TRANSURETHRAL
MICROWAVE THERMOTHERAPY FOR BENIGN PROSTATIC HYPERPLASIA
(
Download pdf )
JONATHAN N. RUBEINSTEIN, KEVIN T. MCVARY
Department of Urology, Feinberg Medical School, Northwestern University, Chicago, Illinois, USA
ABSTRACT
Transurethral resection of the prostate (TURP) remains the gold standard for treatment of benign prostatic hyperplasia (BPH). In general, while this procedure is safe, patients require a spinal, epidural, or general anesthesia and often several days of hospital stay; the potential morbidity and mortality limits the use of TURP in high-risk patients. Pharmacotherapy has been recommended as a first-line therapy for all patients with mild to moderate symptoms. Patients are oftentimes enthusiastic if they are offered a one-time method to treat lower urinary tract symptoms secondary to BPH, provided that the method offers reduced risk and allows an efficacy equal to that of medical therapy. One such method is transurethral microwave thermotherapy (TUMT). TUMT involves the insertion of a specially designed urinary catheter with a microwave antenna, which heats the prostate and destroys hyperplastic prostate tissue. TUMT allows the avoidance of general or regional anesthesia, and results in minimal blood loss and fluid absorption. In this review, the authors discussed the current indications and outcome of TUMT, including the history of the procedure, the mechanism of action, the indications for TUMT, the pre-operative considerations, the patient selection, the results in terms of efficacy, by comparing TUMT vs. Sham, TUMT vs. Alpha-blocker and TUMT vs. TURP. Finally, the complications are presented, as well as other uses and future directions of the procedure. The authors concluded that TUMT is a safe and effective minimally invasive alternative to treatment of symptomatic BPH.
Key
words: prostate; prostatic hyperplasia; transurethral microwave
thermotherapy; transurethral resection of prostate
Int Braz J Urol. 2003; 29: 251-63
INTRODUCTION
Transurethral
resection of the prostate (TURP) remains the gold standard for treatment
of benign prostatic hyperplasia (BPH). In general, while this procedure
is safe, patients require a spinal, epidural, or general anesthesia and
often several days of hospital stay; the potential morbidity and mortality
limits the use of TURP in high-risk patients. Pharmacotherapy has been
recommended as a first-line therapy for all patients with mild to moderate
symptoms. Unfortunately, long-term outcomes are not fully elucidated,
patients must adhere to a strict medication schedule, and outcome indicators
are not reached as well or as reliably as TURP. Despite these inadequacies,
patients choose medications over surgery because of the perceived reduced
risk of adverse events and the desire to avoid surgery.
This trade-off of risk for efficacy is a
common thread running through all elective treatments for BPH. Newer modalities
have been aimed at providing alternatives to pharmacotherapy or watchful
waiting. Patients are oftentimes enthusiastic if they are offered a one-time
method to treat lower urinary tract symptoms secondary to BPH, provided
that the method offers reduced risk and allows an efficacy equal to that
of medical therapy. One such method is transurethral microwave thermotherapy
(TUMT). TUMT involves the insertion of a specially designed urinary catheter
with a microwave antenna, which heats the prostate and destroys hyperplastic
prostate tissue. TUMT allows the avoidance of general or regional anesthesia,
and results in minimal blood loss and fluid absorption. Clinical trials
in the United States and Europe have shown this modality to be safe and
effective, with excellent symptomatic relief seen in as little as one
outpatient setting using only local anesthesia. However, there is less
improvement in urinary functioning seen than with TURP, and long-term
follow-up data is not yet available. Clinical indications and treatment
parameters for TUMT are still evolving as technology advances and more
experience is gained. This manuscript summarizes current knowledge on
indications and efficacy of microwave therapy of the prostate.
PROCEDURE HISTORY
In
the 1980s, the use of heat to treat BPH regained clinical interest as
alternatives to TURP and open prostatectomy were being explored. The modern
use of microwaves has been credited to Yerushalami et al. (1). In 1982,
they performed microwave therapy on a patient with prostatic adenocarcinoma
(1) and later reported the therapeutic use of microwaves by the transrectal
route to treat patients with BPH who were poor operative candidates (2).
The first machines to undergo clinical trials
using hyperthermia employed a transurethral catheter in a series of ten
1-hour sessions. Software and instrumentation allowed only a limited and
often interrupted delivery of energy to the prostate, with intraprostatic
temperatures reaching 40-45ºC. Patients reported improved symptomatology,
likely due to destruction of the alpha-adrenergic nerve fibers around
the prostate, although an objective improvement of voiding parameters
was not observed (3), and prostatic cells were not destroyed.
To reliably destroy cells, temperatures
greater than 45ºC were necessary, which was coined “thermotherapy”
(4). Cells would slough away over a period of weeks to months. Unfortunately,
the urethral pain threshold was realized to be 45ºC. The introduction
of urethral cooling allowed these higher temperatures to be used. Although
heat treatment pattern differed from device to device, antennae were designed
to allow heat to generally follow the anatomical borders of the transition
zone. Both objective and subjective measures produced significant improvement.
However, patients invariably had severe prostatic edema and urinary retention,
requiring the use of a urinary catheter, which became standard practice
after a TUMT.
To improve outcomes even further, high-energy
thermotherapy was introduced. Temperatures greater than 70ºC were
reached, causing thermoablation of prostatic tissue. Today, several different
microwave devices are in use around the world, including the Targis (Urologix,
Inc., Minneapolis, Minnesota, USA), Prostatron (Technomed Medical System,
Lyons, France), Prostalund (Lund Instruments AB, Lund, Sweden), Prostcare
(Bruker Medical, Wissembourg, France), Urowave (Dornier MedTech America,
Kennesaw, Georgia, USA), PRIMUS U+R (Tecnomatix, Monheim, Germany), and
the LEO Microthermer (Laser Electro Optics, London, UK).
MECHANISM OF ACTION
TUMT uses an external power source creating microwaves at a frequency of 915-1296 MHz. Tissue penetration of microwaves leads to electromagnetic oscillations of free charges and the polarization of small molecules, such as water, resulting in the release of kinetic energy, which increases the temperature of the tissue. Cell necrosis, vascular injury, and apoptosis ensue. Urethral cooling in part protects the prostatic urothelium from these effects.
INDICATIONS FOR TUMT
It is widely agreed upon that patients with mild to moderate symptoms should be treated with medical therapy initially, reserving invasive therapies for those with more severe symptoms, those failing medical management, and for patients choosing invasive therapy over medical management. As the criterion standard for invasive therapy, TURP is offered to most patients. Initially, only patients with severe symptoms who were poor anesthetic risks were offered TUMT. Indications have now been expanded to include patients who prefer and outpatient setting rather than for a hospital stay due to the ease of use, minimal anesthesia requirement, and potentially rapid recovery.
PRE-OPERATIVE CONSIDERATIONS
Patient
Selection
A thorough clinical evaluation is vital
to assess the presence and degree of voiding dysfunction and/or the role
played by BPH. The patient’s past urologic history along with surgical
risks and concomitant medical problems need to be evaluated. A history
of sexually transmitted diseases, kidney stones, trauma, previous catheterizations,
genitourinary cancer, renal insufficiency, neurologic disease, and neurogenic
bladder may influence the treatment options. Medical conditions that may
influence bladder functioning mimicking BPH-type symptoms include diabetes
and neurological diseases. Surgical risks predominantly are due to renal
failure, coronary artery disease, and cerebrovascular disease. Medicines
containing alpha-sympathomimetics, such over-the-counter cold remedies,
enhance bladder outlet obstruction. A family history should focus on a
history of urologic cancer, and a social history should focus on risks
for cancer such as a smoking history and occupational exposure.
The physical examination should be systematic
and meticulous, focusing on the presence or absence of distended bladder,
urethral stenosis, meatal stenosis, and anal area and rectal tone. The
prostate is evaluated for its size, the presence or absence of nodularity,
laterality, consistency, and landmarks.
Laboratory
Studies
A hemoglobin determination and platelet
count are not specifically required prior to TUMT unless the patient has
a history of anemia, coagulation disorder, or as suggested by the history
and physical examination. Patients with a renal insufficiency (baseline
creatinine greater than 1.7 mg/dL) due to post-renal obstruction may benefit
more from a more definitive procedure such as TURP. A determination of
serum PSA level may be important in the evaluation of the absence of prostate
cancer. Unlike TURP, no specimen is submitted to pathology after TUMT.
Even with a normal PSA and negative biopsies, patients are at risk for
prostate cancer, which may be observed on the chips. If clinically suggested,
transrectal biopsies should be performed, as alternative therapies may
be suggested. Studies using baseline PSA as a predictive factor for TUMT
success are varied. In addition, all patients should have documented negative
culture before any urethral instrumentation to decrease the risk of urosepsis.
Imaging
Studies
A transrectal ultrasound (TRUS) is suggested
before performing TUMT, for patients with prostate volumes estimated to
be less than 25 cc or greater than 100 cc respond poorly to TUMT. Renal
ultrasound should be performed if there is a history of urinary retention,
have evidence of renal insufficiency, or there is hematuria.
Diagnostic
studies
A cystourethroscopy is mandatory to evaluate
the urethra for stricture disease, prostatic length, and degree of prostatic
hypertrophy. Patients with lateral lobe hypertrophy respond much better
to TUMT than those with middle lobe hypertrophy or a median bar, as marked
middle lobe enlargement distorts the heating pattern. The urethra and
bladder urothelium should also be evaluated for evidence of tumors and
stones. The location of the ureteral orifices should be noted.
The maximal voiding rate (Qmax) is a noninvasive
but nonspecific electronic recording of urinary flow rate. This is mainly
used to monitor response to treatment, although patients may have a weak
urinary stream due to an inadequate detrusor contraction rather than bladder
outlet obstruction. An adult man without evidence of obstruction should
have an average velocity of 12 cc/sec and an average peak velocity of
20 cc/sec. The prediction capacity of Qmax for outcome remains a matter
of debate.
Symptom indices (including the AUA score,
IPSS, Madsen quality of life, and Boyarsky) are available and are used
to confirm the components of the patient’s history, evaluate and
quantify the patient’s response to treatment, and to compare the
results of research protocols. Studies have failed to document a strong
correlation between symptom scores and physiologic changes due to BPH.
A post-void residual (PVR) volume is often
measured. While this does not predict surgical outcome, it may be used
to determine how closely patients need to be followed. Patients with high
PVRs have slightly higher rates of failure of watchful waiting and are
at an increased risk of complications such as urinary tract infections
and renal failure. A pressure-flow study should be performed in patients
whose voiding velocity and PVR measurements are not sufficient to determine
the presence or absence of bladder outlet obstruction or poor detrusor
contraction. This is the study of choice in patients with a history of
neurologic disease or other problems that affect the bladder and may confound
the diagnosis, and for patients with normal flow rates but bothersome
symptoms.
A cystometrogram (CMG) using either gas
or liquid may be of value for patients with known or suspected neurological
impairment. While it is less specific than pressure-flow studies, it allows
the evaluation of bladder capacity, evidence of uninhibited detrusor contractions,
and an estimation of bladder compliance. Patients who have adequate bladder
contractions have better outcomes after TUMT. A urethral pressure profile
measures pressures along the length of the urethra. In patients with known
or suspected urethral obstruction, this test helps determine the location
of the lesion. Finally, videourodynamics may be useful for patients with
complex causes of outlet obstruction or for cases where the sites of obstruction
need to be identified. Patients with neurogenic bladders may benefit as
well as patients undergoing an evaluation for incontinence.
HISTOLOGICAL FINDINGS AFTER TUMT
Bostwick & Larson (5) studied the results of TUMT on 3 dogs, showing that there was periurethral coagulative necrosis acutely, which began to resolve within 18 days. The capsule and urethra were deemed to be intact. Mauroy et al. (6) reported maximal coagulative necrosis at 8 days. Bdesha et al. (7) reported a significant decrease in alpha-1 adrenoreceptors after TUMT. Khair et al. (8) performed radical prostatectomies on at 7 days and 1 year later after TUMT, reporting that hemorrhagic necrosis and devitalized tissues without inflammation was observed in benign, stromal, and cancerous areas early on, with a mean volume of necrosis being 8.8 cc (average 22% of the tissue), but that only nonspecific chronic inflammation and a desquamous metaplasia with evidence of periurethral fibrosis remained at one year. The mean volume of necrosis remaining was 0.2 cc, which was less than 1%, implying that cells were sloughed away.
CONTRAINDICATIONS
All
patients undergoing transurethral procedures must have a documented sterile
urine culture and must be evaluated for prostate or urothelial cancer
if clinically suspected. Patients with neurogenic bladder voiding dysfunction
should have their underlying neurogenic problem evaluated and treated.
Contraindications specific to TUMT are evolving
as the technology changes and outcomes are studied further. Patients with
a history of TURP or pelvic trauma should not undergo TUMT because of
potential alterations in pelvic anatomy. Patients with glands that are
smaller than 25 gm. or a prostatic urethral length of less than 3 cm respond
poorly to TUMT, as do patients with glands greater than 100 gm or patients
with a prominent median bar.
Other contraindications include patients
with penile prosthesis, severe urethral stricture disease, Leriche syndrome/severe
peripheral vascular disease, or an artificial urinary sphincter. Patients
with pacemakers and defibrillators need clearance from their cardiologist
concerning turning their pacemakers off during therapy.
Hip replacement is no longer a contraindication.
Acute urinary retention was previously thought to be a contraindication
to TUMT; however, high-energy TUMT has shown to be promising in this population,
although efficacy has yet to be determined. Those presenting in retention
tend to be ill with greater co-morbidities; thus, they might benefit from
the less invasive nature of TUMT.
PREOPERATIVE DETAILS
In
preparation for TUMT, patients need to be counseled about the risks, benefits,
alternatives, and expectations of the therapy. Patients who have a urinary
catheter in place or had recent urinary tract manipulation should be placed
on appropriate antibiotics. The patient should have nothing by mouth for
6 hours prior to the therapy. An appropriate oral analgesic (such as ibuprofen,
ketorolac, or morphine) and an anxiolytic (benzodiazepine) may be administered
prior to the procedure.
The patient is brought to the therapy suite
and asked to void to completion. The bladder is emptied by straight catheterization,
and 40 cc of sterile water is placed within the bladder. 10-20 cc of 1-2%
Xylocaine gel is inserted within the urethra for anesthesia. The treatment
catheter is then placed within the urethra, confirmed by return of the
sterile water and by transabdominal or transrectal ultrasound, and the
balloon is inflated. This catheter has a curved tip, a temperature sensor,
and a microwave unit near the tip. The distal ports include those for
balloon inflation, urine drainage, coolant, microwave cable, and fiber
optic connector. The rectal probe (if used) is inserted and continuously
monitors the rectal temperature.
POSTOPERATIVE DETAILS
Out
patients and practice are such that all patients are catheterized following
TUMT. They return to the clinic for a trial of decatheterization, varying
by the protocol used. As prostatic edema is nearly universal after microwave
therapy, there are a high number of patients that fail the initial decatheterization
trial if performed too early.
Post-treatment convalescence is relatively
rapid, with most patients able to void in less than 3 days at home and
a mean recovery time of 5 days at home. Catheterization is required in
approximately 60% of patients after low-energy Prostatron 2.0 and 100%
of the high-energy Prostatron 3.5, and often this is extended for over
2 weeks. Patients are typically catheterized for a minimum of 2 days after
Targis thermotherpay. Patients with larger prostates are more prone to
catheterization because of increased edema. The slow process of improvement
is characteristic of high-energy transurethral microwave thermotherapy.
Coagulated tissue must be absorbed and the treated area must be reorganized
before sufficient voiding is achieved. Patients may notice an improvement
over a period of many months. Patients are advised to watch for the inability
to void, painful voiding, high fevers, abdominal pain, or other problems.
Some patients are maintained on alpha-blockade
for a period of time after TUMT. Studies have shown that these patients
have improved symptomatology early on over those not on alpha-blockade
(9), and also have a lower incidence of retention. Another option includes
the placement of a temporary prostatic bridge catheter, which is effective
and well-tolerated option that provides a better immediate peak flow,
IPSS, and quality of life compared to no stent if the patient has adequate
detrusor contraction (10,11).
RESULTS
Efficacy
Microwave thermotherapy has been around
long enough for both short and medium-term results to be reported (Table-1).
In general, patients have noticeable symptomatic improvements as shown
by a reduction in symptom scores, although complementary objective improvements
have not occurred as regularly (12-26). Long-term follow-up in patients
undergoing low-power TUMT reveals subjective symptom score improvement,
although for only limited duration in most cases; in the long-term, only
a minority of patients showed benefit from treatment. Retreatment rates
are substantial, reportedly 57-84% by 5 years. Using the low-energy Prostatron
Prostasoft 2.0, Ohigashi et al. (25) reported a stimated of 67% retreatment
rate with 5 years with only 11% “satisfied” with treatment,
while Tsai et al. (20) reported an overall retreatment rate of 84.4% in
5 years using Prostcare. In both of these studies, patients with high
peak flow rates (i.e. > 6.5 cc/sec) and short urethral lengths (i.e.
< 40 mm) had a lower risk of receiving additional treatments after
TUMT. Interestingly, in both studies, older patients (i.e. > 64 years
old) similarly had a decreased rate of retreatment. Keijzers et al. (24)
similarly reported a decreased risk of retreatment in patients with pre-treatment
maximal urinary flow greater than 10 cc/sec, Madsen symptom score less
than 15, post-void residual urine less than 100 cc, and age greater than
65 years.
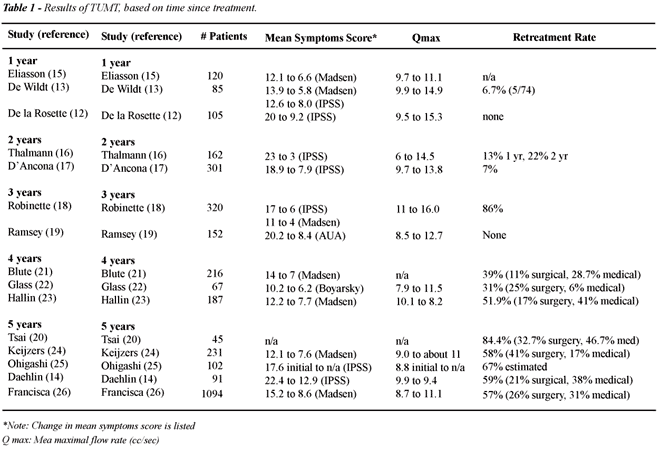
In comparison, higher energy protocols appear
to result in symptomatic improvement similar to that of lower energy protocols
yet the improvement in uroflowometry is much more pronounced. This appears
to be in trade-off for greater irritative symptomatology and longer duration
of catheterization (12). In patients with good initial responses to treatment,
which is achieved in approximately 80%, TUMT provides better long-term
subjective and objective results. Improved urinary flow, decreased post-void
residual urine volume and urodynamic parameters appear to remain stable
at 2 years. In a two-year follow-up using the Targis, Thalmann et al.
(16) reported that only 22% required additional treatments. In the remaining
patients, the IPSS decreased from an average of 23 to 3 at 6 months, while
the maximal flow rate increased from 6 ml/sec to 14.5 at 6 months. Both
results remained durable through 24 months. Post-void residual volumes
also appear to be markedly decreased after high-energy TUMT, as well as
demonstrable decreases in detrusor opening pressures and median detrusor
pressure at maximum flow rate (17). De la Rosette et al. (12) reported
that 6 months after treatment with the Prostasoft 3.5, patients had an
average decrease in the IPSS from 20 to 9.3, an increase in flow rate
from 9.4 cc/sec to 14.6 cc/sec, an average catheter time of 18 days, and
no serious complications.
TUMT
vs. Sham
Several small studies of randomized controlled
trials comparing TUMT versus sham treatments (Table-2) are available,
and generally show improvement of TUMT over sham. In the largest study
(220 men), Roehrborn et al. (27) reported a decrease in AUA symptom score
from 23.6 to 12.7 after TUMT, compared to 23.9 to 18 in the sham group,
while the Qmax increased from 7.7 to 10cc/sec and 8.1 to 9.8 cc/sec in
each group respectively. Blute et al. (28) and Larson et al. (29) similarly
reported a significant improvement in the TUMT group compared to sham
treatment using the Prostasoft 2.0 and Targis respectively. After reporting
their results, de Wildt et al. (30) commented that “a beneficial
effect of heating at thermotherapy temperatures is conclusive and further
sham studies would be unethical”. In contrast, neither Nawrocki
et al. (31) nor Mulvin et al. (32) showed any significant improvement
of TUMT over sham treatment in their studies using the Prostatron 2.0
and Targis respectively.
TUMT
vs. Alpha-blocker
When compared to alpha-blockade, TUMT is
associated with an initially poorer outcome but eventually better result.
Djavan et al. (33,34) prospectively studied 51 patients undergoing high-energy
TUMT and 52 patients on terazosin therapy. Terazosin appeared to have
a more rapid onset of action, and had its maximal effects observed at
6 weeks of therapy. In contrast, the maximal effect of TUMT was not observed
until after 6 months of therapy. Those patients on terazosin therapy similarly
were reported to have a more favorable change in the IPSS, peak flow,
and quality of life at 2 weeks compared to TUMT. However, by 1, 4, and
6 months, while there was a significant improvement in both cohorts, all
patients were more improved with TUMT. At 18 months, the IPSS, Qmax, and
quality of life was 35%, 22%, and 43% better in the TUMT group. Alpha-blocker
therapy was associated with more adverse events (17/52) compared to TUMT
(7/51). Patients on alpha-blockade complained of dizziness, asthenia,
headaches, and lack of effectiveness, prompting discontinuation in 11.5%
of patients. In addition, there was a sevenfold greater treatment failure
rate seen in the terazosin group.
TUMT
vs. TURP
There have been several trials comparing
TUMT and TURP (Table-2). In generally, these studies have shown TUMT to
be an effective modality, but in no way duplicated the results of TURP
in terms of objective or subjective parameters. For example, Ahmed et
al. (35) reported that 60% of a cohort of 30 patients reported symptomatic
improvement after TUMT, but none had objective relief of obstruction.
In comparison, 100% of the patients in the TURP cohort showed relief of
obstruction. At a longer follow-up of 2.5 years, d’Ancona et al.
(36) reported that TURP performed better than TUMT using both subjective
(Madsen score improvement of 76% after TURP compared to 56% after TUMT)
and objective (flow rates increased 105% after TURP compared to 62% after
TUMT) parameters. Floratos et al. (37) showed that these differences became
more pronounced with time, as patients undergoing TURP continued to improve
in both subjective and objective parameters after 1, 2, and 3 years, which
was not the case in the TUMT group. Further procedures were required in
19.8% of patients undergoing TUMT, compared to 12.9% of the TURP group
at 33 months.

In an attempt to bridge this gap, Wagrell
et al. (38) compared patients undergoing TURP to those undergoing TUMT
with the ProstaLund Feedback Treatment, which provides a feedback system
based on intraprostatic temperatures during treatment. In 133 patients
with a minimum of 12 months follow-up, good outcomes were reported in
82% and 86% of the patients in the TUMT and TURP respectively, and symptoms
score and maximal flow rate improvements appeared to be similar. Whether
or not this remains durable in the long-term remains to be seen.
Adverse effects of the 2 modalities vary.
In general, patients requiring further procedures after TUMT were due
to therapy failure, while after TURP was due to complications. Mild and
moderate adverse events were more common in the TUMT group, but serious
adverse events were more common (17% vs. 2%) in the TURP group. The complication
rate of TURP in these series varied, and was generally due to bleeding
and clot retention, urinary retention, urinary tract infections, urethral
strictures, and retrograde ejaculation. In contrast, patients undergoing
TUMT were at higher risk for severe urinary urgency, urinary retention,
and many required secondary procedures. The average catheter time was
3 days with TURP, 1-3 days with low-energy TUMT and 3-14 days using high
energy TUMT.
The reported rate of changes in sexual function
is 17% with TUMT compared to 36% with TURP, which is higher in older men.
One of the most common adverse effects is retrograde ejaculation. This
is reportedly observed in 48-90% of patients after TURP and 0-28% of patients
after TUMT, although even alpha-blockers are associated with a risk of
retrograde ejaculation. If explained prior to therapy, this complaint
is generally not a concern to most patients.
Arai et al. (39) reported a 26.5% rate of
erectile dysfunction for TURP and 18.2% with TUMT using a high-energy
protocol.Overall, satisfaction with sex life seems to be higher in patients
who have had TUMT than in patients who have had TURP, with patients undergoing
microwave thermotherapy reported as being “very satisfied”
in 55% versus 21% of TURP. However, only 27% of this population is satisfied
with their urinary flow after TUMT compared to 74% of patients who are
satisfied after TURP.
TUMT
in patients in urinary retention
Because patients presenting with urinary
retention generally are older, have a larger prostate volume, and have
more renal insufficiency, they are at increase anesthetic risk, increased
risk for secondary procedures, and risk for bleeding. In the past, TUMT
was thought to be contraindicated because of a high failure rate. However,
with the advent of high-energy TUMT, patients are now offered this less-invasive
therapy. Djavan et al. reported a 94% success rate at 12 weeks in 31 patients
presenting in retention (40), although the 1-year retreatment rate is
estimated to be 25% (41). Schelin reported that 80% of their cohort was
relieved of an indwelling catheter after TUMT, and those who failed all
had large median lobes or protruding lateral lobes into the bladder (42).
Robinette et al. (18) reported that 44 of 60 patients in retention were
able to void spontaneously at 6 months after therapy with the Prostatron
2.0 or 2.5, as were 32 of 35 followed at 12 months.
COMPLICATIONS
During
the procedure, patients commonly experience mild perineal warmth, mild
pain, and a sense of urinary urgency. However, only 5% of patients reported
their pain as being severe during Targis therapy. Despite this, more than
one half of these patients required substantial oral analgesics during
treatment. Higher energy protocols appear to have a slightly higher level
of initial pain due to the initial higher power, which appears to resolve
and return to the same level of comfort as the lower-energy protocols
soon into treatment.
Reports of complications vary, and range
from 0 to 38%, based on the study and the investigators’ criteria
for complications. For example, Ohigashi et al. reported no serious side-effects
in 91 patients treated with low-power over 5-year follow-up (25). Others
report complications including acute urinary incontinence, urinary tract
infection, and urinary retention. The risk for urinary tract infections
rises with each day of catheterization. In addition, the necrotic tissue
that remains in the prostatic fossa after TUMT may increase the risk of
colonization and infection. Treatment morbidity of higher energy protocols
is moderate and consists mainly of the need for catheterization and a
higher percentage of retrograde ejaculation (17).
Erectile dysfunction after TUMT is rare
if a patient is previously normal, but is commonly observed in patients
with prior erectile difficulties. Although causes have not been fully
elucidated, psychogenic factors, bladder neck trauma, and neurogenic voiding
dysfunction probably play a role. Lower-energy TUMT protocols have a lower
incidence of erectile dysfunction compared to higher-energy protocols
but at the expense of better urinary results. Francisca et al. (43) reported
no change in sexual performance after low-energy TUMT when compared to
a sham procedure in 147 patients.
A variety of other rare but reported complications
following TUMT occur. This includes, but is not limited to, urethrorectal
fistula (44), bladder perforation, and improper catheter placement. An
emphysematous prostatic abscess (45) has been reported after low-energy
TUMT in a 55-year old man with diabetes mellitus and cirrhosis. Proper
intra-treatment physician and nursing observation are vital to decrease
these risks.
OTHER USES AND FUTURE
DIRECTIONS
Microwave
therapy may be of value to treat other types of prostate pathology. For
example, several investigators are using TUMT to treat chronic prostatitis
(46). Microwave therapy is known to be lethal to many microorganisms because
microwaves are used to sterilize urinary catheters and surgical scalpels.
Patients with nonbacterial prostatitis who are non-responders to traditional
therapy may benefit from TUMT. Early results are promising, with a 25%
complete and sustained improvement and 50% mild improvement in a group
of 45 patients using TUMT.
In the future, because of the risk factors
for patients with symptomatic BPH, patients may be better stratified in
order to determine the optimal choice of therapies (i.e., pharmacotherapy
vs. TURP vs. TUMT vs. other method). Responders and non-responders may
be differentiated better by prostatic biopsy. The optimal combination
of preoperative medicines may allow for an increase in comfort. The optimal
time and energy requirements for therapy will decrease morbidity. The
long-term results of the balance between patient tolerability and efficacy
need to be evaluated adequately in a controlled setting.
Currently underway is the MIST trial, which
is a prospective, National Institute of Health-sponsored trial comparing
the outcomes of transurethral needle ablation - TUNA, TURP, or combination
medical therapy (alpha-blocker and 5-alpha reductase inhibitor). The end-points
and goals of this study are to evaluate the perioperative morbidity (i.e.
pain), complications, and long-term outcomes of these three options in
the treatment of BPH.
CONCLUSIONS
TUMT
is a safe and effective minimally invasive alternative to treatment of
symptomatic BPH. TUMT can be performed in a 1- to 2-hour office visit
without IV sedation. This is a good alternative for patients who are at
high surgical and anesthetic risk. It is not effective for patients with
a large median lobe or a very large prostate and results in less urinary
flow patterns than TURP.
Enthusiastic reassessment of procedures
that may reduce local and overall morbidity and maintain or improve immediate
and long-term physiologic results is understandable and laudable. Currently,
the limited number of patients, evolving selection and technical approaches,
hampers assessment of these efforts and the limited period and nature
of the follow-up information provided. In summary, this minimally invasive
therapy appears to balance efficacy against tolerability, and this balance
might be tenuous for patients in the long term.
REFERENCES
- Yerushalmi A, Servadio C, Leib Z, Fishelovitz Y, Rokowsky E, Stein JA: Local hyperthermia for the treatment of carcinoma of the prostate: a preliminary report. Prostate 1982; 3: 623-30.
- Yerushalmi A, Fishelovitz Y, Singer D: Localized deep microwave hyperthermia in the treatment of poor operative risk patients with benign prostatic hyperplasia. J Urol. 1985; 133: 873-6.
- Sapozink MD, Boyd SD, Astrahan MA: Transurethral hyperthermia for benign prostatic hyperplasia: preliminary clinical results. J Urol. 1990; 143: 944-9.
- Brehmer M, Svensson I: Heat-induced apoptosis in human prostatic stromal cells. BJU Int. 2000; 85: 535-41.
- Bostwick DG, Larson TR: Transurethral microwave thermotherapy. Pathologic findings in canine prostates. Prostate 1995; 26: 116-22.
- Mauroy B, Chive M, Stefaniak X, Demetriou D, Prevost B, Hattab B, et al.: Study of the effects of thermotherapy in benign prostatic hyperplasia. Eur Urol. 1992; 32: 198-208.
- Bdesha AS, Schachter M, Sever P, Witherow RO: Radioligand-binding analysis of human prostatic alpha-1 adrenoreceptor density following transurethral microwave therapy. Br J Urol. 1996; 78: 886-92.
- Khair AA, Pacelli A, Iczkowski KA: Does transurethral microwave thermotherapy have a different effect on prostate cancer than on benign or hyperplastic tissue? Urology 1999; 54: 67-72.
- Djavan B, Shariat S, Fakhari M: Neoadjuvant and adjuvant alpha-blockade improves early results of high-energy transurethral microwave thermotherapy for lower urinary tract symptoms of benign prostatic hyperplasia: a randomized, prospective clinical trial. Urology 1999; 53: 251-9.
- Djavan B, Fakhari M, Shariat S, Ghawidel K, Marberger M: A novel intraurethral prostatic bridge catheter for prevention of temporary prostatic obstruction following High-Energy transurethral microwave thermotherapy in patients with benign prostatic hyperplasia. J Urol. 1999; 161: 144-51.
- Djavan B, Ghawidel K, Basharkhah A, Hruby S, Bursa B, Marberger M: Temporary intraurethral prostatic bridge catheter compared to neoadjuvant and adjuvant alpha-blockade to improve early results of high-energy transurethral microwave thermotherapy. Urology. 1999; 54: 73-80.
- de la Rosette JJ, Francisca EA, Kortmann BB: Clinical efficacy of a new 30-min algorithm for transurethral microwave thermotherapy: initial results. BJU Int. 2000; 86: 47-51.
- de Wildt MJAM, Debruyne MH, de la Rosette JJMCH: High-energy transurethral microwave thermotherapy: a thermoablative treatment for benign prostatic obstruction. Urology. 1996; 48: 416-23.
- Daehlin L, Frugard J: Transurethral microwave thermotherapy in the management of lower urinary tract symptoms from benign prostatic obstruction: follow-up after five years. Scand J Urol Nephrol. 2000; 34: 304-8.
- Eliasson TU, Abramsson LB, Pettersson GT: Responders and non-responders to treatment of benign prostatic hyperplasia with transurethral microwave thermotherapy. Scand J Urol Nephrol. 1995; 24: 183-91.
- Thalmann GN, Mattei A, Treuthardt C, Burkhard FC, Studer UE: Transurethral microwave therapy in 200 patients with a minimum followup of 2 years: urodynamic and clinical results. J Urol. 2002; 167: 2496-501.
- d’Ancona FC, Francisca EA, Debruyne FJM, de la Rosette JJ: High-energy transurethral microwave thermotherapy for men with lower urinary tract symptoms. J Endourol. 1997; 11: 285-9.
- Robinette MA, Honey JD, Buckley RJ, Forder WC, Spevack LM, DiConstanzo JA, et al.: Results of transurethral microwave thermotherapy (TUMT) in patients presenting with: 1. Symptomatic BPH or 2. Urinary retention. J Urol. 1997; 157 (suppl), 375A.
- Ramsey EW, Miller PD, Parsons K: Durability of results with transurethral microwave thermotherapy for symptomatic benign prostatic hyperplasia (BPH) using the Targis (T3) system. J Urol. 1998; 159: 304A.
- Tsai YS, Lin JS, Tong YC, Tzai TS, Yang WH, Chang CC, et al.: Transurethral microwave thermotherapy for symptomatic benign prostatic hyperplasia: long-term durability with Prostcare. Eur Urol. 2001; 39: 688-92.
- Blute ML, Hanson K, Lynch J, Tomera K, Cope P, Sankey N, et al.: United States Prostatron TUMT study – 4 year follow-up and quality of life. J Urol. 1996; 155: 403 A, Abstract 370.
- Glass JM, Bdesha AS, Witherow RO: Microwave thermotherapy: A long-term follow-up of 67 patients from a single center. Brit J Urol. 1988; 81: 377-82.
- Hallin A, Berlin T: Transurethral microwave thermotherapy for benign prostatic hyperplasia: clinical outcome after 4 years. J Urol. 1998; 159: 459-64.
- Keijzers GH, Francisca EA, d’Ancona FC, Kiemeney LA, Debruyne FM, de la Rosette JJ: Long-term results of lower energy transurethral microwave thermotherapy. J Urol. 1998; 159: 1966-72.
- Ohigashi T, Babsa S, Ohki T, Nakashima J, Murai M: Long-term effects of transurethral microwave thermotherpay. Int J Urol. 2002; 9: 141-5.
- Francisca EA, Keijzers GB, d’Ancona FC, Debruyne FM, de la Rosette JJ: Lower energy thermotherapy in the treatment of benign prostatic hypertrophy: long-term follow-up results of a multicenter international study. World J Urol. 1999; 17: 279-84.
- Roehrborn CG, Preminger G, Newhall P: Microwave thermotherapy for benign prostatic hyperplasia with the Dornier Urowave: results of a randomized, double-blind, multicenter, sham-controlled trial. Urology 1998; 51: 19-28.
- Blute ML, Patterson DE, Segura JW, Tomera KM, Hellerstein DK: Transurethral microwave thermotherapy vs. sham treatment. Double-blind randomized study. J Endourol. 1996; 10: 565-73.
- Larson TR, Blute ML, Bruskewitz RC, Mayer RD, Ugarte RR, Utz WJ: A high-efficiency microwave thermoablation system for the treatment of benign prostatic hyperplasia. Results of a randomized, sham-controlled, prospective, double-blind, multicenter trial. Urology 1998; 51: 731-42.
- De Wildt MJ, Hubregtse M, Ogden C, Carter SS, Debruyne FM, de la Rosette JJ: a 12-month study of the placebo effect in transurethral microwave thermotherapy. Brit J Urol. 1996; 77: 221-7.
- Nawrocki JD, Bell TJ, Lawrence WT, Ward JP: A randomized controlled trial of transurethral microwave thermotherapy. Br J Urol. 1997; 79(3): 389-93.
- Mulvin M, Creagh T, Kelly D, Smith J, Quinlan D, Fitzpatrick J: Transurethral microwave thermotherapy vs. transurethral catheter therapy for benign prostatic hyperplasia. Eur Urol. 1994; 26: 6-9.
- Djavan B, Roehrborn CG, Shariat S: Prospective randomized comparison of high energy transurethral microwave thermotherapy versus alpha-blocker treatment of patients with benign prostatic hyperplasia. J Urol. 1999; 161: 139-43.
- Djavan B, Seitz, C, Roehrborn CG, Remzi M, Fakhari M, Waldert M, et al.: Trargeted transurethral microwave thermotherapy versus alpha-blockade in benign prostatic hyperplasia: outcomes at 18 months. Urology 2001; 57: 66-70.
- Ahmed M, Bell T, Lawrence WT, Ward JP, Watson GM: Transurethral microwave thermotherapy (Prostatron version 2.5) compared to transurethral resection of the prostate for the treatment of benign prostatic hyperplasic: a randomized, controlled, parallel study. Br J Urol. 1997; 79: 181.
- d’Ancona FC, Francisca EA, Witjes EP, Welling L, Debruyne FM, de la Rosette JJ: Transurethral resection of the prostate versus high-energy thermotherapy of the prostate in patients with benign prostatic hyperplasia: long-term results. Br J Urol. 1998; 81: 259.
- Floratos DL, Kiemeney LA, Rossi C, Kortmann BB, Devruyne FM, de la Rosette JJ: Long-term followup of randomized transurethral microwave thermotherapy versus transurethral prostatic resection. J Urol. 2001; 165: 1533-8.
- Wagrell L, Schelin S, Nordling J, Richthoff J, Magnusson B, Schain M, et al.: Feedback microwave thermotherapy versus TURP for clinical BPH—a randomized controlled multicenter study. Urology 2002; 60: 292-9.
- Arai Y, Aoki Y, Okubo K: Impact of interventional therapy for benign prostatic hyperplasia on quality of life and sexual function: a prospective study. J Urol. 2000; 164: 1206-11.
- Djavan B, Seitz C, Ghawidel K, Basharkhah A, Bursa B, Hruby S, et al.: High-energy randomized transurethral microwave thermotherapy in patients with acute urinary retention due to benign prostatic hypertrophy. Urology 1999; 54: 18-22.
- Floratos DL, Sonke GS, Francisca EA: High energy transurethral microwave thermotherapy for the treatment of patients in urinary retention. J Urol. 2000; 163: 1457-60.
- Schelin S: Microwave thermotherapy in patients with benign prostatic hyperplasia and chronic urinary retention. Eur Urol. 2001; 39: 400-4.
- Francisca EA, d’Ancona FC, Meuleman EJ, Debruyne FM, de la Rosette JJ: Sexual function following high energy microwave thermotherapy: Results of a randomized controlled study comparing transurethral microwave thermotherapy to transurethral prostatic resection. J Urol. 1999; 161: 486-90.
- Norby B, Frimodt-Moller PC: Development of a urethrorectal fistula after transurethral microwave thermotherapy for benign prostatic hyperplasia. BJU Int. 2000; 85: 554-5.
- Lin DC, Lin TM, Tong YC: Emphysematous prostatic abscess after transurethral microwave thermotherapy. J Urol. 2001; 166: 625.
- Liatsikos EN, Dinlenc CZ, Kapoor R, Smith AD: Transurethral microwave thermotherapy for the treatment of prostatitis. J Endourol. 2000; 14: 689-92.
________________________
Received: November 8, 2002
Accepted: December 5, 2002
_______________________
Correspondence address:
Dr. Kevin T. McVary
Dept Urology, Feinberg Medical School
Northwestern University, Tarry 11-715
300 E Superior St
Chicago, Illinois, 60611, USA
Fax: + 312 908-7275
E-mail: k-mcvary@northwestern.edu