PELVIC
ACTINOMYCOSIS. UROLOGIC PERSPECTIVE
(
Download pdf )
VENKATA K. MARELLA, OMID HAKIMIAN, GILBERT J. WISE, DAVID A. SILVER
Section of Urology, Maimonides Medical Center, Brooklyn, New York, USA
ABSTRACT
Purpose:
Actinomycosis is a chronic granulomatous infection caused by the gram-positive
anaerobic bacteria, Actinomyces israelli. This paper reviews the etiology
and clinical presentation associated with Actinomycosis that often presents
as a pelvic mass that mimics a pelvic malignancy.
Materials and Methods: A combination of
patients treated by the authors in the recent past and a literature review
of patients with pelvic Actinomycosis were assessed for demographic, clinical
and predisposing co-factors. An analysis is made of age distribution,
gender, diagnostic methods and treatment concepts.
Results: Thirty-three patients were included
in the study that included 2 current patients and 31 obtained from literature
review. There were 27 females (age range 16 - 69 years, mean 38 years)
and 6 males (16 - 55 years, mean 36 years). Presenting signs and symptoms
were lower abdominal mass in 28 (85%); lower abdominal pain in 21 (63%);
vaginal discharge or hematuria in 7 (22%). Two patients developed fistulae
(entero-vesico 1; vesico-cutaneous 1). Nineteen (70%) of the 27 female
patients had intra-uterine contraceptive devices (IUD). Four patients
(12.5%) (3 males and 1 female) had urachus or urachal remnants. Cystoscopy
in 12 patients noted an extrinsic mass effect, bullous edema and in one
patient “vegetative proliferation” proven to be a chronic
inflammatory change. Exploratory laparotomy was performed in 32 of the
33 patients who had excision of mass and involved organs. Diagnosis was
established by histologic examination of removed tissue. Penicillin (6
weeks) therapy was utilized to control infections.
Conclusion: Pelvic actinomycosis mimics
pelvic malignancy and may be associated with the long-term use of intra-uterine
contraceptive devices, and persistent urachal remnants. Removal of infected
mass and antibiotic therapy will eradicate the inflammatory process.
Key
words: pelvis; Actinomycosis; infection; tumor
Int Braz J Urol. 2004; 30: 367-76
INTRODUCTION
Actinomycosis
is a chronic granulomatous infection caused by the gram-positive anaerobic
bacteria, Actinomyces israelli, which often occur in clusters of tangled
filaments called sulfur granules. Actinomyces is a natural inhabitant
of the gastrointestinal tract. Clinical actinomycosis includes cervicofacial
(60%), thoracic (15%), abdominal/pelvic (25%) (1).
Actinomycosis is a chronic infection characterized
by the presence of dense fibrous tissue and pus. This infection does not
invade intact mucous membranes, and commonly requires tissue trauma or
the presence of foreign body. Abdominal surgery, ruptured viscus, tubo-ovarian
abscess and intra-uterine contraceptive devices (IUD) are recognized risk
factors for abdominal/pelvic Actinomycosis (2). In females, actinomyces
are thought to be induced by oro-genital contact (3).
The Actinomycetes have been considered as
“transitional forms between bacteria and fungi” however, their
metabolic and morphologic characteristics classify them as higher forms
of monera (bacteria). Actinomycosis is a chronic suppurative infection,
which rarely involves the genitourinary system. Only 2 cases of abdominal
Actinomycosis were encountered in 5 major hospitals with 640,000 admissions
during 1955 to 1964 (4).
Recent presentations of 2 patients with
genito-urinary Actinomycosis and a review of literature indicate that
pelvic infection may be more prevalent. This paper presents an assessment
of patient with pelvic Actinomycosis, and the associated risk factors.
Diagnosis is essential to differentiate
the inflammatory mass or pseudotumor from malignant process.
MATERIALS AND METHODS
A literature review of 31 patients and 2 recent patients were evaluated to determine demographics, etiologic and clinical patterns of patients with pelvic infection caused by Actinomycosis.
RESULTS
A
total of 33 patients with pelvic Actinomycosis are cited (Table-1). Twenty-seven
patients (81%) were female with age range of 16 - 69 years (mean 38 years)
and 6 male (18%) with age range of 16 - 55 (men 36 years). Four patients
(3 male and 1 female) had urachus or urachal remnants. Nineteen (70%)
of the 27 females had intra-uterine contraception devices.
Cystoscopy was performed in 12 patients.
Findings included extrinsic mass effect, bullous edema and in one patient
“vegetative proliferation” that revealed chronic inflammatory
change. Exploratory laparotomy was performed in 32 of the 33 patients
with excision of mass and involved organs (salpinx - 6; hysterectomy -
2; cystectomy or partial cystectomy - 10) Two patients required urinary
diversion and 1 undiversion following determination that the mass was
inflammatory.
The authors recently treated 2 patients
with pelvic Actinomycosis. A 43-year old female presented with 2 months
history of right lower quadrant abdominal pain associated with weight
loss. Past history included 2 caesarian-sections and intrauterine device
for 9 years, which was removed 1 month prior to admission.
Physical examination revealed a firm, tender
right lower quadrant abdominal mass. No cervical motion tenderness was
appreciated. Patient’s laboratory evaluation revealed no leukocytosis,
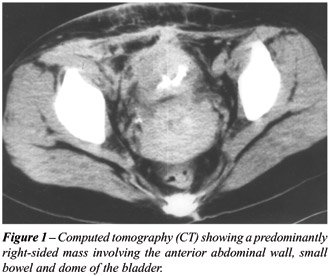
and normal urinalysis. Computed tomography (CT) showed a predominantly
right sided mass involving the anterior abdominal wall, small bowel and
dome of the bladder (Figure-1).
Laparotomy confirmed a firm mass, extending
from anterior abdominal wall to apex of bladder, adherent to right ovary,
and ileum. En-bloc resection of bladder dome, right ovary, terminal ileum,
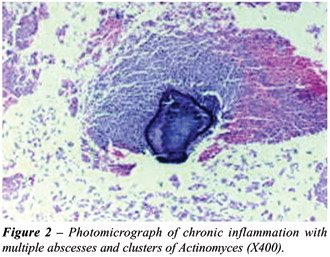
appendix and cecum was performed. Histology revealed chronic inflammation
with multiple abscesses and clusters of Actinomyces (Figure-2). This patient
was treated with intravenous penicillin derivative. There was no recurrence.
Another patient a 39-year old woman had
a 2 years history of lower abdominal pain associated with severe urinary
frequency. The patient reported a 7-year use of IUD and history of penicillin
allergy. Physical examination was unremarkable. Laboratory evaluation
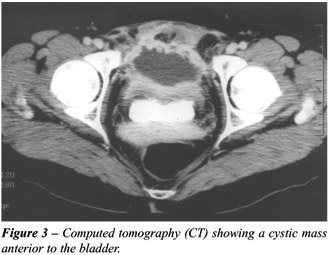
revealed no leukocytosis, pyuria, with sterile urine culture. CT showed
a cystic mass anterior to bladder (Figure-3). Cystoscopy revealed intact
bladder mucosa with edema and impression of extrinsic mass anterior to
bladder. Transurethral biopsy was consistent with inflammatory process.
Elective CT guided drainage of cystic mass disclosed purulent contents.
Although drainage culture was sterile, cytology revealed inflammatory
cells with aggregates of Actinomyces. Patient was treated with prolonged
drainage of abscess cavity and 6 weeks of oral ciprofloxacin. Two months
after removal of the percutaneous drain, patient had recurrence of the
same previous symptoms, and the persistent cystic mass was seen on CT
imaging. Patient underwent laparotomy, and enbloc excision of cystic mass
including the bladder dome. The patient recovered completely after surgery.
A review of 33 patients indicated that the
diagnosis was established in 31 (94%) following surgical exploration and
removal of tissue mass and affected organs that included portions of bladder,
female reproductive organs uterus, salpinx and ovary. Three patients required
urinary, bowel reconstruction or fecal diversions.
DISCUSSION
An
actinomycotic lesion may present as hard mass with induration that extends
to the adjacent organs, resulting in difficult differentiation from malignancy.
Genitourinary symptoms are usually non-specific, and may include abdominal
pain, urinary frequency, and repetitive cystitis.
There is usually a long interval between
the onset of symptoms and establishment of diagnosis. The diagnosis is
primarily histological. Cultures are often unsuccessful in determining
causative factors.
During the past 3 decades, the medical literature
has cited a number of case reports of Actinomycosis of the genito-urinary
system. Actinomycosis has caused primary infections of the kidney (5-7),
including a horseshoe kidney (8), renal duodenal fistula (9), and reno-colic
fistula (10).
Abdominal-pelvic forms of Actinomycosis
have mimicked carcinomatosis with presentations of large pelvic mass and
multiple peritoneal nodules (11). Other manifestations of Actinomycosis
have included retroperitoneal and para-spinal masses (12,13).
Actinomycosis has been known to cause scrotal
masses (14,15), infections of the prostate (16) and penile pilonidal sinus
(17).
Bladder and pelvic infections have been
well documented in the literature. Most patients were women who presented
with lower abdominal mass and suprapubic pain (18-20). Pelvic mass was
the presentation in most of female patients with actinomycosis, with the
history of IUD use. (21-23).
Ureteric obstruction related to pelvic infection
has been reported (24). Brown et al. reported a 34-year old female, with
ureteric obstruction secondary to IUD-associated actinomycosis, which
responded well to tetracycline therapy alone (25). Another 45-year old
female, with IUD use for more than 13 years, presented with bilateral
ureteric obstruction secondary to a large pelvic mass involving the uterus,
tubes, ovaries, and rectum (26). Fulton et al. reported 2 cases with ureteric
obstruction, secondary to IUD-associated Pelvic actinomycosis (27). Tubo-ovarian
abscess was reported by Actinomyces bovis, was reported in a 38-year old
female with IUD use (28).
One patient’s (20) cystoscopy demonstrated
edematous mucosal however this patient presented with a supra-pubic mass
that was similar in presentation as the 2 case reports. King et al. reported
a bladder mass limited to serosa, associated with IUD use for more than
13 years (29). Other presentations of pelvic actinomycosis with bladder
involvement are vesico-cutaneous fistula (30), anterior pelvic masses
involving bladder dome (31), and lateral pelvic masses causing ureteral
compression (32). Actinomycosis involving the urachus or urachal remnant
has been reported. One patient was a 68 year-old woman while the other
cases involved men aged 16, 22 and 55 years (18,33-36).
A 16 year-old male developed an urachal
Actinomycosis that penetrated into the ileum (37). Ureteric obstruction
secondary to retroperitoneal and pelvic Actinomycosis was reported (38,39).
Ileo-vesical fistula was reported in a 69 year-old woman due to Actinomycosis
(40). All patients presented with suprapubic masses thought to be either
an urachal abscess of malignancy. Diagnosis was established following
histologic examination of the resected tissue. All patients responded
to antibiotic therapy. In most cases of female infection the presence
of a long standing (> 6 years) intra-uterine contraceptive device was
incriminated as the putative cause of pelvic infection. A number of cases
have been related to the use of intra-uterine devices.
In only one patient was the diagnosis of
supra vesical Actinomycosis established prior to surgical exploration.
This was achieved by CT guided aspiration of supra-vesical abscess. Although
the patient had 6 weeks of ciprofloxacin therapy, the mass persisted and
the patient subsequently required surgical exploration with excision of
supra-vesical mass and partial cystectomy. The literature cites another
patient in whom diagnosis was established by drainage from a vesico-cutaneous
fistula.
These case reports indicate the potential
for Actinomycosis to develop in women with IUD’s. Actinomyces is
usually found in gastrointestinal tract. The access to the genital tract
is likely to be by oral contact or via blood stream. The disease may become
established in the presence of chronic tissue injury from an IUD, and
some copper containing IUD’s may have a differential effect on bacterial
colonization by anaerobic flora. Luff et al. demonstrated Actinomyces
in 40% of hysterectomy specimens removed from IUD wearers for various
reasons (41). Thus, with chronic inflammation and mucosal breakdown associated
with IUDs appears to favor colonization, and subsequent development of
suppuration. Urachus or urachal remnants also appear to be a co-factor.
Intestinal perforation with ileo-cecal involvement is also a potential
risk factor for pelvic actinomycosis. Awareness of these contributing
factors may interdict the need for aggressive surgery utilized in the
treatment of malignant processes.
The treatment is a combination of extensive
surgical debridement and/or long-term antibiotic therapy. Penicillin is
the drug of choice. If diagnosed pre-operatively, should start with 10
to 20 million units of intravenous penicillin per day for 4 to 6 weeks.
Then wide excision of the infected tissues
and exposure of any sinus tracts should be performed. After surgery, the
patient should receive 1 to 2 million units of oral penicillin continued
for 12 to 18 months. If the diagnosis is made at the time of surgery,
10 to 20 million units of intravenous penicillin, and then long-term oral
penicillin therapy is needed. Tetracycline, erythromycin, clindamycin
and ciprofloxacin are the alternative antibiotics in the management of
patients with penicillin allergy.
CONCLUSIONS
The
presentation of 2 recent case reports and review of 31 cases cited in
the literature reiterate the importance of this diagnostic entity when
the urologist encounters a patient with an unusual pelvic mass. The presence
of long -standing contraceptive intra-uterine device is the most significant
contributing factor in the female patient. An urachus or urachal remnant
is also an associated risk factor.
Surgical resection combined with penicillin
therapy was proven to be successful. In most patients, the diagnosis was
established after exploratory surgery following extensive resection of
infected tissue. Awareness of this diagnostic entity may enable the urologist
to implement antibiotic therapy and minimize the need for major organ
resection.
REFERENCES
- Cope, Z.: Actinomycosis. London, Oxford University Press, 1938.
- Schiffer MA, Elguezabal A, Sultana M, Allen AC: Actinomycosis infections associated with intrauterine contraceptive devices. Obstet Gynecol. 1975; 45: 67-72.
- McCormick JF, Scorgie RD: Unilateral tubo-ovarin actinomycosis in the presence of an intrauterine device. Am J Clin Pathol. 1977; 68: 622-6.
- Weese, W.C., Smith, IM: A study of 57 cases of Actinomycosis over 36 year period. A diagnostic “failure” with good prognosis and treatment. Arch Int Med. 1975; 135: 1562-8.
- Patel BJ, Moskowitz H, Hashmat A: Unilateral renal actinomycosis. Urology. 1983; 21: 172-4.
- Crosse JE, Soderdahl DW, Schamber DT: Renal actinomycosis. Urology. 1976; 7: 309-11.
- Khalaff H, Srigley JR, Klotz LH: Recognition of renal actinomycosis: nephrectomy can be avoided. Report of a case. Can J Surg. 1995; 38: 77-9.
- McGibney D, Clarke PB: Primary renal actinomycosis in the presence of horseshoe kidney. Br J Urol. 1986; 58: 566.
- Cvetkov MC, Elenkov C, Georgiev M, Topov U, Stefanova G: Renal actinomycosis complicated by renoduodenal fistula and diabetes mellitus. Br J Urol. 1995; 75: 104-5.
- Yu HH, Yim CM, Leong CH: Primary actinomycosis of kidney presenting with reno-colic fistula. Br J Urol. 1978; 50: 140.
- Cameron AE, Menon GG, Wyatt AP: Abdominal actinomycosis mimicking carcinomatosis. J R Soc Med. 1988; 81: 231-2.
- Schlech WF 3rd, Gelfand M, Alper B, Kaiser AB: Medical management of visceral actinomycosis. South Med J. 1983; 76: 921-2.
- Levine LA, Doyle CJ: Retroperitoneal actinomycosis: a case report and review of the literature. J Urol. 1988; 140: 367-9.
- Sarosdy MF, Brock WA, Parsons CL: Scrotal actinomycosis. J Urol. 1979; 121: 256-7.
- Jani AN, Casibang V, Mufarrij WA: Disseminated actinomycosis presenting as a testicular mass: a case report. J Urol. 1990; 143: 1012-4.
- de Souza E, Katz DA, Dworzack DL, Longo G: Actinomycosis of the prostate. J Urol. 1985; 133: 290-1.
- Rashid AM, Williams RM, Parry D, Malone PR: Actinomycosis associated with pilonidal sinus of the penis. J Urol. 1992; 148 (2 Pt 1): 405-6.
- Ellis LR, Kenny GM, Nellans RE: Urogenital aspects of actinomycosis. J Urol. 1979; 122: 132-3.
- Makar AP, Michielsen JP, Boeckx GJ, Van Marck EA: Primary actinomycosis of the urinary bladder. Br J Urol. 1992; 70: 205-6.
- Ozyurt C, Yurtseven O, Kocak I, Kandiloglu G, Elmas N: Actinomycosis simulating bladder tumour. Br J Urol. 1995; 76: 263-4.
- Beedham T, Ellice R, Smith H, Usherwood MM: Female genital actinomycosis. Eur J Obstet Gynecol Reprod Biol. 1979; 9: 341-5.
- Spagnuolo PJ, Fransioli M: Intrauterine device-associated actinomycosis simulating pelvic malignancy. Am J Gastroenterol. 1981; 75: 144-7.
- Lomax CW, Harbert GM Jr, Thornton WN Jr: Actinomycosis of the female genital tract. Obstet Gynecol. 1976; 48: 341-6.
- Maeda H, Shichiri Y, Kinoshita H, Okubo K, Okada T, Aoki Y, et al.: Urinary undiversion for pelvic actinomycosis: a long-term follow up. Int J Urol. 1999; 6: 111-3.
- Brown R, Bancewicz J: Ureteric obstruction due to pelvic actinomycosis. Br J Surg. 1982; 69: 156.
- Jackson AE, Parry JR, Shah PJ: Ureteric obstruction secondary to pelvic actinomycosis. Br J Urol. 1988; 62: 85-7.
- Fulton IC, Paterson WG, Crucioli V: Pelvic actinomycosis causing ureteric obstruction. Case reports. Br J Obstet Gynaecol. 1981; 88: 1044-50.
- Henderson SR: Pelvic actinomycosis associated with an intrauterine device. Obstet Gynecol. 1973; 41: 726-32.
- King DT, Lam M: Actinomycosis of the urinary bladder. Association with an intrauterine contraceptive device. JAMA. 1978; 240: 1512-3.
- Deshmukh AS, Kropp KA: Spontaneous vesicocutaneous fistula caused by actinomycosis: case report. J Urol. 1974; 112: 192-4.
- Perez Garcia MD, Rodriguez Alonso A, Nunez Lopez A, Ojea Calvo A, Alonso Rodrigo A, Rodriguez Iglesias B, et al: Abdominal-pelvic actinomycosis with urinary tract involvement, secondary to gynecologic infection caused by intrauterine device. Actas Urol Esp. 2000; 24: 197-201.
- Marco Perez LM, Lopez Costea MA, Trilla Herrera E, Condom Mundo E, Franco Miranda E, Serrallach Mila N: Primary bladder actinomycosis. Actas Urol Esp. 2000; 24: 573-5.
- Grierson JM, Zelas P: Actinomycosis involving the urachal remnant. Med. J. Aust. 1977; 1: 849-52.
- Micheli E, Hurle R, Losa A, Chinaglia D, Ranieri A, Lembo A: Primary actinomycosis of the urachus. BJU Int. 1999; 83: 144-5.
- Gotoh S, Kura N, Nagahama K, Higashi Y, Fukui I, Takagi K: Actinomycosis of urachal remnants. J Urol. 1988; 140: 1534-5.
- Asano S, Fujii Y, Inokuchi S: Actinomycosis of the bladder wall with transitional cell carcinoma of the bladder. Nippon Hinyokika Gakkai Zasshi. 1977; 68: 983-6.
- Nagy V, Sokol L, Baca M, Valansky L, Zelenak P, Bodnar J: Actinomycosis of the urachus persistens penetrating into the ileum. Int Urol Nephrol. 1997; 29: 627-31.
- Isaacson, P, Jennings, M., Bilateral ureteric obstruction in a patient with ileocecal Crohn’s disease complicated by actinomycosis. Br J Urol. 1977; 49: 410.
- Willscher MK, Mozden PJ, Olsson CA: Retroperitoneal fibrosis with ureteral obstruction secondary to Actinomyces israeli. Urology. 1978; 12: 569-71.
- Piper JV, Stoner BA, Mitra SK, Talerman A: Ileo-vesical fistula associated with pelvic actinomycosis. Br J Clin Pract. 1969; 23: 341-3.
- Luff RD, Gupta PK, Spence MR, Frost JK: Pelvic actinomycosis and the intrauterine contraceptive device. A cyto-histomorphologic study. Am J Clin Pathol. 1978; 69: 581-6.
_____________________
Received: June 28, 2004
Accepted: August 20, 2004
_______________________
Correspondence address:
Dr. Venkata K. Marella
Section of Urology
Maimonides Medical Center
953, 49 st, # 301
Brooklyn, New York, 11219, USA
Fax: + 1 718 635-7093
E-mail: vkmarella@hotmail.com