UROVYSIONTM
TESTING CAN LEAD TO EARLY IDENTIFICATION OF INTRAVESICAL THERAPY FAILURE
IN PATIENTS WITH HIGH RISK NON-MUSCLE INVASIVE BLADDER CANCER
(
Download pdf )
doi: 10.1590/S1677-55382009000600005
JARED M. WHITSON, ANNA B. BERRY, PETER R. CARROLL, BADRINATH R. KONETY
Departments of Urology (JMW, PRC, BPK) and Pathology (ABB), University of California San Francisco, San Francisco, California, USA
ABSTRACT
Purpose:
In this study, we investigated the ability of UroVysionTM to assess response
to intravesical therapy in patients with high risk superficial bladder
tumors.
Materials and Methods: We performed a retrospective
review of patients undergoing intravesical therapy for high risk superficial
bladder tumors. Urine specimens were collected for UroVysionTM analysis
before and immediately after a course of intravesical therapy. Cytology
and cystoscopy were performed six weeks after treatment, using either
a positive cytology or visible abnormality on cystoscopy as a prompt for
biopsy. The operating characteristics of the UroVysionTM test were then
determined.
Results: 41 patients were identified in
whom 47 cycles of induction and 41 cycles of maintenance intravesical
therapy were given during the study period. This yielded a total of 88
treatment and evaluation cycles. Median follow-up was 9 months per induction
(range 1-21 months) and 13 months per patient (range 1-25 months). A total
of 133 urine samples were collected for UroVysionTM of which 40 were positive.
Based upon standard clinical evaluation, 41 biopsies were performed which
detected 20 recurrences. UroVysionTM testing performed immediately upon
completion of therapy for the 41 patients undergoing biopsy yielded a
sensitivity, specificity, and accuracy of 85%, 61%, and 71%.
Conclusions: The use of UroVysionTM following
intravesical therapy for high-risk superficial bladder tumors helps to
identify patients at high risk of refractory or recurrent disease who
should undergo immediate biopsy under anesthesia.
Key
words: bladder neoplasms; superficial; BCG; interferons; chemotherapy;
follow-up
Int Braz J Urol. 2009; 35: 664-72
INTRODUCTION
In
the United States, there are approximately 68,000 new cases of urothelial
carcinoma of the bladder (UC) diagnosed each year, resulting in 14,000
deaths annually (1). A recent population based study found that 32-47%
of all bladder cancer deaths may be preventable, and that preventable
deaths are more common in patients who initially present with non-muscle
invasive disease (2). Currently, the standard of care for high risk superficial
bladder tumors (HRSBT) is transurethral resection of bladder tumor (TURBT)
and intravesical therapy (IVT). Following a 6 week course of IVT, patients
usually undergo a 6 week waiting period prior to cytology and cystoscopy
to allow any abnormalities caused by IVT to normalize. However, for patients
in whom IVT fails, this is merely a period during which lymph node metastasis
could occur or the bladder disease can progress. Therefore, a test which
is able to accurately predict which patients have responded favorably
to IVT within a week of completion of IVT could lead to both earlier initiation
of second line therapy and potentially significant improvements in survival.
Fluorescence in situ hybridization (FISH)
is a technique that uses fluorescently labeled DNA probes to assess cells
for chromosomal alterations. UroVysionTM (Vysis, Downers Grove, IL., USA)
is a Food and Drug Administration approved FISH probe set which detects
gain in copy number of chromosomes 3, 7, and 17 and homozygous deletion
of 9p21. Multiple studies have shown a significantly higher sensitivity
than cytology for detecting UC, including even in high-grade cancers,
while it maintains the high specificity of cytology (3). Additionally,
UroVysionTM is a useful test in cells with atypical or suspicious cytology,
as is often observed during IVT, because it relies on DNA alterations
rather than morphologic changes (4).
In this study, we assessed the ability of
UroVysionTM FISH performed before and at the completion of an induction
cycle of IVT to predict the results of biopsy prompted by standard clinical
evaluation - cytology and cystoscopy - performed 6 weeks after the last
intravesical dose.
MATERIALS AND METHODS
The
University of California San Francisco (UCSF) Urologic Oncology Database
has Institution Review Board approval to collect clinical, pathologic,
and follow-up data on consenting patients who have been seen and treated
for genitourinary cancer at UCSF. The database was queried for patients
who were treated with IVT for HRSBT between 2006 and 2008, a time which
corresponded to the implementation of routine UroVysionTM testing in these
patients. This procedure identified a total of 41 patients who comprised
the study cohort.
Patients were followed-up according to institutional
standard of care. In general, this included voided cytology and UroVysionTM
prior to initiation of IVT. Following the induction course of 6 weekly
doses, a repeat voided urine specimen was collected for UroVysionTM at
or within one week of completion of IVT. Voided/barbotage cytology and
cystoscopy were performed 3 months after initiation of IVT. A positive
cytology or visible abnormality on cystoscopy was a prompt for biopsy.
Documented superficial recurrence on biopsy was an indication for re-induction
or radical cystectomy. Maintenance IVT was administered in 3 weekly doses
6 weeks after completion of the induction course and repeated in 3 weekly
doses at 6 monthly intervals for the following 18-24 months. Routine surveillance
cystoscopy and cytology were performed at 3 monthly intervals for the
first 2 years and every 6 months thereafter up to 5 years.
UroVysionTM Analysis
The UroVysionTM test consists of commercially available DNA probes in the pericentromeric regions of chromosomes 3, 7, and 17 as well as to the 9p21 locus. Slides were interpreted by the same molecular cytopathologist (A.B.). They were diagnosed as positive based on = 4 cells showing polysomy of chromosome 3, 7 or 17, or = 12 cells demonstrating hypodiploid 9p21 content. A minimum of 25 cells were considered as a sufficient sample for the test.
Statistical Analysis
The primary objective was to calculate the sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy of an UroVysionTM test performed immediately after 6 weeks of intravesical therapy. Biopsy results were considered as the reference standard. The secondary objective was to determine whether a change in UroVysionTM over the course of therapy had greater predictive capabilities.
RESULTS
Demographic and clinical data of the 41 patients in this study are shown in Table-1. A total of 47 cycles of induction IVT and 41 cycles of maintenance IVT were given during the study period. Six patients underwent re-induction for biopsy proven superficial recurrence. This yielded a total of 88 treatment and evaluation cycles. In total, the patients were a relatively high risk group for IVT with 56% already having failed at least one course of prior IVT, and 67% of the patients with a high grade T1 disease and/or carcinoma in situ (CIS). Of the patients who began induction with HG T1 disease, 11 out of 19 had repeat TURBT prior to receiving intravesical therapy. Of the patients who did not undergo repeat TURBT, 4 had focal lamina propria invasion alone, 2 were nonagenarians with multiple medical problems, and 2 were referred after induction had already begun.
Median follow-up was 9 months per induction
cycle (range 1-21 months) and 13 months per patient (range 1-25 months).
Forty-one biopsies were performed which detected 20 recurrences. Five
patients underwent radical cystectomy for disease refractory to multiple
courses of intravesical therapy (n = 4), or inability to tolerate induction
intravesical therapy (n = 1). One patient had progressed to muscle invasive
disease. In addition, 2 patients developed upper tract recurrences with
1 who underwent a nephroureterectomy and 1 who was awaiting surgery.
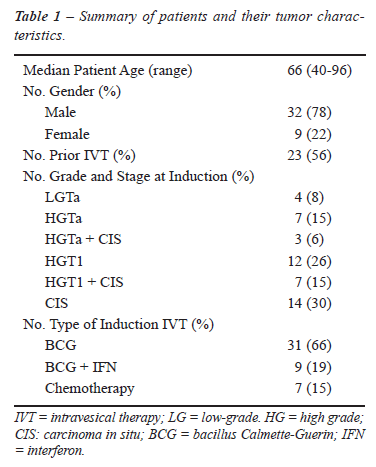
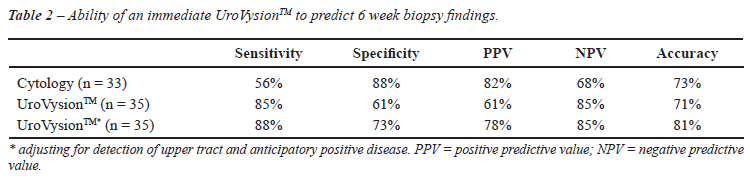
A total of 133 voided urine samples were collected for UroVysionTM. Fifty-two tests were performed prior to IVT (29 before induction, and 23 before maintenance), of which 13 were positive. Eighty-one were performed after IVT (36 after induction and 45 after maintenance), and 27 were positive. The results of testing for the patients who underwent biopsy are illustrated in Figure-1. A total of 34% of patients after IVT had an equivocal cytology. The characteristics of immediate UroVysionTM performed 6 weeks after the last intravesical dose appear in Table-2. Correlation testing before IVT revealed that there was no correlation between cytology and UroVysionTM results (r = 0.15 p = 0.57). Correlation testing after IVT showed a weak correlation between cytology and UroVysionTM results (r = 0.27 p = 0.06). We also evaluated the characteristics of the UroVysionTM test with anticipatory positive and upper tract recurrences included as “true positives”. These data are shown in Table-2. We defined anticipatory positive as those patients with a positive UroVysionTM-negative biopsy at 3 months who later had a positive UroVysionTM-positive biopsy within 6 months.
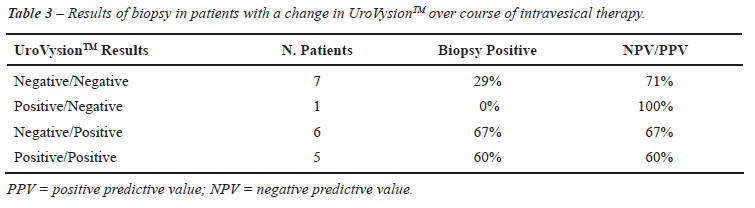
During 44 cycles, the patients had an UroVysionTM
test performed both before and after therapy. Of these patients, 19 had
a biopsy performed based upon abnormal standard clinical evaluation. The
results are shown in Table-3. The NPV of a change in UroVysionTM from
positive to negative compared with a post-IVT negative value alone increased
from 85% to 100%. The PPV of a change in UroVysionTM from negative to
positive compared with a post-IVT positive value alone increased from
59% to 67%.
In 2 patients, UroVysionTM testing did not
predict disease recurrence, which was detected by biopsy (true false negative).
In one patient disease recurrence was detected by cystoscopy, while in
1 patient this was missed by clinical tests as well. This patient was
1 of 2 patients included in the study who had a biopsy performed because
they had been receiving a novel chemotherapy regimen following immunotherapy
failure. In a total of 7 patients, UroVysionTM testing predicted disease
recurrence even though the biopsy was negative. Only 3 of these patients
had no evidence of recurrence in follow-up (true false positive). Three
patients had disease recurrence in the bladder at 3 or 6 month surveillance
(anticipatory positive FISH). One patient had an upper tract recurrence
3 months after the positive FISH and negative biopsy. One patient is still
awaiting a 3 month evaluation. In a total of 4 patients, a positive UroVysion
test was the only predictor of recurrent disease (2 in the bladder and
2 in the upper tract), as both cytology and cystoscopy were negative in
these patients.
COMMENTS
Despite
improvements in surgical technique, refinements in adjuvant intravesical
therapy, and small, but real increased disease specific survival with
chemotherapy, there has been no real change in the age-adjusted total
mortality rate in urothelial carcinoma in the past 20 years (5). Patients
with low grade stage Ta disease have as little as a 5% chance of progression
and even smaller risk of bladder cancer specific mortality (6). Patients
who present with high grade stage = T2 disease may have a somewhat fixed
70% 5 year disease specific survival. In contrast, patients who present
with HRSBT have a lethal disease in a potentially curable form.
There are only a few series of patients
with HRSBT that have undergone cystectomy at diagnosis; therefore, the
83% 10 year disease specific survival rate reported in one early cystectomy
study could have been underestimated (7). In comparison, the 10 year disease
specific survival rate of patients with HRSBT in a randomized clinical
trial of Bacillus Calmette-Guerin therapy was only 70% (8). A further
study has shown that as use of IVT for HRSBT increased, survival of patients
who eventually undergo radical cystectomy has dramatically decreased (9).
In fact, patients with HRSBT who progress to muscle invasive disease while
undergoing IVT have a 10 year disease specific survival of only 27% (7).
Reasons for such dramatic differences in survival are multifactorial;
however, there is evidence to support the concept that the risk of death
from under or untreated high risk disease increases with time.
Three prior studies have evaluated the use
of FISH in monitoring response to IVT in patients with HRSBT (10-12).
The conclusion of these studies was that a positive post-IVT FISH is useful
in predicting eventual relapse, with one study also showing a higher chance
of progression. While these studies showed the important prognostic efficacy
of FISH, no studies have suggested the use of FISH in order to prompt
a change in management. Given the unsatisfactory high mortality in patients
who progress during IVT, we believe that changes in the management of
this group of UC patients are needed. With the goal of decreasing the
time required to detect refractory or recurrent disease, we conducted
this study to evaluate the usefulness of UroVysion FISH in patients undergoing
IVT.
We found that a voided UroVysionTM performed
immediately after completion of an IVT cycle had an accuracy of 71% in
predicting findings on biopsy 6 weeks later. This is in agreement with
the combined accuracy of cytology and cystoscopy in other reported studies
(65-84%), (13-15) but can be achieved without any waiting period. Importantly,
cytology was equivocal in one out of every three patients after IVT, potentially
limiting its usefulness in this setting. The accuracy of UroVysionTM was
lowered mainly by a PPV of 61%. However, in this group of patients with
high risk disease, the ideal test may be one with a high NPV. In this
study, the NPV of UroVysionTM testing was 85%. Thus, under these conditions,
a negative UroVysionTM could be useful to select a group of patients who
could be monitored for 6 more weeks prior to standard evaluation with
cytology and cystoscopy. In addition, the accuracy of UroVysionTM in this
study was significantly affected by both anticipatory positive FISH tests
and by upper tract recurrence. Including these as “true positive”
results this would increase the PPV to 78% and the overall accuracy of
UroVysion to 81%. Patients who change from positive to negative or negative
to positive have even higher NPV and PPV, respectively, although these
data are based on a small sample size.
Although there has been a tendency to move
away from post-IVT protocol biopsies based on published reports, (15)
many randomized trials continue to employ protocol biopsy as the standard
method of evaluation rather than cystoscopy alone (16). In our series,
biopsy of all patients with a positive UroVysionTM test alone (negative
cytology and cystoscopy) would have resulted in 6 extra biopsies. However,
biopsy (or upper tract investigation) of all patients with only a positive
UroVysionTM would have detected disease in 3 patients missed by routine
cystoscopy and cytology. In addition, our data suggest that patients with
a negative UroVysionTM can likely be safely monitored until the standard
3 month time point, as the 2 “false negative” cases showed
“dysplasia” on biopsy which were treated clinically as a recurrence.
Therefore, there were no failures of the UroVysion test to detect biopsy
proven recurrence.
Potentially more importantly, we believe
there is value in determining which patients need further evaluation with
biopsy earlier than the standard schedule. A total of 70% of our patients
with a positive UroVysionTM had recurrent HGTa/T1 disease. While CIS has
been observed in late responders in 11% of patients, (17) it is unlikely
that this would be the case for patients with a frank tumor. Thus, waiting
6 more weeks for these patients would only have led to potential disease
progression. Therefore, the positive UroVysionTM would lead to earlier
TURBT and potentially earlier re-induction, or possibly earlier cystectomy.
It is important to note that the most recent reported guidelines for the
management of non-muscle invasive bladder cancer states that cystectomy
should be considered for initial therapy of select patients, (18) let
alone patients who have likely already failed one induction course of
IVT. According to the European Organization of Research and Treatment
of Cancer risk tables, this might include (in this setting of already
high risk patients) in particular those patients with multifocal disease,
large tumors, or HGT1 with CIS (19).
Limitations of our study include its retrospective
nature, lack of availability of UroVysion test in all patients, particularly
before initial diagnostic TURBT, and that all patients did not undergo
a biopsy. We now have a protocol that includes routine pre-TURBT UroVysion
testing, followed by tests prior to and after induction IVT as well as
before and after maintenance therapy. Another critique might our hipothesis
that earlier detection and treatment of refractory disease improves outcome.
Given the added cost of UroVysionTM, an important next step will be to
perform a prospective study to determine whether or not improvement in
disease specific and overall survival is observed.
CONCLUSIONS
In patients with HRSBT undergoing IVT, UroVysion testing performed immediately upon completion of therapy can predict 6 weeks post IVT biopsy result with a sensitivity, specificity, and accuracy of 85%, 61%, and 71% respectively.
CONFLICT OF INTEREST
None declared.
REFERENCES
- Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, et al.: Cancer statistics, 2008. CA Cancer J Clin. 2008; 58: 71-96.
- Morris DS, Weizer AZ, Ye Z, Dunn RL, Montie JE, Hollenbeck BK: Understanding bladder cancer death: tumor biology versus physician practice. Cancer. 2009; 115: 1011-20.
- Konety BR: Molecular markers in bladder cancer: a critical appraisal. Urol Oncol. 2006; 24: 326-37.
- Pycha A, Mian C, Hofbauer J, Haitel A, Wiener H, Marberger M: Does topical instillation therapy influence chromosomal aberrations in superficial bladder cancer? J Urol. 1998; 159: 265-9.
- Surveillance, Epidemiology and End Results (SEER) Program (www.seer.cancer.gov) SEER *Stat Database: Mortality - All COD, Public-Use With State, Total U.S. (1969-2004), National Cancer Institute, DCCPS, Surveillance Research Program, Cancer Statistics Branch, released April 2007. Underlying mortality data provided by NCHS (www.cdc.gov/nchs).
- Holmäng S, Hedelin H, Anderström C, Holmberg E, Busch C, Johansson SL: Recurrence and progression in low grade papillary urothelial tumors. J Urol. 1999; 162: 702-7.
- Herr HW, Sogani PC: Does early cystectomy improve the survival of patients with high risk superficial bladder tumors? J Urol. 2001; 166: 1296-9.
- Cookson MS, Herr HW, Zhang ZF, Soloway S, Sogani PC, Fair WR: The treated natural history of high risk superficial bladder cancer: 15-year outcome. J Urol. 1997; 158: 62-7.
- Lambert EH, Pierorazio PM, Olsson CA, Benson MC, McKiernan JM, Poon S: The increasing use of intravesical therapies for stage T1 bladder cancer coincides with decreasing survival after cystectomy. BJU Int. 2007; 100: 33-6.
- Mengual L, Marín-Aguilera M, Ribal MJ, Burset M, Villavicencio H, Oliver A, et al.: Clinical utility of fluorescent in situ hybridization for the surveillance of bladder cancer patients treated with bacillus Calmette-Guérin therapy. Eur Urol. 2007; 52: 752-9.
- Kipp BR, Karnes RJ, Brankley SM, Harwood AR, Pankratz VS, Sebo TJ, et al.: Monitoring intravesical therapy for superficial bladder cancer using fluorescence in situ hybridization. J Urol. 2005; 173: 401-4.
- Savic S, Zlobec I, Thalmann GN, Engeler D, Schmauss M, Lehmann K, et al.: The prognostic value of cytology and fluorescence in situ hybridization in the follow-up of nonmuscle-invasive bladder cancer after intravesical Bacillus Calmette-Guérin therapy. Int J Cancer. 2009; 124: 2899-904.
- Guy L, Savareux L, Molinié V, Botto H, Boiteux JP, Lebret T: Should bladder biopsies be performed routinely after bacillus Calmette-Guérin treatment for high-risk superficial transitional cell cancer of the bladder? Eur Urol. 2006; 50: 516-20; discussion 520.
- Skemp NM, Fernandes ET: Routine bladder biopsy after bacille Calmette-Guérin treatment: is it necessary? Urology. 2002; 59: 224-6.
- Dalbagni G, Rechtschaffen T, Herr HW: Is transurethral biopsy of the bladder necessary after 3 months to evaluate response to bacillus Calmette-Guerin therapy? J Urol. 1999; 162: 708-9.
- O’Donnell MA, Lilli K, Leopold C; National Bacillus Calmette-Guerin/Interferon Phase 2 Investigator Group: Interim results from a national multicenter phase II trial of combination bacillus Calmette-Guerin plus interferon alfa-2b for superficial bladder cancer. J Urol. 2004; 172: 888-93. Erratum in: J Urol. 2004; 172: 2485.
- Lamm DL, Blumenstein BA, Crissman JD, Montie JE, Gottesman JE, Lowe BA, et al.: Maintenance bacillus Calmette-Guerin immunotherapy for recurrent TA, T1 and carcinoma in situ transitional cell carcinoma of the bladder: a randomized Southwest Oncology Group Study. J Urol. 2000; 163: 1124-9.
- Hall MC, Chang SS, Dalbagni G, Pruthi RS, Seigne JD, Skinner EC, et al.: Guideline for the management of nonmuscle invasive bladder cancer (stages Ta, T1, and Tis): 2007 update. J Urol. 2007; 178: 2314-30.
- Sylvester RJ, van der Meijden AP, Oosterlinck W, Witjes JA, Bouffioux C, Denis L, et al.: Predicting recurrence and progression in individual patients with stage Ta T1 bladder cancer using EORTC risk tables: a combined analysis of 2596 patients from seven EORTC trials. Eur Urol. 2006; 49: 466-5; discussion 475-7.
____________________
Accepted after revision:
July 19, 2009
_______________________
Correspondence address:
Dr. Jared M Whitson
Box 0738
400 Parnassus Ave, A-631
San Francisco, CA, 94123, USA
Fax: + 1 415 885-8849
E-mail: jwhitson@urology.ucsf.edu
EDITORIAL COMMENT
This article
retrospectively analyses the ability of UroVysionTM FISH test performed
immediately after last intravesical therapy instillation to predict outcome
of follow-up evaluation after 6 weeks. Aim is to reduce standard 6 weeks
waiting while disease may progress.
Unique characteristic of UroVysionTM FISH test lies in its ability to
actually demonstrate / identify genetically abnormal - pathological cells.
Unfortunately, mere presence of genetically abnormal cells is not synonymous
with tumor presence, therefore, test performance, also according to the
present study results, is not ideal, but still promising and in agreement
with other reports (1).
One should avoid the temptation to act solely on results of this test
as in a rather bizarre case I recently observed. Nephroureterectomy was
performed solely based on positive Urovysion FISH result from upper urinary
tract and only dysplasia, no malignancy was found on the pathology sample.
Approach authors preliminary evaluated - using UroVysion FISH for early
identification of non-responders to intravesical therapy, who are at high
risk for disease progression and therefore dismal prognosis - is promising,
but at the moment far from proven or tested. There are more questions
open than answered (for example basic science reasoning, why testing after
6th, not 5th instillation, what is dynamics of treatment response, etc.).
However, in the future, role of UroVysion FISH test, sometimes disputed
(2), may become established just in such scenarios, which deserve further
studies.
REFERENCES
- Hajdinjak T: UroVysion FISH test for detecting urothelial cancers: meta-analysis of diagnostic accuracy and comparison with urinary cytology testing. Urol Oncol. 2008; 26: 646-51.
- Nieder AM, Soloway MS, Herr HW: Should we abandon the FISH test? Eur Urol. 2007; 51: 1469-71.
Dr.
T. Hajdinjak
Department of Urology
Center UROL
Maribor, Slovenia
E-mail: tine.hajdinjak@gmail.com
EDITORIAL COMMENT
This study
assesses the use of the UroVysionTM FISH test in order to predict whether
intravesical Bacillus Calmette-Guerin (BCG) treatment was effective shortly
after completion of the therapy course. This allowed identification of
patients that are refractory to BCG treatment who should be considered
for cystectomy with a sensitivity of 88% and a negative predictive value
of 85%. The advantage of this evaluation by FISH is that earlier assessment
of refractory disease, i.e. directly after completion of a series of intravesical
therapy, may prompt earlier cystectomies. This may then theoretically
shorten the time during which metastatic disease may develop and lives
may be saved. Although the patient group in this study is small, these
results are promising and warrant further extended and prospective studies.
Such a study might also include a FISH test on the primary tumor in order
to be able to select patients whose tumors do show chromosomal abnormalities
in the FISH test.
Another advantage is that FISH and other urine tests can be “anticipatory
positive” that is they can detect tumors that were not seen by cystoscopy.
This phenomenon can be explained by the fact that cystoscopy is not 100%
sensitive (sensitivity estimates range from 63-85%) (1,2) or that some
tumors are yet too small to be seen. Hence, a urine test may identify
more patients that have to be followed more stringently. In addition,
urine tests are able to detect upper tract recurrences that cannot be
seen by cystoscopy as was also the case in this study (3).
REFERENCES
- Schmidbauer J, Witjes F, Schmeller N, Donat R, Susani M, Marberger M, et al.: Improved detection of urothelial carcinoma in situ with hexaminolevulinate fluorescence cystoscopy. J Urol. 2004; 171: 135-8.
- Jocham D, Witjes F, Wagner S, Zeylemaker B, van Moorselaar J, Grimm MO, et al.: Improved detection and treatment of bladder cancer using hexaminolevulinate imaging: a prospective, phase III multicenter study. J Urol. 2005; 174: 862-6; discussion 866.
- Van Tilborg AA, Bangma CH, Zwarthoff EC: Bladder cancer biomarkers and their role in surveillance and screening. Int J Urol. 2009; 16: 23-30.
Dr.
Ellen C. Zwarthoff
Department of Pathology
Josephine Nefkens Institute
Erasmus MC
Rotterdam, The Netherlands
E-mail: e.zwarthoff@erasmusmc.nl