumcised patients,
25 (54.4%) had grade I tumors, 18 (40%) grade II tumors and only 3 (6.6%)
grade III tumors. Of the 230 patients evaluated, 31 (13.4%) reported history
of sexually transmitted diseases, 17 (54.8%) patients reporting a history
of urethritis and 14 (45.2%) of previous HPV infection.
In
relation to pathological variables studied, all patients present squamous
cell carcinoma of the penis. The lesion size ranged from 0.3 cm to 15
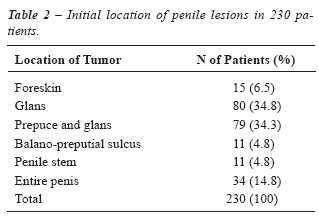
cm (mean 4 cm). The initial location of the lesions is shown in Table-2.
Based on Broders’ classification, 87 patients (37.8%) had grade
I tumors, 131 (56.9%) grade II and only 12 (5.3%) grade III. The clinical
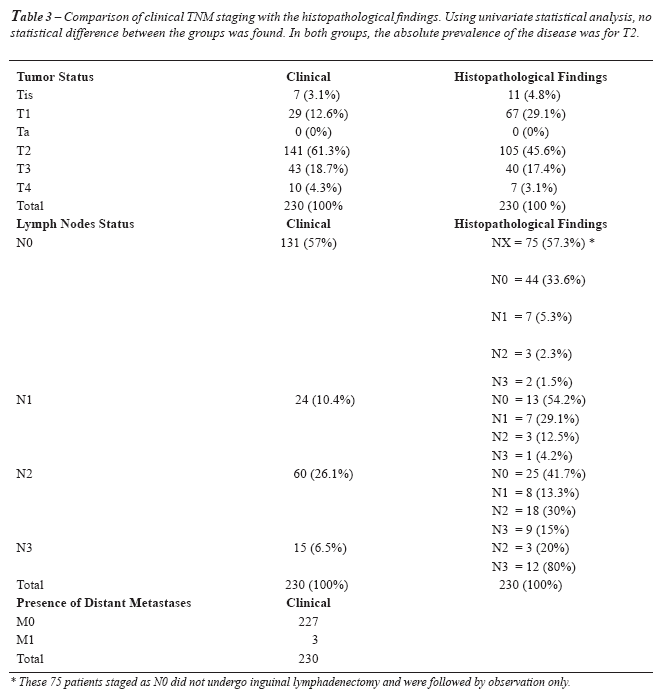
and pathological TNM classification is shown in Table-3. Of these patients,
95 (41.3%) had corpora cavernosa or corpus spongiosum infiltration and
40 (17.4%) had urethral invasion. Lymphovascular embolization was observed
in 63 patients (27.3%) and koilocytosis in 124 (53.9%). Only 3 patients
had lung metastases at diagnosis.
The
treatment for the patients varied according to the presentation of the
primary tumor. Six (2.6%) patients were treated with topical 5-fluorouracil
cream 5% due to the presence of carcinoma in situ, 15 (6.5%) patients
underwent circumcision due to lesions limited to the foreskin and 23 (10%)
patients were submitted to resection of the primary lesion of superficial
tumors less than 4 cm. Partial penile amputation was performed in 142
(61.8%) patients with tumors larger than 4 cm and/or signs of invasive
disease, while a total penectomy was performed in 34 (14.8%) patients
with extensive lesions and/or signs of invasive disease involving the
penile shaft. Only 10 (4.3%) patients underwent emasculation due to large
tumors with extensive involvement of the penile shaft and scrotum. Of
the 230 patients evaluated in this series, 155 (67.4%) underwent bilateral
inguinal radical lymphadenectomy to complement treatment of the primary
lesion. Of these, 56 (36.1%) underwent prophylactic lymphadenectomy, 84
(54.2%) therapeutic lymphadenectomy and 15 (9.7%) lymphadenectomy for
advanced disease palliation. The median time between the lesion onset
and clinical diagnosis was 13.2 months. After a mean follow-up of 28.8
months we observed a cancer-specific survival of 95.8%, 73.4%, 40% and
35.7% respectively for patients with lymph node status N0, N1, N2 and
N3.
COMMENTS
Cancer
of the penis is a rare neoplasm with low overall incidence. In the United
States, it accounts for approximately 0.4% of men malignancies. In Brazil,
despite the high incidence in some regions, this disease accounts for
about 2.1% of male malignancies. (2,7). The incidence of penile cancer
varies according to the study area, with its highest incidence reported
in the Northeast, representing approximately 5.7% of malignant neoplasms
in men (2). In our study we found that 167 (72.7%) patients were from
the Southeast, with 153 (91.6%) born in state of Rio de Janeiro and only
45 (19.5%) from the Northeast. A recent study by Favorito et al. (8) showed
the prevalence of penile cancer in the Southeast and Northeast, with rates
of 45.54% and 41.07%, respectively. Despite the large migration to the
Southeast, because it is the most developed economic region in the country,
in this study the incidence of the disease was more prevalent in patients
born in the state of Rio de Janeiro. These data suggest that many patients
with penile cancer receive specific treatment at their home states, with
a decline in the interstate migration.
When
the cancer of penis is present, it is prevalent in elderly men, with an
abrupt increase in incidence during the sixth decade of life and a new
peak around 80 years of age (9). In our series, we observed only 1.7%
of cases among patients aged between 21-30 years. The percentage increased
in the fifth decade of life (20%) and peaked in the sixth, with an incidence
of 26.5%.
The
practice of neonatal circumcision seems to be a protective factor in the
genesis of cancer of the penis (10). The incidence of penile cancer in
the Jewish population, where the practice of neonatal circumcision is
universal, is close to zero. In Muslim countries, where circumcision is
performed in childhood beyond the neonatal period, the incidence is up
to three times higher (11). In our study, the patients were predominantly
Catholic, representing 74.8% of all cases. Brazil is the largest Catholic
country in South America, explaining the high incidence of disease in
this religious group. There are only nine reports of penile cancer in
circumcised Jews in the neonatal period reported in the literature (12).
Interestingly, we had the opportunity to treat an Israeli Jewish patient,
who had undergone neonatal circumcision, with an advanced-stage tumor
(Figure-3).

Several
studies have shown an association between penile cancer and smoking. Hellberg
et al. found a relationship between penile cancer and smoking that was
direct, dose-related and independent of other known risk factors (12).
Harish and Ravi extended these observations by demonstrating that the
consumption of products made from tobacco is also related to the incidence
of penile cancer independent of other factors (13). In our series, we
observed a predominance of smokers, representing 56.5% of cases. In assessing
the degree of tumor differentiation between smokers and nonsmokers, we
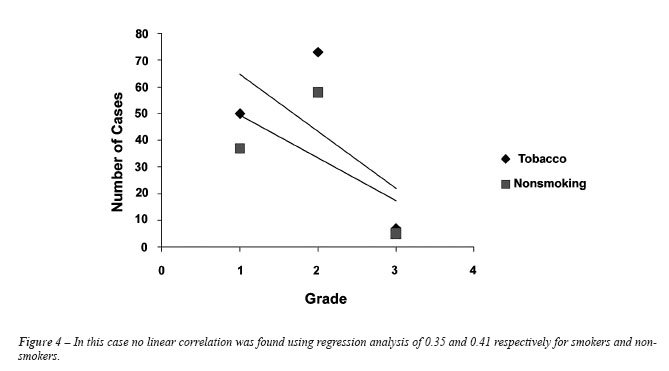
found no statistical difference between the 2 groups (Table-4 and Figure-4).
This study, despite not having used a control group, showed that more
than half of the patients with tumor of the penis were smokers, suggesting
that smoking may represent a risk factor for the development of penile
cancer. However, the degree of tumor differentiation may not be related
to smoking in this series.
An
interesting finding in this work concerns the marital status of patients:
58.6% were married and 24.7% were single. Since it was not possible to
determine any relationship between single marital status and sexual behavior,
we were unable to identify any predisposing factor for the genesis of
penile cancer related to marital status. It would be reasonable to imagine
a lower incidence of penile cancer in patients who were married that theoretically
would have a single sexual partner. It is possible that this finding is
coincidental and that the only factor associated with this observation
is the low economic level of patients and inadequate hygiene conditions,
which did not differ between married and single patients.
In the United States, a study by the National
Cancer Registries Program revealed an average incidence of 0.7 new cases
per 100,000 men in 2001. The incidence ranged from 0.8 for whites, 0.5
for blacks and 0.7 for Hispanics. Although some series have shown no racial
predisposition (14), Muir and Nectoux (15) observed a preponderance of
2:1 in black men. In Brazil, due to the great racial miscegenation is
hard to separate the patients by ethnicity since one cannot accurately
differentiate blacks, browns and whites. Some authors suggest a higher
likelihood of African-Americans to develop more aggressive forms of the
disease than white patients (16). In our series, only 20 patients (8.8%)
were black, and of these, twelve had grade 1 tumors, seven had grade 2
tumors and only one had a grade 3 tumor. Thus, we did not observe more
aggressive disease in black patients compared to whites.
The mechanism of tumor induction and promotion
related to human papilloma virus (HPV) infection is not completely understood.
It is believed that the incorporation of viral DNA to the human genome
leads to hyper-expression of E6 and E7 and inactivates the host cell’s
tumor suppressor gene products p53 and pRb (17). The identification of
HPV in specimens of penile tumors varies with the investigatory technique,
ranging from 30 to 100% (18-20). Using koilocytosis to detect the presence
of HPV in tumor tissue, we found positive readings in 124 patients (53.9%).
The cytological and histological diagnoses, despite showing good specificity
(90%), showed low sensitivity. Only 30% to 60% of patients with HPV infection
are correctly identified by these methods (17,21,22). In another study
conducted by our group, it was possible using a more sophisticated method
to detect HPV DNA in 75% of patients with invasive carcinomas (23).
Sexually transmitted diseases like herpes,
urethritis and syphilis have been implicated as a possible risk factor
for the development of penile cancer, however no convincing evidence was
found linking them to this disease (5,12). In our series 13.4% of patients
reported having had in the past at least one episode of sexually transmitted
diseases (STDs), 17 (54.8%) had urethritis and 14 (45.2%) presented HPV
infection. A possible explanation of the association between penile cancer
and STDs is the fact that the patients with STDs have a higher number
of sexual partners, increasing the likelihood of HPV infection.
Phimosis is considered an important risk
factor for the development of penile cancer, and is found in approximately
25-75% of patients with this cancer in the largest series (3-5). It has
been proposed that inadequate hygiene of the preputial sac with consequent
accumulation of smegma leads to a chronic local inflammatory process,
contributing to the genesis of penile cancer. In our study we found 68
patients (29.6%) with phimosis and 46 patients (20%) who had been circumcised.
Of these patients, 45 (97.8%) had undergone the procedure in childhood
or adulthood and only one (2.2%) in the neonatal period, corroborating
the literature data and suggesting the inefficiency of circumcision after
the neonatal period to prevent cancer of the penis (5,7,8,12,24). There
are few studies in the literature that correlate the degree of tumor differentiation
in the presence or absence of prior circumcision. Favorito et al. (8)
reported in their study that among 37 patients with squamous cell carcinoma
of penis circumcised before the appearance of the tumor, 31 had grade
1 tumor (83.8%), 2 (5.4%) had grade 2 tumor and 4 had (10.8%) grade 3
tumor. A study conducted by Seyam et al. (25) demonstrated that among
21 patients with squamous cell carcinoma of penis with a history of previous
circumcision, the incidence of grade 1, 2 and 3 tumors was respectively,
36.4%, 50% and 4.5%. In our series, more than half (54.4%) of our circumcised
patients presented grade 1 tumors. Although late circumcision does not
confer any protection against the squamous cell carcinoma of the penis,
its performance may be related to the development of less aggressive lesions.
One possible explanation is the exposure of the glans in a period that
precedes the appearance of the lesions, allowing for earlier identification
of tumor, and the elimination of chronic local irritating factors.
There is a strong association between the
clinical stage of the primary penile lesion and the development of inguinal
metastases. Involvement of the corpus cavernosum, the corpus spongiosum
and/or urethra are considered important risk factors, predisposing the
development of inguinal metastases in 61% to 75% of cases (1,26,27). Lymphovascular
embolization is also related to poor prognosis. In contrast, patients
who present koilocytosis have shown better survival (28). In this series
Lymphovascular embolization was observed in 63 patients (27.3%) and koilocytosis
in 124 (53.9%).
In our study we found that 152 patients
(66.1%) had invasive disease (pt2, pt3 and pt4) and the average time elapsed
between the lesion onset and clinical diagnosis was 13.2 months. This
long delay in diagnosis and treatment of patients is associated with poor
access to public health services and little available information about
the disease, reflecting the low socioeconomic level of patients most affected
by this disease.
The 2002 TNM classification for the staging
of tumors of the penis has been criticized by several authors (17,29-31).
Because it is essentially a pathological assessment it is virtually impossible
to clinically determine the precise level of tumor invasion and the real
lymph node status. In the study by Petralia et al. (30), physical examination
was able to properly stage the primary tumor in only eight of 13 patients
(61.5%), with overstaging in 2 (15.4%) and understaging in the other three
(23.1%) patients. Likewise de Kerviler et al. (32) only obtained a correct
clinical staging of penile lesions in 66.6% of patients in their series.
In our study we observed clinical staging accuracy of the primary tumor
in 75.2% of cases. When stratifying patients according to the primary
tumor, understaging was observed in 14.3% of patients with Tis and overstaging
in 17.2%, 29.8%, 13.9% and 30% respectively for T1, T2, T3 and T4 tumors.
Misinterpretation of the degree of tumor infiltration of the primary lesion
on physical examination could be attributed to local edema and infectious
processes that arise at tumor site.
The presence and extent of inguinal metastases
are the most important prognostic factors related to survival of patients
with squamous cell carcinoma of the penis (1,4,16,17). In our series,
of the 230 patients evaluated we found that 131 (57%) presented clinical
lymph node status N0, 24 (10.4%) were at stage N1, 60 (26.1%) were at
stage N2 and 15 (6 5%) were at stage N3. Despite the presence of clinically
positive lymph nodes in 43% of the cases, one must take into account the
inaccuracy of inguinal clinical staging, where under-staging errors of
up to 20% are observed in patients with lymph node status N0 and over-staging
in 50% of patients with palpable lymph nodes (33,34). In our series we
observed a failure leading to understaging in 21.4% of patients with clinical
N0 lymph node status. Overstaging occurred in 38.4% of patients with palpable
lymph nodes (Table-3).
CONCLUSION
Cancer of the penis is a rare neoplasm in Rio de Janeiro, mainly affecting patients born in this state and with low socioeconomic status. The epidemiological profile of these patients revealed that they were white, married, smoker, uncircumcised, Catholic and sixty or older. It was not possible to accurately determine the prevalence of HPV infection based only on detection of koilocytosis in tumor tissue. Poorer patients with less education tend to delay longer in seeking medical help, and therefore the diagnosis of the disease is frequently performed in the advanced stages.
CONFLICT OF INTEREST
None declared.
REFERENCES
- Solsona E, Algaba F, Horenblas S, Pizzocaro G, Windahl T; European Association of Urology: EAU Guidelines on Penile Cancer. Eur Urol. 2004; 46: 1-8.
- Brunini R: Câncer no Brasil: Dados histopatológicos: 1976-80, Ministério da Saúde - Campanha Nacional de Combate ao Câncer, 1982.
- Barrasso R, De Brux J, Croissant O, Orth G: High prevalence of papillomavirus-associated penile intraepithelial neoplasia in sexual partners of women with cervical intraepithelial neoplasia. N Engl J Med. 1987; 317: 916-23.
- Maiche AG: Epidemiological aspects of cancer of the penis in Finland. Eur J Cancer Prev. 1992; 1: 153-8.
- Maden C, Sherman KJ, Beckmann AM, Hislop TG, Teh CZ, Ashley RL, et al.: History of circumcision, medical conditions, and sexual activity and risk of penile cancer. J Natl Cancer Inst. 1993; 85: 19-24.
- Cubilla AL, Reuter V, Velazquez E, Piris A, Saito S, Young RH: Histologic classification of penile carcinoma and its relation to outcome in 61 patients with primary resection. Int J Surg Pathol. 2001; 9: 111-20.
- Parkin DM and Muir CS: Cancer Incidence in Five Continents. Comparability and Quality of Data. Lyon. IARC 1992; pp. 45-173.
- Favorito LA, Nardi AC, Ronalsa M, Zequi SC, Sampaio FJ, Glina S: Epidemiologic study on penile cancer in Brazil. Int Braz J Urol. 2008; 34: 587-91; discussion 591-3.
- Persky L: Epidemiology of cancer of the penis. Recent Results Cancer Res. 1977; 60: 97-109.
- Licklider S: Jewish penile carcinoma. J Urol. 1961; 86: 98.
- Tan RE: Observations on frequency of carcinoma of the penis at Macassar and its environs (South Celebes). J Urol. 1963; 89: 704-5.
- Hellberg D, Valentin J, Eklund T, Nilsson S: Penile cancer: is there an epidemiological role for smoking and sexual behaviour? Br Med J (Clin Res Ed). 1987; 295: 1306-8.
- Harish K, Ravi R: The role of tobacco in penile carcinoma. Br J Urol. 1995; 75: 375-7.
- Beggs JH, Spratt JS Jr: Epidermoid carcinoma of the penis. J Urol. 1964; 91: 166-72.
- Muir CS, Nectoux J: Epidemiology of cancer of the testis and penis. Natl Cancer Inst Monogr. 1979; 53: 157-64.
- Busby JE, Pettaway CA: What’s new in the management of penile cancer? Curr Opin Urol. 2005; 15: 350-7.
- Peclat de Paula AA, Neto JCA, Cruz AD, Júnior RF: Carcinoma epidermoide do pênis: considerações epidemiológicas, histopatológicas, influência viral e tratamento cirúrgico. Revista Brasileira de Cancerologia. 2005; 51: 243-52.
- Bezerra AL, Lopes A, Santiago GH, Ribeiro KC, Latorre MR, Villa LL: Human papillomavirus as a prognostic factor in carcinoma of the penis: analysis of 82 patients treated with amputation and bilateral lymphadenectomy. Cancer. 2001; 91: 2315-21.
- McCance DJ, Kalache A, Ashdown K, Andrade L, Menezes F, Smith P, et al.: Human papillomavirus types 16 and 18 in carcinomas of the penis from Brazil. Int J Cancer. 1986; 37: 55-9.
- Sarkar FH, Miles BJ, Plieth DH, Crissman JD: Detection of human papillomavirus in squamous neoplasm of the penis. J Urol. 1992; 147: 389-92.
- Trofatter KF Jr: Diagnosis of human papillomavirus genital tract infection. Am J Med. 1997; 102: 21-7.
- de Paula AA, Netto JC, Freitas R Jr, de Paula LP, Mota ED, Alencar RC: Penile carcinoma: the role of koilocytosis in groin metastasis and the association with disease specific survival. J Urol. 2007; 177: 1339-43; discussion 1343.
- Scheiner MA, Campos MM, Ornellas AA, Chin EW, Ornellas MH, Andrada-Serpa MJ: Human papillomavirus and penile cancers in Rio de Janeiro, Brazil: HPV typing and clinical features. Int Braz J Urol. 2008; 34: 467-74; discussion 475-6.
- Thomas JA, Small CS: Carcinoma of the penis in Southern India. J Urol. 1968; 100: 520-6.
- Seyam RM, Bissada NK, Mokhtar AA, Mourad WA, Aslam M, Elkum N, et al.: Outcome of penile cancer in circumcised men. J Urol. 2006; 175: 557-61; discussion 561.
- Slaton JW, Morgenstern N, Levy DA, Santos MW Jr, Tamboli P, Ro JY, et al.: Tumor stage, vascular invasion and the percentage of poorly differentiated cancer: independent prognosticators for inguinal lymph node metastasis in penile squamous cancer. J Urol. 2001; 165: 1138-42.
- McDougal WS: Carcinoma of the penis: improved survival by early regional lymphadenectomy based on the histological grade and depth of invasion of the primary lesion. J Urol. 1995; 154: 1364-6.
- Ornellas AA, Nóbrega BL, Wei Kin Chin E, Wisnescky A, da Silva PC, de Santos Schwindt AB: Prognostic factors in invasive squamous cell carcinoma of the penis: analysis of 196 patients treated at the Brazilian National Cancer Institute. J Urol. 2008; 180: 1354-9.
- Horenblas S, van Tinteren H: Squamous cell carcinoma of the penis. IV. Prognostic factors of survival: analysis of tumor, nodes and metastasis classification system. J Urol. 1994; 151: 1239-43.
- Petralia G, Villa G, Scardino E, Zoffoli E, Renne G, de Cobelli O, et al.: Local staging of penile cancer using magnetic resonance imaging with pharmacologically induced penile erection. Radiol Med. 2008; 113: 517-28.
- Leijte JA, Gallee M, Antonini N, Horenblas S: Evaluation of current TNM classification of penile carcinoma. J Urol. 2008; 180: 933-8; discussion 938.
- de Kerviler E, Ollier P, Desgrandchamps F, Zagdanski AM, Attal P, Teillac P, et al.: Magnetic resonance imaging in patients with penile carcinoma. Br J Radiol. 1995; 68: 704-11.
- Ornellas AA, Seixas AL, Marota A, Wisnescky A, Campos F, de Moraes JR: Surgical treatment of invasive squamous cell carcinoma of the penis: retrospective analysis of 350 cases. J Urol. 1994; 151: 1244-9.
- McDougal WS, Kirchner FK Jr, Edwards RH, Killion LT: Treatment of carcinoma of the penis: the case for primary lymphadenectomy. J Urol. 1986; 136: 38-41.
____________________
Accepted
after revision:
September 27, 2010
_______________________
Correspondence
address:
Dr. Antonio Augusto Ornellas
Department of Urology
Instituto Nacional de Câncer
Praça da Cruz Vermelha, 23
Rio de Janeiro, RJ, Brazil
E-mail: ornellasa@hotmail.com
EDITORIAL COMMENT
Koifman
et al. report on their experience with a relatively large prospective
series of men with penile cancer, a rare disease. The findings of this
contemporary series are interesting and useful for public health strategies.
So what is new on demographics and treatment of penile cancer? As it has
been previously shown, the Northern regions of Brazil have the highest
rates of penile cancer (1), and national prevention campaigns have focused
these regions. However, it has been observed that men treated in Rio de
Janeiro were mostly from Rio de Janeiro and not migrants as in past decades,
and therefore local campaigns are also important.
Maybe the most important aspect when treating men with penile cancer remains
inguinal nodes staging. Koifman et al. report about 10% of false negatives
and close to 50% false positive nodes. However, only when better staging
modalities become available can treatment become less aggressive. In this
series, all patients who were indicated for adjunctive inguinal treatment
underwent radical bilateral inguinal lymphadenectomy, what we see as a
good adjunctive approach. When modified procedures were described, initial
experience made us believe that they could be advantageous, but the possibility
of leaving disease behind has reduced interest for the modified procedures.
For this reason radical procedures seem to become a trend again in contemporary
series (2,3).
REFERENCES
1. Favorito
LA, Nardi AC, Ronalsa M, Zequi SC, Sampaio FJ, Glina S: Epidemiologic
study on penile cancer in Brazil. Int Braz J Urol. 2008; 34: 587-91; discussion
591-3.
2. Caso JR, Rodriguez AR, Correa J, Spiess PE: Update in the management
of penile cancer. Int Braz J Urol. 2009; 35: 406-15.
3. Korkes F, Moniz RR, Castro MG, Guidoni LRM, Fernandes RC, Perez MDC:
Modified inguinal lymphadenectomy for penile carcinoma has no advantages.
JAS. 2009; 16: 33-6.
Dr. Fernando
Korkes
Assistant Professor
Division of Urology, ABC Medical School
Av. Príncipe de Gales, 821
Santo André, SP, 09060-650, Brazil
E-mail: fkorkes@terra.com.br
EDITORIAL COMMENT
In
this epidemiological study by Koifman et al., the authors detail the prevalence
of penile cancer in 3 public hospitals within Brazil over a 6 year period.
Of the 230 patients with penile cancer described in this series, the majority
of patients originated from the southeast region of Brazil, with only
a small subset (2.2%) having undergone neonatal circumcisions. Phenotypically,
these patients presented in most instances with low to intermediate grade
tumors, with one-quarter of patients exhibiting lymphovascular invasion.
This study highlights that delay in diagnosis remains a major limitation
in the care of penile cancer patients, with the median time to diagnosis
being 13.2 months.
I
applaud the authors for their very insightful study. There are several
important clinical lessons learned from the present study. Firstly, teaching
and public education in the prevention, signs, and symptoms of penile
cancer remains a major limitation in optimizing the outcomes of this disease
on patients. This is probably the one area in which we as clinicians can
most greatly impact the care of our patients and in the community at large.
Secondly, I was somewhat surprised to note that only a small subset of
the patients within this study had high grade (Grade 3) tumors whereby
illustrating that there may in fact exist significant heterogeneity within
the pathophysiology of penile cancer worldwide as other series have reported
a greater proportion (typically 25-50%) of patients exhibiting higher
grade penile tumors. This may similarly impact our surgical approach to
penile cancer. As we embark in this new era of risk-adapted and personalized
cancer care, the goals of treatment have become to optimize oncological
outcome while preserving quality of life. In this regard, a highly selective
approach to penile preserving surgery and/or ablation for low grade/stage
primary penile tumors is gaining popularity and acceptance among penile
cancer experts. In contrast, a multimodal approach (typically using systemic
chemotherapy followed by surgery) for locally advanced and metastatic
penile cancer is rapidly becoming accepted as the most suitable approach
for advanced disease. In addition, targeted therapy (aimed at the EGFR
or other altered pathways) will likely redefine the therapeutic armamentarium
to advanced penile cancer in the coming years.
Dr. Philippe
E. Spiess
Assistant Professor of Urologic Oncology
H. Lee Moffitt Cancer Center
Tampa, FL, USA
E-mail: Philippe.Spiess@moffitt.org
EDITORIAL COMMENT
While
science is advancing at a tremendous speed and most research is focusing
on a molecular basis and treatment improvement including minimally invasive
resources (1) as well as new technologies for penile reconstruction, a
holistic view is fundamental, especially for diseases carrying obscure
facets such as the misunderstood behavior involved in penile cancer patients.
Though
the current work presents important limitations once data showed are deemed
to selection and measurement biases among others, this study has the potential
to highlight and put forward the main aspects to future studies including
interventional protocols.
Why over a year for a diagnosis in a much required (urinary, sexual and
reproductive functions), external and easily auto-examinable organ?
The
median time between the lesion onset and clinical diagnosis was 13.2 months
which involved most patients presenting invasive disease, diagnosed in
advanced stages, imposing mutilating and devastating treatments and certainly
impacting on quality and quantity of life.
Authors
have stated that the long delay in diagnosis and treatment of patients
is associated with poor access to public health services and little available
information about disease, reflecting the low socioeconomic level of patients
most affected by this disease.
This
is possibly the most alarming information presented and should be better
explored in future studies in a more comprehensive manner.
Previous
studies have identified that although there is strong evidence of an association
between lower socio-economic status and delay for urological cancers,
diagnosis and treatment non-recognition of symptom seriousness is the
main patient-mediated factor resulting in increased time to presentation.
Additionally, fear of cancer is a contributor to delayed presentation.
On the other hand, ‘misdiagnosis’ occurring either through
treating patients symptomatically or relating symptoms to a health problem
other than cancer is important and this could also be linked to inadequate
patient examination, use of inappropriate tests or failing to follow-up
negative or inconclusive test results (2).
For
penile cancer, while the major source of delay results from patient reluctance
to seek medical advice (3), approximately one-fifth of patients with penile
cancer are first referred to specialties other than Urology. This diagnostic
delay potentially affects the overall prognosis and thus, the greatest
impact in this condition is likely to be achieved by increased public
awareness and education.
In this regard epidemiological characteristics could be a small evident
part of something largely hidden, the clues to the tips of the iceberg
masked behind psychological and emotional aspects possibly structured
by a mixture of social and faith taboos.
This
is the (very high) cost of a miscellaneous of fear, ignorance and deep-rooted
taboos warranting further studies urgently. Bias and preconception related
to the penile cancer diagnosis and treatment are under recognized and
poorly understood; further studies are needed once the epidemiological
aspects suggest a great and important role for prejudice in penile cancer
dangerousness.
The
penis being a noble organ and cancer a serious illness, the fact that
penile cancer is neglected is a paradox. There is probably some mystic
significance for penis and cancer that could explain in part the upsetting
data shown. The over a year delay in diagnosis is conjectured to be related
to male and target organ factors once other urological cancers are more
readily diagnosed compared to penile cancer.
In
this scenario, as for prostate cancer (4), the discovery of the factors
behind it will serve to render the patient information more fundamental
towards a less deterministic and less self-destructive behavior. Added
to this is an endless behavior world to be unveiled as recognized recently
(5), opening a broad and under explored avenue in the study of penile
cancer.
Greek god Priapus teaches that the phallus is the source of life, beauty,
joy, and pleasure. The symbolic version of the phallus, a phallic symbol
is meant to represent male generative powers. Men are positioned as men
insofar as they are seen to have the phallus. The symbolic phallus is
the concept of being the ultimate man, and having this is compared to
having the divine gift of God.
Although
there is a worldwide geographic variation in incidence that could be caused
by differences in socioeconomic status, hygiene, religious and cultural
conditions, fear of mutilation or damage caused by the treatment, and
other misunderstanding about the possible causes of cancer, impede the
process of seeking testing for early diagnosis and treatment. Consequent
anxiety leads to distortion in communication, creating difficulty in the
comprehension of the information and recommendations, with detriment to
the doctor-patient relationship (4).
Future
studies should be concerned with the vital issue in human research: the
cultural scenario that clearly manifests global inequality and contrasting
behavioral differences around the world (6).
It highlights the need for individualized approaches to help men address
their thoughts and feelings about being diagnosed with urological cancers.
These efforts should include strategies that address cultural beliefs
and values related to temporal orientation.
More
research and public education are necessary, with information campaigns
addressing men’s emotional attitudes. It is our hope that by changing
perceptions, providing empathy, respect and focusing on the perception
of the person’s own body, it is possible that each individual could
find their own way to a satisfactory way of living, resulting in a better
quality of life, significantly impacting penile cancer.
REFERENCES
- Tobias-Machado M, Correa WF, Reis LO, Starling ES, Neves OC, Juliano RV, Pompeo ACL. Single-Site Video Endoscopic Inguinal Lymphadenectomy (SSVEIL): Initial Report. J Endourol. (in press).
- Macleod U, Mitchell ED, Burgess C, Macdonald S, Ramirez AJ: Risk factors for delayed presentation and referral of symptomatic cancer: evidence for common cancers. Br J Cancer. 2009; 101(Suppl 2): S92-S101.
- Lucky MA, Rogers B, Parr NJ: Referrals into a dedicated British penile cancer centre and sources of possible delay. Sex Transm Infect. 2009; 85: 527-30.
- Naccarato AM, Reis LO, Matheus WE, Ferreira U, Denardi F: Barriers to prostate cancer screening: psychological aspects and descriptive variables – is there a correlation? Aging Male. 2010; 12. [Epub ahead of print]
- Zequi SC, Guimaraes GC, da Fonseca FP, Lopes A, Matheus WE, Reis LO, et al. Sex with Animals (SWA): Characteristics and Possible Associations with Penile Cancer and Sexually Transmitted Diseases. Actual Results of a Multicentric Brazilian Case-Control Study. J Urol 2010; 183(Suppl 4): e8.
- Oliveira Reis L: Translating questionnaires for use in crosscultural research--are we doing the right thing?. Actas Urol Esp. 2009; 33: 5-7.
Dr. Leonardo
Oliveira Reis
Division of Urologic Oncology, School of Medicine
University of Campinas, UNICAMP
Campinas, SP, Brazil
E-mail: reisleo@unicamp.br