COMPARISON
OF VASOVASOSTOMY WITH CONVENTIONAL MICROSURGICAL SUTURE AND FIBRIN ADHESIVE
IN RATS
(
Download pdf )
WILSON F. BUSATO JUNIOR, AMANDIA M. MARQUETTI, LUIZ C. ROCHA
Section of Urology (WFBJ), School of Medicine, UNIVALI and Catarinense Institute of Urology (WFBJ, AMM), Itajai, Santa Catarina, Section of Urology (LCR), School of Medicine, Federal University of Parana, Curitiba, Brazil
ABSTRACT
Objective:
Microsurgical procedures are currently the gold standard for vasovasostomy
with excellent results, but it takes an increased operative time demanding
special training and experience in microsurgery. The objective of this
study is to reach the same results with reduced operative time.
Materials and Methods: Male adults Wistar
rats were divided into 3 groups: I) 20 rats in control group, II) 20 with
conventional one-layer microsuture and III) 20 with fibrin glue. After
four weeks each rat was left for two weeks with two female rats.
Results: The fertility rates were 80% in
group I, 70% in group II and 65% in group III (p > 0.05). In group
II granuloma was found in 75% of the rats, while in group III in 85% (p
< 0.05). Anastomosis was considered with patency in 75% and 80% of
the rats in groups II and III (p > 0.05). Overall operative time was
41.7 ± 2.49 and 28.55 ± 1.14 minutes in groups II and III
(p < 0.05) and the time to anastomosis of 24.6 ± 1.8 and 9.35
± 0.78 minutes (p < 0.05), respectively.
Conclusions: We can conclude that vasovasostomy
with fibrin adhesive has the same results of the conventional microsurgery
technique but with a reduced operative time and a simplified procedure.
Key
words: rats; vasovasostomy; fibrin tissue adhesive; microsurgery;
infertility
Int Braz J Urol. 2007; 33: 829-36
INTRODUCTION
Currently,
more than 30 million couples worldwide use vasectomy as a method of birth
control. It is estimated that up to 6% of men who undergo vasectomy will
request reversal, and 3.5% will in fact receive a vasovasostomy (1,2).
In the United States, from 600,000 to 1 million vasectomies are performed
annually, which results in the expectation of 21,000 to 35,000 reversals
(3,4).
A number of techniques have been described
to restore vas deferens patency, but no consensus has been reached as
to which technique is the best (5). Several techniques using microanastomosis
show good results, with respect to both anastomosis patency and pregnancy
rate, between 80 to 90%, and 50 to 80%, respectively (5-11). Thus, the
main objective of this study is to devise a new technique or to improve
an existing one that would allow a faster procedure (12,13).
Advances in welding of human tissues have
been incorporated in experimental animal studies (14-16). This study was
undertaken to prospectively compare the use of conventional suture with
the fibrin glue technique in vas deferens reanastomosis, and to determine
if these techniques present similar results but with a reduced procedure
time and ease of execution.
MATERIALS AND METHODS
Sixty
male Wistar rats (Rattus norvegicus Albinus, Rodentia, Mamalia) initially
weighing between 220 to 230 grams and aged 90 days were included in the
experiments. To confirm fertility, 120 90-days-old female Wistar rats
in proestrus, determined by the analysis of vaginal secretions, were used.
The ethical guidelines on animal experimentation of the National Council
for Control of Animal Experimentation of Brazil were observed. The animals
were grouped as follows:
Group I - 20 (twenty) male rats that underwent
exposure of the left vas deferens for 5 to 10 minutes. Group II - 20 (twenty)
male rats presenting left vasovasostomy with a conventional one layer
suture with 10-0 nylon and Group III - 20 (twenty) male rats that underwent
a left vasovasostomy with fibrin glue.
Surgical Technique - The animals were anesthetized
intraperitoneally with sodium pentobarbital and the procedures were performed
under aseptic conditions. The surgery started after the loss of corneal
reflex. A bilateral incision was made in the scrotum, the vasa deferentia
were identified and isolated in all rats. In the three groups, the vasectomy
was performed on the right side.
In group I, the vas deferens was released
and the skin was sutured using 4-0 chromic catgut. In group II, the left
side vas deferens was divided, followed by a one-layer anastomosis with
five sutures using 10-0 nylon, according to the technique described by
Leonard & Thomas (17), with surgical microscope at 16 to 25X magnification.
In group III, on the left side the cut ends were reattached through a
transmural suture with one anchor points using 8-0 nylon suture and application
of 1 to 2 drops of fibrin glue..
The animals were kept in individual cages
for 30 days. At the end of this period, every male was put in a cage together
with two females for 2 weeks. After this period the males were anesthetized
and underwent a scrotal exploration. At the completion of the experiment,
the animals were euthanized with sulphuric ether in a bell jar.
The criteria for data collection and analysis
were fertility rate determined by the pregnancy of at least one female,
granuloma formation at the site of the anastomosis, amount of neutrophilic
exudate at the cut edges of the anastomosis, epithelial alterations (denaturation
and tearing), duration of surgery and anastomosis, and anastomosis patency.
The last one was evaluated immediately after the removal from the anastomosis
site in three ways: 1) observation through surgical microscope of intravasal
fluid produced by manual compression (milking) next to the anastomosis,
2) microscopic examination at 40 X 3.3 magnification of the fluid collected
from the distal end of the vas deferens with a glass slide to determine
the presence of sperm, and 3) catheterization of the proximal end at the
anastomosis site with 24-gauge cannula and gravity injection (70 cm height)
of methylene blue diluted in saline to 50% (18). In order to consider
the anastomosis patent to the passage of sperm, at least two criteria
were met.
The Z-test with 95% significance level was
used in the statistical analysis of the fertility rate, the granuloma
formation and the patency rate. The standard deviation was calculated
for the duration of the surgery and anastomosis. The exact Fischer’s
test was used in the correlation between the presence of granulomas and
the patency rate, and in the evaluation of neutrophilic infiltrate. The
χ2 test incorporating Yates correction for continuity
was used in the analysis of the reepithelialization and collagen reorganization.
RESULTS
Four
animals presented infection of the surgical wound, determined by the direct
inspection of the suture line; two animals belonged to group I, one to
group II and another to group III. One of the rats from group I showed
dehiscence of the skin suture, without infection. This rat (rat G1-4)
impregnated two females.
Fertility rate - Some females became pregnant
before four weeks were completed, the others were removed on the thirtieth
day and their pregnancies were confirmed thereafter. Seventeen female
rats (85%) from group I, 14 (70%) from group II and 13 (65%) from group
III became pregnant (p > 0.05).
Granulomas - Only the rats from group II
and III were evaluated. The total amount of macro- and microscopic granulomas
found in every rat was larger in the group that was vasovasostomized with
fibrin glue (Figure-1). Fifteen rats (75%) from group II had from 1 to
7 granulomas with an average of 3.6 granulomas/rat. Seventeen rats (85%)
from group III had from 2 to 8 granulomas with an average of 4.2 granulomas/rat
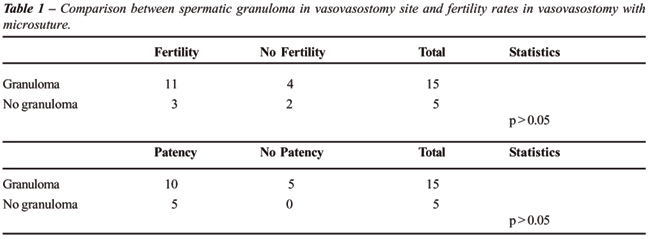
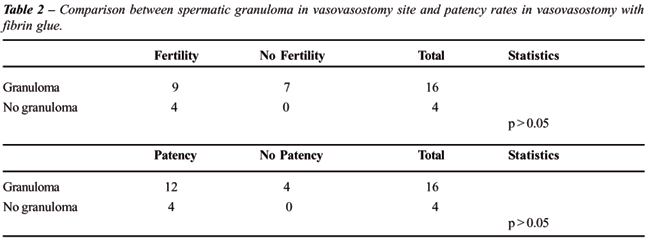
(p < 0.05). The anastomotic patency was not correlated with the presence
of granulomas in group II (Table-1) and group III (Table-2).
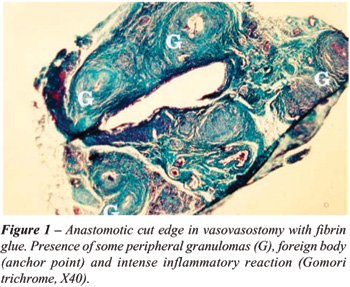
Histology - The evaluation of neutrophilic
exudate related to an inflammatory process at the anastomosis cut edges
can be seen in Table-3. The statistical comparison showed no significant
differences (p > 0.05) between vasovasostomy with conventional suture
and with fibrin glue. Intense neutrophilic exudate did not alter significantly
anastomosis patency, which suggests that the neutrophilic inflammatory
reaction is not an obstructive factor to the reanastomosed lumen (Figure-1).

The reepithelialization process of the vas
deferens lumen for groups II and III showed no significant difference
(p > 0.05). In same way, collagen fiber reorganization showed no significant
difference (p > 0.05).
Patency - Anastomotic patency was demonstrated
in 50 rats. Of these, 19 (95%) were in the control group, 15 (75%) in
the vasovasostomy-with-conventional-suture group, and 16 (80%) in the
fibrin-glue group (Table-4).

Surgery Duration - There was a statistical
significant difference in operative time when the different surgical procedures
were compared. Anastomosis with fibrin glue took less time to be performed
when compared to the conventional suture technique (Table-5).

COMMENTS
There
is no uniformity as to which is the best vasovasostomy technique (19,20).
Initially, the controversy was about the use of sutures in one or two
layers. The analysis of these two possibilities in the literature shows
similar results with respect to fertility and patency rates (8,10,21,22).
The fundamental difference is related to operative time, required training
and costs. In this study, the reanastomosis were performed immediately
after vasotomy and in vivo experience usually reveals a significant discrepancy
in lumen sizes, hence, the theoretical reason for 2-layer closures. When
both methods are compared, one would opt for the faster and easier method
to be performed (11).
Based on these reasons, anastomosis with
fibrin glue was compared to the one-layer technique with mucosa-to-mucosa
alignment. However, it has been questioned the need of a few stitches
before the fibrin glue application in order to keep the cut ends in alignment.
It was decided to use two transmural anchor points in order to improve
the vasovasostomy outcome, as previously described (23).
Conventional anastomoses that use microsurgical
techniques show high rates of success, with patency rates of about 90%
and fertility rates between 40 and 80% (12). Kücükaydin et al.
(24) obtained fertility rates of 70% for the fibrin glue or laser vasovasostomy
and 80% in control groups. In this experiment, we obtained a fertility
rate of 70% for the conventional suture group and 65% for the fibrin glue
group.
The presence of granulomas at the vasovasostomy
site still does not have a conclusive meaning (25). Granulomas are described
in 20 to 35% of the ducts in vasovasostomies (26). Silverstein et al (12)
report granulomas in 28% of the fibrin glue vasovasostomies, but they
observed a higher rate (61%) with conventional suture technique. These
results are similar to the ones found in this study where 30% of the rats
with anastomosis with fibrin glue and 15% with conventional suture presented
macroscopic spermatic granulomas. The most plausible explanation for a
larger number of granulomas when the fibrin glue was used is the amount
of glue that was applied. The excess of glue surrounding the anastomosis
can lead to granuloma formation. An experiment in dose titration could
clarify this question.
Some authors have emphasized the importance
of a patent anastomosis that prevents sperm leakage (23,24,26). The use
of fibrin glue guarantees the anastomotic patency, since the glue contacts
the full circumference of the vas deferens and not only 4 or 6 points
as in the conventional suture technique. However, it is important to note
that negative results can occur if the glue is applied to the two ends
of the vas deferens before they are reconnected.
At present, the majority of studies demonstrate
that anastomosis patency is not altered by the presence of granulomas
(18,27), this data is similar to what was observed in this experiment,
since no correlation was found between the presence of granulomas or their
number with an obstruction of the anastomosis.
It was observed that the presence of sperm
in the distal vas segment occurred in 75% of the anastomosis with conventional
suture and in 80% of the ones with fibrin glue. These results are similar
to those reported in literature, which ranges from 80 to 90% (12,16,23,24).
The vas deferens is lined internally by
a pseudostratified epithelium, composed mainly of small basal hemispherical
cells and columnar principal cells with microvilli (28). The exact physiological
role of the vasa deferentia in men is not completely understood, but it
is expected that after a reconstruction of the vasa one obtains a histological
evaluation as similar as possible as it was observed before the vasectomy.
We observe that the majority of vasa deferentia from the conventional
suture and fibrin glue groups showed lack of or initial epithelialization,
60 and 50% respectively. In spite of the fact that there was no statistical
difference showing unequivocally that the highest pregnancy and patency
rates are related to the ducts that showed a higher degree of reepithelialization,
it was observed at least a trend in this direction. It is noteworthy to
mention that the evaluation at 45 days may partially explain these results.
Although approximately 20% of the original
mass of fibrin glue is dissolved by fibrinolysis within 72 hours following
the application, a local inflammatory reaction may be observed. This tissular
reaction is a result of the combination of trauma and the physical and
chemical properties of the materials used in the synthesis (29). The permanence
of neutrophils indicates a persistent inflammatory process. It was detected
a predominance of a moderate inflammatory reaction in the anastomosis
with conventional suture and in the ones with fibrin glue, 90 and 85%
respectively. Similar results have been reported in the literature related
to the evaluation of inflammatory reactions in surgical wounds (30). Also,
the number of neutrophils at about six weeks may represent a response
to the surgical trauma (12,23,24,29,31). The inflammatory process, as
a result of the surgical manipulation and of the synthesis material, did
not seem to affect the anastomosis patency. Few studies in the literature
make this correlation, but those that evaluate it do not believe in cause-effect
relationship either (28).
In the functional analysis of vasovasostomy
we need to keep in mind that there are several factors that may alter
the patency and not only the quality of the anastomosis itself. The denervation
of the vas deferens during the vasectomy may halt the peristaltic propulsion
of sperm through the anastomosis. Our results, with a 75% patency rate
for the vasectomy with conventional suture and an 80% patency rate for
the vasectomy with fibrin glue, agree with the literature that reports
a 80 to 90% patency rate (5-8,10-12,26).
The results from different techniques are
equivalent in terms of patency and pregnancy rates, but it is expected
a reduction in the operative time. There is no clear definition in literature
about surgery duration. Some authors do not mention how time was measured
in methods (12,24,32), others start time measurement at the incision (16,20)
,and there are those who measure time from the first vasovasostomy stitch
(13). In spite of these differences, average operative time for a conventional
suture vasovasostomy ranges from 24 to 96 minutes, while in the fibrin
glue or laser technique it is reduced to 10 to 35 minutes (12,13). The
results of this experiment are 24.6 ± 1.8 minutes for the conventional
anastomosis and 9.35 ± 0.78 minutes for the one with fibrin glue,
and they are comparable to literature. The experiments of longer duration
refer to the total duration of surgery. In this case, our time measurements
of 41.7 ± 2.49 minutes for conventional suture and 28.55 ±
1.14 minutes for the fibrin glue technique still agree with literature.
The fibrin glue technique is simpler, requires
less microsurgery experience and may transform the vasovasostomy a procedure
within reach of a general urologist (12), since it shows results similar
to the conventional microsuture technique, but with reduction of operative
time. It is expected that the reduction in surgery time validates this
technique, but caution needs be taken when the surgeon encounters no fluid
and needs to perform vasoepididymostomy.
ACKNOWLEDGEMENT
Catarinense Institute of Urology provided financial support.
CONFLICT OF INTEREST
None declared.
REFERENCES
- Potts JM, Pasqualotto FF, Nelson D, Thomas AJ Jr, Agarwal A: Patient characteristics associated with vasectomy reversal. J Urol. 1999; 161: 1835-9.
- Engelmann UH, Schramek P, Tomamichel G, Deindl F, Senge T: Vasectomy reversal in central Europe: results of questionnaire of urologists in Austria, Germany and Switzerland. J Urol. 1990; 143: 64-7.
- Roberts HJ: “Is Vasectomy Safe?. West Palm Beach. Florida. Sunshine Academic Press. 1979.
- Heidenreich A, Altmann P, Engelmann UH: Microsurgical vasovasostomy versus microsurgical epididymal sperm aspiration/testicular extraction of sperm combined with intracytoplasmic sperm injection. A cost-benefit analysis. Eur Urol. 2000; 37: 609-14.
- Fenster H, McLoughlin MG: Vasovasostomy-microscopy versus macroscopic techniques. Arch Androl. 1981; 7: 201-4.
- Owen E, Kapila H: Vasectomy reversal. Review of 475 microsurgical vasovasostomies. Med J Aust. 1984; 140: 398-400.
- Silber SJ: Pregnancy after vasovasostomy for vasectomy reversal: a study of factors affecting long-term return of fertility in 282 patients followed for 10 years. Hum Reprod. 1989; 4: 318-22.
- Fox M: Vasectomy reversal—microsurgery for best results. Br J Urol. 1994; 73: 449-53.
- Claro JA, Kesselring G, Ferreira U: Reversão microscópica da vasectomia. Rev Bras Ginecol Obst. 1996; 18: 485-7.
- Goldstein M, Li PS, Matthews GJ: Microsurgical vasovasostomy: the microdot technique of precision suture placement. J Urol. 1998; 159: 188-90.
- Schroeder-Printzen I, Diemer T, Weidner W: Vasovasostomy. Urol Int. 2003; 70: 101-7.
- Silverstein JI, Mellinger BC: Fibrin glue vasal anastomosis compared to conventional sutured vasovasostomy in the rat. J Urol. 1991; 145: 1288-91.
- Seaman EK, Kim ED, Kirsch AJ, Pan YC, Lewitton S, Lipshultz LI: Results of laser tissue soldering in vasovasostomy and epididymovasostomy: experience in the rat animal model. J Urol. 1997; 158: 642-5.
- Shanberg A, Tansey L, Baghdassarian R, Sawyer D, Lynn C: Laser-assisted vasectomy reversal: experience in 32 patients. J Urol. 1990; 143: 528-9.
- Anidjar M, Desgrandchamps F, Martin L, Cochand-Priollet B, Cussenot O, Teillac P, et al.: Laparoscopic fibrin glue ureteral anastomosis: experimental study in the porcine model. J Endourol. 1996; 10: 51-6.
- Ball RA, Steinberg J, Wilson LA, Loughlin KR: Comparison of vasovasostomy techniques in rats utilizing conventional microsurgical suture, carbon dioxide laser, and fibrin tissue adhesives. Urology. 1993; 41: 479-83.
- Leonard SA, Thomas R: New technique for microscopic vasovasostomy. Urology. 1987; 23: 46-7.
- Carey PO, Howards SS, Flickinger CJ, Herr JC, Gallien TN, Caloras D, et al.: Effects of granuloma formation at site of vasovasostomy. J Urol. 1988; 139: 853-6.
- Flam TA, Roth RA, Silverman ML, Gagne RG: Experimental study of hollow, absorbable polyglycolic acid tube as stent for vasovasostomy. Urology. 1989; 33: 490-4.
- Borau Duran A, Ponce de Leon Roca J: Comparison of microsurgical vasovasostomy techniques in the rat. Eur Urol. 1990; 17: 241-2.
- Sharlip ID: Vasovasostomy: comparison of two microsurgical techniques. Urology. 1981; 17: 347-52.
- Silber SJ: Microsurgery for vasectomy reversal and vasoepididymostomy. Urology. 1984; 23: 505-24.
- Vankemmel O, Rigot JM, Burnouf T, Mazeman E: Delayed vasovasostomy: experimental study using fibrin glue. Eur Urol. 1997; 31: 182-6.
- Kucukaydin M, Okur H, Kontas O, Patiroglu TE: Fibrin glue and conventional sutured vasal anastomosis in the rat. J Surg Res. 1995; 59: 601-5.
- Chatterjee S, Rahman MM, Laloraya M, Kumar GP: Sperm disposal system in spermatic granuloma: a link with superoxide radicals. Int J Androl. 2001; 24: 278-83.
- Silber SJ: Microscopic vasectomy reversal. Fertil Steril. 1977; 28: 1191-202.
- Flickinger CJ, Howards SS, Herr JC, Carey PO, Yarbro ES, Sisak JR: Factors that influence fertility after vasovasostomy in rats. Fertil Steril. 1991; 56: 555-62.
- Andonian S, Hermo L: Cell- and region-specific localization of lysosomal and secretory proteins and endocytic receptors in epithelial cells of the cauda epididymidis and vas deferens of the adult rat. J Androl. 1999; 20: 415-29.
- Shekarriz B, Stoller ML: The use of fibrin sealant in urology. J Urol. 2002; 167: 1218-25.
- Naughton CK, Myles J, Thomas AJ Jr: The use of URYX for reversible vasectomy in a rabbit model. J Androl. 2004; 25: 545-53.
- Cavallaro G, Cavallaro E: Vasectomy reversal and spermatic granuloma: experimental investigation. Microsurgery. 2003; 23: 437-9.
- McCallum S, Li PS, Sheynkin Y, Su LM, Chan P, Goldstein M: Comparison of intussusception pull-through end-to-side and conventional end-to-side microsurgical vasoepididymostomy: prospective randomized controlled study in male wistar rats. J Urol. 2002; 167: 2284-8.
____________________
Accepted after revision:
November 30, 2006
________________________
Correspondence address:
Dr. Wilson F S Busato Junior
Instituto Catarinense de Urologia
Marcos Konder, 1120
Itajai, SC, 88301-303, Brazil
E-mail: wbusato@melim.com.br