SUBMUCOSAL BLADDER NECK INJECTIONS FOR MANAGEMENT OF STRESS URINARY INCONTINENCE
ARNULF STENZL, HANNES STRASSER
Department of Urology, University of Innsbruck Medical School, Innsbruck, Austria
ABSTRACT
Submucosal deposition of bulking agents
is increasingly used for the treatment of intrinsic stress urinary incontinence
both in male and female patients. Bulking agents should ideally be non-immunogenic,
biocompatible, non-migratory, injectable, cause a minimal inflammatory
reaction, retain the submucosal bulging effect, and reasonably cheap.
None of the agents available to date fully meets these criteria. The rationale
and clinical results of artificial, heterologous and autologous agents
currently used for treatment are analyzed and the technique of transurethral
submucosal injection applied by the authors is described. Perioperative
imaging using transrectal or transvaginal ultrasonography is important
for the exact placement of the injected material, and is one of the key
factors for a satisfactory result.
Depending on length of follow-up, the route
of application (transurethral, antegrade transvesical, periurethral),
the etiology of the patients, the amount of injected material, the number
of procedures and the bulking agent used continence and improvement rates
vary from 21-83% and 40-100%, respectively.
A future outlook describes new agents and
techniques that may be promising for broad clinical use.
Key words:
incontinence, bulking agents, urethra, injectables, submucosal injection,
ultrasonography.
Braz J Urol, 26: 199-207, 2000
INTRODUCTION
The concept of using injectables to increase urethral resistance and thus treat urinary incontinence was already known more than sixty years ago (1). The first agents, such as paraffin or Dondren, actually worked as sclerosing medium causing a difficult to control urethral stenosis. In the early seventies the first bulking agent in the actual sense – polytetrafluoroethylene (Teflonâ) – was introduced as a possible means for treating incontinence (2). Several encouraging reports (3,4) were soon followed by reports about disappointing long-term results (5) and complications, especially migration of substance particles into the lungs (6,7) and brain (8) causing at times severe morbidity. More recently developed alloplastic injectables for the urethra which have a bulging effect onto the urethral mucosa are collagen, suspended silicone particles and newer potential substances like polyvinyl alcohol foam (9), dextranomers in hyaluronan (10), and bioglass (11). Potential long-term immunogenic side effects (12) and possible migration of these substances have led to the developments of techniques whereby autologous materials such as the patients’ own fat (13), collagen (14), smooth muscle cells, or chondrocytes (15) are used for suburethral injection. The problem with non-vascularized free tissue transplants is a significant loss of implant volume over time. This may be overcome by the use of precursor cells such as stem cells, myoblasts (16) or chondrocytes (17), which according to recent preliminary results induce new tissue formation at the injection site, rendering uniform and stable implants after several months.
RELEVANT ANATOMY
Female
The goal of injectables is to restore the
relatively thick, highly vascular submucosa, which contributes coaptation
and mucosal seal to the continence mechanism. In female patients coaptation
and mucosal seal is most effective in the proximal urethra where it is
supported by urethral smooth muscle tone and dense fascial structures
supporting the bladder neck and adjacent urethra (18). Smooth musculature
extends along the entire length of the urethra, whereas the striated fibers
of the rhabdosphincter are located only in the mid and distal portion
(19,20). These are the sites where the greatest increase in intraurethral
pressure occurs. The rhabdosphincter together with the surrounding extrinsic
striated muscles contribute to the resting tone but also to the pressure
rise that precedes coughing and Valsalve maneuvers and which is exerted
when inhibiting micturition. These zones of increased intraluminal pressures
therefore less depend on the sealing effect of a thick submucosa.
Male
A lack of watertightness in male patients
in the majority of cases is a consequence of either lower urinary tract
surgery or trauma. During transurethral and radical prostatectomy almost
all of the relevant smooth muscle sphincter is removed or destroyed and
continence depends on the omega-shaped layer of the rhabdosphincter (21).
Inadvertent cutting, deep bites during placement of the anastomotic sutures,
hematoma, or extensive inflammation may cause scars and functional impairment
of the rhabdosphincter. In addition some of the various amounts of fibers
that extend ventrally to the base of the bladder will also be eradicated
during open surgery. Anatomical studies have shown that the striated sphincter
is not a ring-like structure but a complex interdigitating system intimately
connected to the prostatic smooth musculature.
INDICATIONS
It is evident that bulking agents will restore neither a weakened pelvic floor nor a damaged sphincter. Its presumed mode of action is the restoration of the submucosal cushion in the urethra, which has been lost either due to age, previous surgery or chronic disease. In female patients therapy with injectables is therefore best indicated in pure intrinsic sphincter deficiency, but may be combined with reconstructive pelvic floor surgery as an adjuvant (22). In male patients a contractile external sphincter damaged by previous surgery or trauma is the most rewarding indication. Endoscopically visible indentations may in these cases delineate the site of preferred injection.
MECHANISM OF ACTION
The
first injected substances used led to a sclerosis with destruction of
the urethral musculature and non-compliance of the urethral wall instead
of a submucosal bulge. It was only more recently that inert substances
applied into the submucosa resulted in its actual bulging at and above
the level of the maximum sphincter pressure.
Polytetrafluoroethylene (TeflonÒ)
paste is usually encapsulated but not penetrated by vascular and fibrous
tissue creating at times sequester in the urethral wall (23,24). Commercially
available collagen preparations consist of sterile, nonpyrogenic bovine
dermal collagen cross-linked with glutaraldehyde and dispersed in phosphate-buffered
physiologic saline (25). Glutaraldehyde cross-linked (GAX) collagen starts
to degrade within 12 weeks. At the same time neovascularization and deposition
of fibroblasts and autologous collagen takes place inside the implant
(26). A similar albeit more rapid process is assumed after injection of
autologous fat. Experimental studies have shown, however, that in the
long term up to 90% of the fat implant is lost (27).
Silicone preparations consist of biphasic
co-polymers of fully polymerized and vulcanized particles of polydimethylsiloxane
compounds suspended in a hydrogel carrier (28). The size of the particles
ranges from 100 to 600mm (average 150mm), which makes particle migration
practically impossible. After absorption of the hydrogel carrier within
the first few days the innumerous irregularly shaped silicone particles
are solidly encapsulated by collagenous tissue thus creating the therapeutic
mucous bulge.
TECHNIQUE OF TRANSURETHRAL INJECTION
In
order to achieve our current success rate we developed a standardized
transurethral technique using three-dimensional ultrasound (29).
Implantations were performed using three
dimensional, transrectal ultrasound with a 7.5 MHz Voluson 3D multiplanar
endorectal transducer (30) before, during and after the procedure. Intraoperative
transrectal ultrasound will show the position of the needle and subsequently
the position of the injected material. This may also be useful for any
additional procedures to show which deposits remained inside and which
ones may have been lost.
The bulking agent initially was a collagen
preparation and consists now of solid textured silicon particles suspended
in a carrier gel (31).
Technique
in Male Patients
In patients with stress urinary incontinence
after radical prostatectomy the external sphincter zone needs to be outlined.
This can be done by scoping the patient under local anesthesia during
which the patient is asked to contract the sphincter. In addition transrectal
ultrasound may delineate the morphology of the rhabdosphincter and sometimes
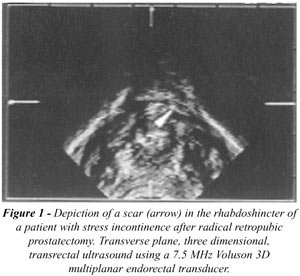
sonographically scars can be depicted (Figure-1). The needle is then inserted
in the bladder, drawn back into the proximal urethra, turned appropriately
and injected at an angle of at least 45°.
A regular rigid cystoscope with an Albarran
deflector, a 0° to 30° lens and a 5F working channel are used.
First the defect is visualized endoscopically and its relationship to
the external sphincter zone is clarified. The endoscope is advanced into
the bladder, the bladder is filled, and an 18g needle is inserted through
the working channel of the scope until it can be seen through the lens
thereby avoiding any lacerations of urethral or vesical mucosa. After
turning the needle it is brought to the location of the desired injection,
angled at least 45° from the urethral axis, and inserted into the
submucosa for approximately 1 cm or any visible marking of the needle,
e.g., beginning of a white coating. If transrectal sonography is used
during the procedure the insertion of the needle and its exact position
can be followed on the ultrasound screen.
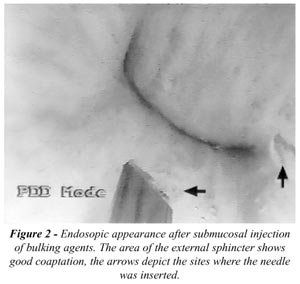
Whenever a defect in the sphincter zone
is apparent, the endoscopically visible defect is undermined and cushioned
with 1 to 2.5 cc of the agent (Figure-2). In addition or in cases where
there is no apparent defect, the material is injected at the 3, 9 and
if possible 6 o’clock position. After each injection the needle should
be left in place for at least one minute to allow the material to settle.
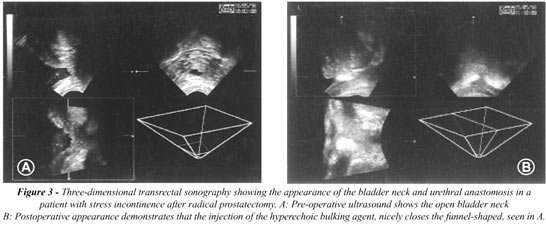
Three-dimensional transrectal sonography
improves the exact transurethral deposition of the implants. After injection
the suspended collagen or polydimethylsiloxane particles present as hyperechogenic
structures which can be depicted in all three dimensions (Figure-3).
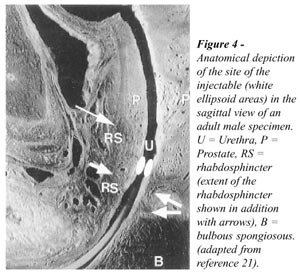
Usually patients where the rhabdosphincter
can be outlined fare better than those where this is sonographically not
possible. The goal of the procedure is to restore the coaptation of the
mucosa from the level of the external sphincter cranially (Figure-4).
After the injections the cystoscope should not be passed beyond the area
of injection nor should any catheter be inserted afterwards. The immediate
success of the injection can be tested with a simple trick. When the sphincteric
zone is sufficiently closed, irrigation fluid of the scope placed in the
urethra will not be able to advance into the bladder but instead will
be seen coming out along side the cystoscope shaft at the tip of the penis.
Technique
in Female Patients
The injection technique in female patients
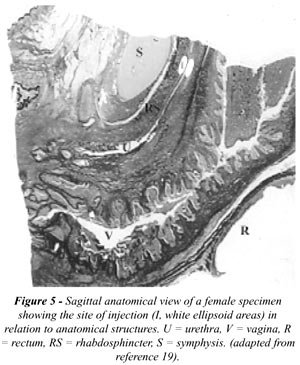
is slightly different because no distinct sphincteric zone can be seen
(Figure-5), and due to the length of the urethra injections in the caudal
part of the urethra are difficult because of insufficient irrigation.
A rigid cystoscope with an Albarran deflector,
a 5F working channel, but a 30° to 70° lens are therefore advisable.
A specially designed short endoscope where the working channel opening
is at the level of the optical lens is currently developed.
The injection procedure is basically the
same with the exception of the location and the number of deposits. Smaller
but more deposits (usually four) are placed at the bladder neck and cranial
third of the urethra at the 3, 6, 9 and 12 o’clock position.
Follow-up
Catheterizations should be avoided in the
early postoperative period. If urinary retention occurs or may be anticipated
at the time of injection, a 10 Fr. suprapubic catheter should be inserted
until the patient resumes spontaneous voiding. Patients should receive
oral antibiotics for 1 week. Mechanic pressure to the perineum such as
hard seat covers, hard stools, etc. should be avoided for 2 weeks, bicycling
should be avoided for 6 weeks. If a second or third injection is necessary,
it should be performed after an interval of at least 3 months to allow
the previous implant to heal in.
Pitfalls
The needle can be inserted in the urethra
too far cranially resulting in a partial or total loss of material into
the bladder lumen. It may not always be visible endoscopically, but could
definitely be seen if ultrasound were used during the procedure. If the
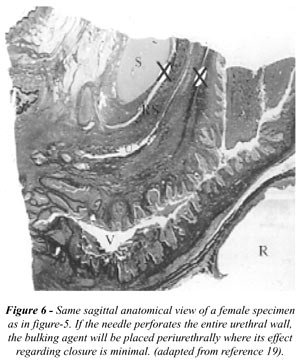
needle perforates the entire urethral wall the bulking agent will be placed
periurethrally where its effect regarding closure is minimal (Figure-6).
It is important to remember to outline sphincteric
defects when injecting bulking agents transurethrally. Injections into
the remaining rhabdosphincter in male patients may even aggravate incontinence
postoperatively.
The insertion of the needle in the urethra
may cause lacerations of the urethral mucosa with subsequent bleeding
and blurred vision.
RESULTS
Many
urologists have abandoned the use of Teflon in recent years due to disappointing
long-term results and severe co-morbidity related to distant migration
end ensuing embolization of the particles (5,7). Collagen has replaced
Teflon over the last 10 years, and has definitely increased the safety
of submucosal urethral injections (25). In a multicenter trial injection
related transient urinary retention in women was observed in 8%, urinary
tract infection in 6%, hematoma formation at the injection site in 2%,
and pain in 1% (25). Hypersensitivity reaction to the collagen material,
however, is possible, and a skin test prior to the injection is mandated
(12).
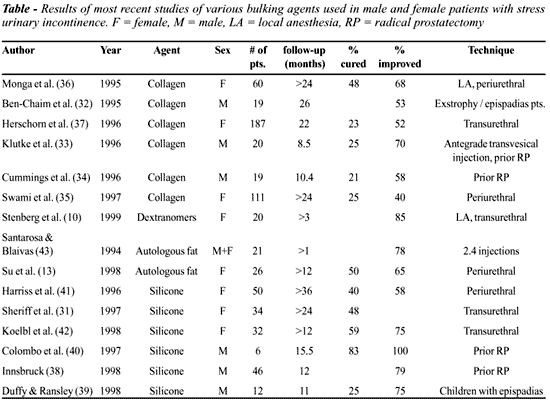
Cure and improvement rates with submucous
collagen in male patients with stress incontinence due to exstrophy/epispadias
complex or prior radical prostatectomy ranges from 21-25% and 53-70%,
respectively (32-34). In female patients 23 – 48% and 40 – 68%
are being reported as cured and improved, respectively (25, 35-37) (Table).
The reported continence rates for male patients
using vulcanized silicone particles ranges from 25 to 83%, and improvement
rates extend from 75 to 100% (38-40). In female patients after a minimum
follow-up of at least 12 months cure rates between 40 and 59% and improvement
rates between 58 and 77% are reported (31,41,42). Only minor complications
comparable to those seen with collagen were seen. To date there have been
no reports about hypersensitivity. Experimental studies demonstrated no
migration of silicon particles larger than 70mm after periurethral injection
(28). Due to an average silicone particle size of 150 mm in the clinically
used preparation neither migration nor any migratory-related adverse effects
have been observed.
Whether dextranomer microspheres suspended
in a sodium hyaluronan solution (10) are going to be superior to any currently
used agent with regard to continence, durability and side effects, needs
to be demonstrated in larger studies.
The most extensively studied autologous
substance to date is fat (13,27,43,44). Its advantages are that autologous
fat is absolutely biocompatible, readily available, and inexpensive. The
drawback is that in the long-term 10-50% of the injected fat will survive,
repeated injections may become necessary, and extensive periurethral scarring
may result.
FUTURE OUTLOOK
In
order to overcome the problem of biocompatibility, migration, and short
and long-term immunogenic reactions, the use of autologous tissues and
cells was the focus of recent experimental studies (15). As outlined above
autologous fat despite promising experimental results did not prove to
be as good clinically.
A better option is apparently chondrocytes
(17), which possess the ability to form viable cartilage. Chondrocytes
are harvested from the patient’s auricular surfaces, submersed in
an alginate polymer delivery vehicle, and injected submucosally into the
urethra, where they are supposed to act as a stable bulking material.
Its clinical applicability is currently assessed in a multicenter clinical
trial in the United States.
Combining the technique of submucosal urethral
injection with tissue engineering principles (45) may be an interesting
variant. A possible advantage might not only be a permanent, perfectly
biocompatible bulking material, but also a periurethral expansion of the
injected cells. A further step could be to make bulking agents functional
by provoking, e.g., nerve ingrowth. To date animal studies using autologous
smooth muscle cells, myoblasts (16), and stem cells have been performed
using various carrier substances. Stability of the implants, and surprising
differentiation into viable muscle cells when using precursor cells, could
be demonstrated. The material in which the cells are suspended prior to
injection has to be carefully selected regarding a possible immunogenic
reaction, maintenance of cell viability, and viscosity necessary for injection
through a reasonably small needle. Whether bulking agents need to be injected
through a needle at all is also a matter of debate. The authors of this
paper currently undertake experiments with new injection modalities.
CONCLUSION
Treatment of intrinsic stress incontinence with endoscopic submucosal injection is an effective method and is the least invasive of all surgical procedures. New developments, especially in combination with tissue engineering, may render it as one of the primary treatment modalities for stress urinary incontinence in the future.
REFERENCES
- Murless B: The injection treatment of stress urinary incontinence. J Obstet Gynaecol Br Emp, 45: 67, 1938.
- Berg S: Polytef augmentation urethroplasty. Correction of surgically incurable urinary incontinence by injection technique. Arch Surg, 107: 379-381, 1973.
- Lopez AE, Padron OF, Patsias G, Politano VA: Transurethral polytetrafluoroethylene injection in female patients with urinary continence. J Urol, 150: 856-858, 1993.
- Politano VA: Transurethral polytef injection for post-prostatectomy urinary incontinence. Br J Urol, 69: 26-28, 1992.
- Beckingham IJ, Wemyss-Holden G, Lawrence WT: Long-term follow-up of women treated with perurethral Teflon injections for stress incontinence. Br J Urol, 69: 580-583, 1992.
- Mittleman RE, Marraccini JV: Pulmonary Teflon granulomas following periurethral Teflon injection for urinary incontinence [letter]. Arch Pathol Lab Med, 107: 611-612, 1983.
- Claes H, Stroobants D, Van Meerbeek J, Verbeken E, Knockaert D, Baert L: Pulmonary migration following periurethral polytetrafluoroethylene injection for urinary incontinence. J Urol, 142: 821-822, 1989.
- Borgatti R, Tettamanti A, Piccinelli P: Brain injury in a healthy child one year after periureteral injection of Teflon. Pediatrics, 98: 290-291, 1996.
- Merguerian PA, McLorie GA, Khoury AE, Thorner P, Churchill BM: Submucosal injection of polyvinyl alcohol foam in rabbit bladder. J Urol, 144: 531-533, 1990.
- Stenberg A, Larsson G, Johnson P, Heimer G, Ulmsten U: DiHA Dextran Copolymer, a new biocompatible material for endoscopic treatment of stress incontinent women. Short term results. Acta Obstet Gynecol Scand, 78: 436-442, 1999.
- Walker RD, Wilson J, Clark AE: Injectable bioglass as a potential substitute for injectable polytetrafluoroethylene. J Urol, 148: 645-647, 1992.
- Stothers L, Goldenberg SL: Delayed hypersensitivity and systemic arthralgia following transurethral collagen injection for stress urinary incontinence. J Urol, 159: 1507-1509, 1998.
- Su TH, Wang KG, Hsu CY, Wei HJ, Yen HJ, Shien FC: Periurethral fat injection in the treatment of recurrent genuine stress incontinence. J Urol, 159: 411-414, 1998.
- Cendron M, DeVore DP, Connolly R, Sant GR, Ucci A, Calahan R, Klauber GT: The biological behavior of autologous collagen injected into the rabbit bladder. J Urol, 154: 808-811, 1995.
- Kershen RT, Atala A: New advances in injectable therapies for the treatment of incontinence and vesicoureteral reflux. Urol Clin North Am, 26: 81-94, 1999.
- Hward J, Yokoyama T, Lavelle J, Teahan S, Watanabe T, Ozawa H, Yoshimura N, de Groat W, Qu Z, Chancellor M : Differentiation of primary myoblast injection into the lower urinary tract; creation of detrusor cellular myoplasty. J Urol, 161:66, 1999.
- Atala A, Cima LG, Kim W, Paige KT, Vacanti JP, Retik AB, Vacanti CA: Injectable alginate seeded with chondrocytes as a potential treatment for vesicoureteral reflux. J Urol, 150:745-747, 1993.
- DeLancey JO: The pathophysiology of stress urinary incontinence in women and its implications for surgical treatment. World J Urol, 15: 268-274, 1997.
- Colleselli K, Stenzl A, Eder R, Strasser H, Poisel S, Bartsch G: The female urethral sphincter: a morphological and topographical study. J Urol, 160: 49-54, 1998.
- Strasser H, Colleselli K, Stenzl A, Frauscher F, Helweg G, Bartsch G: Transurethrale Sonographie: Evaluierung des Rhabdosphinkters der weiblichen Urethra bei Streßinkontinenz. J Urol Urogynäkol, 4: 36, 1997.
- Strasser H, Klima G, Poisel S, Horninger W, Bartsch G: Anatomy and Innervation of the Rhabdosphincter of the male urethra. Prostate, 28: 24-31, 1996.
- McGuire EJ, English SF: Periurethral collagen injection for male and female sphincteric incontinence: indications, techniques, and result. World J Urol, 15: 306-309, 1997.
- Dewan PA, Byard RW: Histological response to injected Polytef and Bioplastique in a rat model. Br J Urol, 73: 370-376, 1994.
- Smith DP, Beegle BE, Noe HN, Wilson EA: Does technique or material used affect bladder tissue reactions when injecting teflon or silicone paste? Urology, 48: 119-123, 1996.
- Winters JC, Appell R: Periurethral injection of collagen in the treatment of intrinsic sphincteric deficiency in the female patient. Urol Clin North Am, 22: 673-678, 1995.
- Leonard MP, Canning DA, Epstein JI, Gearhart JP, Jeffs RD: Local tissue reaction to the subureteral injection of glutaraldehyde cross-linked bovine collagen in humans. J Urol, 143: 1209-1212, 1990.
- Olson ME, Morck DW, Ceri H, Lee CC, Chancellor MB: Evaluation of autologous fat implantation in the rat urinary bladder submucosa. Urology, 52: 915-919, 1998.
- Henly DR, Barrett DM, Weiland TL, O’Connor MK, Malizia AA, Wein AJ: Particulate silicone for use in periurethral injections: local tissue effects and search for migration. J Urol, 153: 2039-2043, 1995.
- Stenzl A, Strasser H, Bartsch G: Ultrasound-guided transurethral injection of implants for treatment of stress incontinence. Eur Urol, 35 (Suppl.2): 181 (V24), 1999.
- Strasser H, Frauscher F, Helweg G, Stenzl A, Neururer R, Bartsch G: Three-dimensional transrectal and transurethral ultrasound of the male rhabdosphincter. Eur Urol, 35 (suppl.2): 94, 1999.
- Sheriff MK, Foley S, McFarlane J, Nauth-Misir R, Shah PJ: Endoscopic correction of intractable stress incontinence with silicone micro-implants. Eur Urol, 32: 284-288, 1997.
- Ben-Chaim J, Jeffs RD, Peppas DS, Gearhart JP: Submucosal bladder neck injections of glutaraldehyde cross-linked bovine collagen for the treatment of urinary incontinence in patients with the exstrophy/epispadias complex. J Urol, 154: 862-864, 1995.
- Klutke CG, Nadler RB, Tiemann D, Andriole GL: Early results with antegrade collagen injection for post-radical prostatectomy stress urinary incontinence. J Urol, 156: 1703-1706, 1996.
- Cummings JM, Boullier JA, Parra RO: Transurethral collagen injections in the therapy of post-radical prostatectomy stress incontinence. J Urol, 155: 1011-1013, 1996.
- Swami S, Batista JE, Abrams P: Collagen for female genuine stress incontinence after a minimum 2-year follow-up. Br J Urol, 80: 757-761, 1997.
- Monga AK, Robinson D, Stanton SL: Periurethral collagen injections for genuine stress incontinence: a 2- year follow-up. Br J Urol, 76: 156-160, 1995.
- Herschorn S, Steele DJ, Radomski SB: Followup of intraurethral collagen for female stress urinary incontinence. J Urol, 156:1305-1309, 1996.
- Strasser H, Stenzl A, Bartsch G : Intraurethral Macroplastique® injections in treatment of urinary stress incontinence. Eur Urol, 35 (Suppl.2):114, 1999.
- Duffy PG, Ransley PG : Endoscopic treatment of urinary incontinence in children with primary epispadias. Br J Urol, 81: 309-311, 1998.
- Colombo T, Augustin H, Breinl E, Schips L, Hubmer G: The use of polydimethylsiloxane in the treatment of incontinence after radical prostatectomy. Br J Urol, 80: 923-926, 1997.
- Harriss DR, Iacovou JW, Lemberger RJ: Peri-urethral silicone microimplants (Macroplastique) for the treatment of genuine stress incontinence. Br J Urol, 78: 722-725, 1996.
- Koelbl H, Saz V, Doerfler D, Haeusler G, Sam C, Hanzal E: Transurethral injection of silicone microimplants for intrinsic urethral sphincter deficiency. Obstet Gynecol, 92: 332-336, 1998.
- Santarosa RP, Blaivas JG: Periurethral injection of autologous fat for the treatment of sphincteric incontinence. J Urol, 151: 607-611, 1994.
- Matthews RD, Christensen JP, Canning DA: Persistence of autologous free fat transplant in bladder submucosa of rats. J Urol, 152: 819-821, 1994.
- Atala A, Freeman MR, Vacanti JP, Shepard J, Retik AB: Implantation in vivo and retrieval of artificial structures consisting of rabbit and human urothelium and human bladder muscle. J Urol, 150: 608-612, 1993.
______________________
Received: August 30, 1999
Accepted: September 5, 1999
_______________________
Correspondence address:
Arnulf Stenzl
Department of Urology, Universität Kliniken
Anichstrasse 35
A-6020, Innsbruck, Austria
Fax: ++ (43) (512) 504-8411
E-mail: arnulf.stenzl@uibk.ac.at