THE
ISOLATED GAMMA PROBE TECHNIQUE FOR SENTINEL NODE PENILE CARCINOMA DETECTION
IS UNRELIABLE
(
Download pdf )
LUCIO F. GONZAGA-SILVA, JOSE M. TAVARES, FERNANDO C. FREITAS, MANOEL E. TOMAS FILHO, VLADMIR P. OLIVEIRA, MARCOS V. LIMA
Department of Surgery, Federal University of Ceara and Section of Urology, Cancer Hospital of Ceara, Fortaleza, Ceara, Brazil
ABSTRACT
Purpose:
Penile carcinoma is a common disease in northeast Brazil. This paper shows
the results of the use of isolated gamma probe and discusses the incidence
of false negative rates.
Materials and Methods: From July 2000 to
September 2003, 27 newly diagnosed penile carcinoma patients (T1, T2,
N0) were included in this prospective study. The isolated gamma probe
technique uses the sodium phytate technetium as a tracer and inguinal
scanning with probe and after identified the lymph node it is removed.
Lymphadenectomies were performed for positive inguinal lymph nodes metastasis.
Results: There were 27 patients (mean age
59.6). Follow up was 37 months. Patients from country were 72% and illiterate
or semi-illiterate were 56.7%. The tumors were mostly located in the glans
(81.4%). They were T1, 52 % and T2, 48 %. 81.4% of the patients underwent
partial penectomy, and 18.6% underwent postectomy and excision with wide
margins. In 48% of the patients, the highest radioactive count rate was
located on the left side, while in 41% was located on the right side.
Only one patient had a positive pathological lymph node metastasis at
the moment of the surgery. Additionally 3 patients became inguinal lymph
node positive at the follow up. This date yielded a sensibility rate of
25% and a false-negative rate of 42.8%.
Conclusion: Isolated gamma probe technique
for sentinel node penile carcinoma has a very low sensibility and a high
false negative rate. Therefore it is highly advisable the addition of
others methods such as lymphoscintigraphy, vital blue, ultrasonography
and so on. The isolated gamma probe technique for sentinel node penile
carcinoma detection is unreliable.
Key
words: penile cancer; lymphatic metastasis; sentinel lymph node
biopsy; gamma probe technique
Int Braz J Urol. 2007; 33: 58-67
INTRODUCTION
Though
rare in developed countries, penile carcinoma is relatively common in
Latin America (1). On the average, 3 new cases have been identified at
our Hospital every month over the last 5 years (2).
The lymphatic system is the primary route
for metastasis. Tumors spread loco-regionally and stepwise (3). Survival
of patients with penile carcinoma clearly depends on the status of the
inguinal lymph node (4). Thus, the overall 5-year survival rate is 73-95%
for patients with negative inguinal lymph nodes and 19-62% for patients
with positive inguinal lymph nodes (5). When associated with pelvic disease,
the 5-year survival rate is less than 10%. If left untreated, patients
with metastasis rarely survive for longer than 2 years (6).
In patients with metastasis confined to
the regional lymph nodes, inguinal lymphadenectomy is very often curative,
with disease-free rates of 30-90% (7). Inguinal lymphadenectomy has been
associated, in some reports, with significant levels of morbidity (30-50%)
and up to 3% of mortality (8,4). Moreover, prophylactic bilateral inguinal
lymph node dissection is considered unnecessary in up to 80% of penile
carcinoma patients with clinically negative regional lymph nodes (9).
Elective bilateral inguinal lymph node dissection
is the most invasive approach and the least invasive is a “wait
and see” policy (10). However, the latter is associated with a poorer
prognosis if tumor-involved lymph nodes become palpable during follow-up
(11,12).
Dynamic sentinel node biopsy provides a
means of assessing lymph node status in the management of penile carcinoma,
through a minimally invasive procedure. It has important diagnostic, prognostic
and therapeutic value at the cost of only minor morbidity and is an attempt
to diminish the risk of complications without jeopardizing the oncological
results (10).
The gamma probe technique has been adopted
at our institution since July 1999. However, it was used alone (without
lymphoscintigraphy and vital dye) on the first 27 patients mainly because
our hospital had not yet been officially authorized to use the gamma camera.
After the use of the equipment had been
authorized (September 2003), the complete approach (gamma probe, lymphoscintigraphy
and vital blue dye) became the routine procedure in the investigation
of penile carcinoma sentinel nodes.
The purpose of this paper was to show the
results of the isolated use of gamma probe technique for sentinel node
investigation in patients with penile carcinoma at our institution, before
the complete technique was introduced in September 2003, and to analyze
its value as a less invasive lymphatic staging method.
MATERIALS AND METHODS
Between
July 2000 and September 2003, 27 patients with newly diagnosed penile
squamous cell carcinoma were prospectively enrolled for sentinel node
detection by gamma probe technique. Informed consent was obtained from
all patients and the protocol was approved by the research ethics committee
at our institution.
The 27 patients enrolled in the study did
not differ significantly from subjects described in similar studies published
in the literature. The mean age was 59.6 years (range 41-80). The Mean
follow-up time was 37 months (range: 24-62). Five patients were lost to
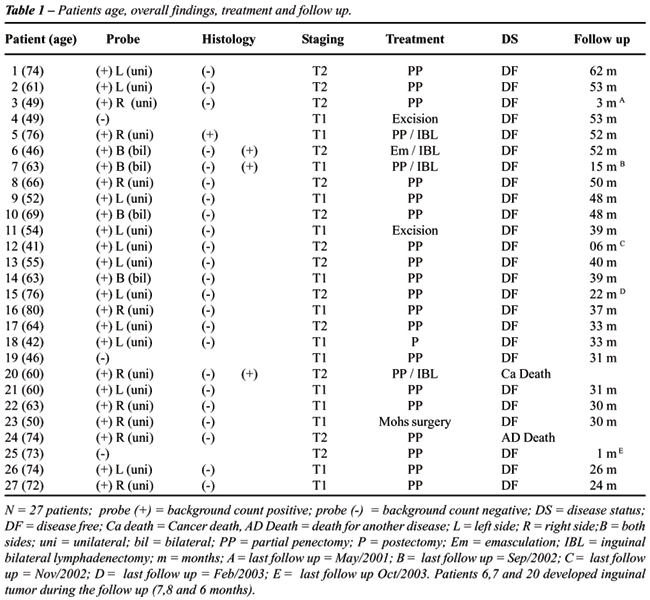
follow up (Table-1).
Patients were eligible when staged T1-T2,
N0, according to the 2002 TNM classification system of the International
Union Against Cancer.
At our hospital, technetium-99-m-labeled
sodium phytate (99mtc) is used in colloidal presentation as a tracer.
A dose of 0.2 mL was injected subdermally at the four cardinal points
around the penile lesion, totaling 0.8 mL. The average radioactive dose
was (59 MBq) (13).
The bilateral inguinal probe scanning was
performed about 30 minutes after the injection. First, background count
is established by measuring radioactivity over a neutral site. Then the
handheld gamma detection probe was used to identify the sentinel node
sites. A sentinel node was defined as a node with radioactivity three
times over the background.
Finally, the focus of activity in the inguinal
area (the so-called hot point) was marked off. A small incision was then
made and the sentinel node was removed. When sentinel lymph nodes are
removed successfully, the radioactivity count rate on the excision site
should not exceed 10% of the highest ex-vivo radioactivity count.
The excised sentinel lymph node was sent
to the pathologist after making sure it was the one (on either side) with
the highest background radioactivity count (corresponding to the highest
quantity of radio colloid).
The sentinel node was bisected, fixed in
formalin, embedded in paraffin and sectioned at 8 levels, on the average
(5-µm sections). Paraffin sections were stained with hematoxylin and eosin
(13).
Inguinal lymphadenectomies were performed
immediately for all inguinal lymph node metastasis proving positive. Negative
cases, (i.e. without lymph nodes metastasis) were followed up with check-ups
every three months for three years. Patients subjected to inguinal lymphadenectomy
were kept under careful observation to identify early or late complications.
Disease-specific survival is defined as
the percentage of people in a study who have survived a particular disease
(in this case, penile cancer) since diagnosis or treatment. Disease-free
survival is the length of time after treatment during which no disease
(in this case nodal or distant recurrence) is found.
RESULTS
Most
of the patients were illiterate or semi-illiterate (56.7%) (From country
72%). The tumors were mostly located in the glans (81.4%) and in the prepuce
(38%). Some tumors affected the glans and prepuce (18.5%).
As to the tumor stage, 51.9% were T1 and
48.1% were T2. With regard to treatment, 81.4% of the patients underwent
to partial penectomy, while the remainder had postectomy and excision
with wide margins (18.6%). In eleven patients (40.7%), the highest radioactive
count was located only on the left side; in nine patients (33.3%), it
was located only on the right. The radioactive count was too low for sentinel
node identification in three patients (11.1%) (Table-1).
No complications related to the isolated
gamma probe technique for sentinel node penile carcinoma detection were
observed.
Only one patient exhibited lymph node metastasis
at the time of the surgery. The patient was given a bilateral inguinal
lymphadenectomy during the same session. The pathological examination
of the specimen revealed that the excised sentinel lymph node was the
only node affected (Table-1).
In addition, three patients who were negative
for pathological lymph node metastasis at the moment of the surgery became
inguinal lymph node-positive in the course of follow up. Thus, the sensitivity
rate was 25% and the false-negative rate of 42.8% (Table-1).
Of the four patients submitted to bilateral
inguinal lymphadenectomy three are disease-free at the time of writing
(September 2005) and one died of cancer.
COMMENTS
Prophylactic
bilateral inguinal lymph node dissection is considered unnecessary in
up to 80% of penile carcinoma patients with clinically negative regional
lymph nodes. The challenger lies in identifying the remaining 20% of occult
metastasis and thereby offers such patients an opportunity for cure (9).
The isolated use of familiar predictive
prognostic factors for the diagnosis of occult lymph node metastasis,
such as depth of invasion, differentiation grade, and vascular invasion,
results in considerable false-positive and false-negative rates (10).
On the other hand, dynamic sentinel node
biopsy has proved very useful in the detection of occult lymph node metastasis.
The procedure is minimally invasive, preventing unnecessary lymph node
dissections, and moreover, improves substantially the staging with a morbidity
rate of only 8% (10,14).
Although dynamic sentinel node biopsy for
penile carcinoma was adopted at our hospital in July 2000, the procedure
was restricted to the intra-operative gamma ray technique. Twenty seven
patients were studied by this method until September 2003 when the complete
technique (including preoperative lymphoscintigraphy, intra-operative
gamma ray detection and a vital blue dye) became available.
Sentinel lymph node biopsy is best performed
by a committed team with experience in surgery, pathology and nuclear
medicine. The nuclear physician is responsible for preparing and measuring
the radioactive material and for controlling the measuring and mapping
equipment. The surgeon is responsible for handling the probe during the
surgery, for the surgical procedures and for the management of the case
during follow-up (15).
The overall findings, treatment and follow-up
of these initial 27 patients (examined with intraoperative gamma probe
only) are illustrated in the Table-1.
Unlike the studies by Horenblas (14), which
excluded patients with T1 tumors in view of the low risk of occult metastasis,
our study was designed so as to include patients with penile carcinoma
staged both T1 and T2 N0, considering the local incidence of late recurrences
in such patients (T1).
The Table-1 shows that in all but three
patients the sentinel nodes were visualized by the intraoperative gamma
probe technique. At this point, no measures were taken for patients with
radioactive count below the background count rate (3 patients). However,
as recommended by Horenblas and coworkers, it is now considered mandatory
to examine such patients for tumor deposits blocking the passage of tracer
(5).
The vast majority of our patients underwent
to partial penectomy, while three patients with very small tumors (4,
11 and 23) were given conservative surgery. Our research team soon is
publishing a description of this approach and its indications.
Only one of 27 T1 and T2 patients had a
positive histology test (5). He was submitted to inguinal bilateral lymphadenectomy
shortly after penectomy, and was alive and disease-free after a follow-up
of 36 months.
The Table-1 shows the number of positive
histology tests observed during the total follow-up period (July 2000
to September 2003). The histological positivity was observed in four patients.
Thus, the sensitivity of the isolated intraoperative gamma probe technique
was 25%.
Inguinal tumor outgrowth after excision
of a classified tumor-negative sentinel node or non-visualization is classified
as false-negative result (10). The Table-1 show that three patients presented
these features i.e. three patients (6,7 and 20) developed inguinal tumor
after a negative sentinel node biopsy.
The false-negative rate is defined as the
number of false-negative results divided by the total of positive results
plus the false-negative results (10). Our three false-negative results
occurred clustered around the beginning of the study and resulted in a
false-negative rate of 42.8%.
A false-negative rate of this order clearly
indicates that the isolated gamma probe technique is not a reliable way
of detecting sentinel nodes in penile carcinoma.
The Netherlands Institute of Cancer (Horenblas
et al.) found an initial false-negative rate of 18% (6 of 34 cases). The
technique revealed metastasis in 28 of 123 patients and was false-negative
in 6 patients (13).
In 2001 important adjustments were made
to the procedure of dynamic sentinel node biopsy in penile carcinoma patients
at the Netherlands Institute of Cancer (Pathological analysis by serial
sectioning and immunohistochemical staining, preoperative ultrasonography
with fine-needle biopsy aspiration cytology and preoperative lymphoscintigraphy
besides exploration of nonvisualized groin) leading to eradication of
false-negative results.
In a study of 70 T2-T3 patients submitted
to dynamic sentinel node biopsy (pre-operative lymphoscintigraphy, intra-operative
gamma probe and vital blue dye), Perdoná and coworkers (2005) found
a false negative rate of 11% and a sensitivity of 90% (16).
At our Hospital we had also made adjustments
to the procedure (as of September 2003), which now includes preoperative
lymphoscintigraphy and intra-operative injection of vital blue dye besides
gamma ray detection. These improvements are expected to reduce false-negative
rates.
Several studies are presently conducted
on a variety of tumors and sites. These studies will no doubt, cautiously
endorse sentinel lymph node biopsy. However, sentinel lymph node biopsy
can be difficult to master. Most surgeons will agree that it takes considerable
experience to correctly identify sentinel lymph nodes; in fact, the Oncology
Group of the American College of Surgeons recommends that physicians perform
at least 30 sentinel lymph node biopsies as part of their training (17).
CONCLUSION
Our findings show that the isolated gamma probe technique for sentinel node penile carcinoma is associated with very low sensitivity (25%) and high false-negative rates (42.8%). The inclusion of other techniques, such as lymphoscintigraphy, vital blue, ultrasonography, is therefore highly advisable.
CONFLICT OF INTEREST
None declared.
REFERENCES
- Donald F: Tumors of the Penis. In: Campbell’s Urology, 8th edition. Philadelphia, WB Saunders. 2002; pp. 2945.
- Gonzaga-Silva LF: Histopathology – Biopsy and Prognostic Factors. In: Practical Guide of Urology, Rio de Janeiro, Segmento. 2003; pp. 561-2. [in Portuguese]
- Tabatabaei S, Harisinghani M, McDougal WS: Regional lymph node staging using lymphotropic nanoparticle enhanced magnetic resonance imaging with ferumoxtran-10 in patients with penile cancer. J Urol. 2005; 174: 923-7; discussion 927.
- Ornellas AA, Seixas AL, Marota A, Wisnescky A, Campos F, de Moraes JR: Surgical treatment of invasive squamous cell carcinoma of the penis: retrospective analysis of 350 cases. J Urol. 1994; 151: 1244-9.
- Srinivas V, Morse MJ, Herr HW, Sogani PC, Whitmore WF Jr: Penile cancer: relation of extent of nodal metastasis to survival. J Urol. 1987; 137: 880-2.
- Ravi R: Correlation between the extent of nodal involvement and survival following groin dissection for carcinoma of the penis. Br J Urol. 1993; 72: 817-9.
- Kossow JH, Hotchkiss RS, Morales PA: Carcinoma of penis treated surgically. Analysis of 100 cases. Urology. 1973; 2: 169-72.
- Akduman B, Fleshner NE, Ehrlich L, Klotz L: Early experience in intermediate-risk penile cancer with sentinel node identification using the gamma probe. Urology. 2001; 58: 65-8.
- Abi-Aad AS, deKernion JB: Controversies in ilioinguinal lymphadenectomy for cancer of the penis. Urol Clin North Am. 1992; 19: 319-24.
- Kroon BK, Horenblas S, Meinhardt W, van der Poel HG, Bex A, van Tinteren H, et al.: Dynamic sentinel node biopsy in penile carcinoma: evaluation of 10 years experience. Eur Urol. 2005; 47: 601-6; discussion 606.
- Theodorescu D, Russo P, Zhang ZF, Morash C, Fair WR: Outcomes of initial surveillance of invasive squamous cell carcinoma of the penis and negative nodes. J Urol. 1996; 155: 1626-31.
- McDougal WS: Carcinoma of the penis: improved survival by early regional lymphadenectomy based on the histological grade and depth of invasion of the primary lesion. J Urol. 1995; 154: 1364-6.
- Kroon BK, Horenblas S, Estourgie SH, Lont AP, Valdes Olmos RA, Nieweg OE: How to avoid false-negative dynamic sentinel node procedures in penile carcinoma. J Urol. 2004; 171: 2191-4.
- Horenblas S, Jansen L, Meinhardt W, Hoefnagel CA, de Jong D, Nieweg OE: Detection of occult metastasis in squamous cell carcinoma of the penis using a dynamic sentinel node procedure. J Urol. 2000; 163: 100-4.
- Lima MV, Tavares JM, Silveira RA, Tomas Filho ME, Silva FA, Silva LF: Inintraoperative use of gamma probe for identification of sentinel node in penile carcinoma. Int Braz J Urol. 2002; 28: 123-9.
- Perdona S, Autorino R, De Sio M, Di Lorenzo G, Gallo L, Damiano R, et al.: Dynamic sentinel node biopsy in clinically node-negative penile cancer versus radical inguinal lymphadenectomy: a comparative study. Urology. 2005; 66: 1282-6.
- Cantin J, Scarth H, Levine M, Hugi M, Steering Committee on Clinical Practice Guidelines for the Care and Treatment of Breast Cancer: Clinical practice guidelines for the care and treatment of breast cancer: 13. Sentinel lymph node biopsy. CMAJ. 2001; 165: 166-73. Erratum in: CMAJ 2001; 165: 744.
____________________
Accepted after revision:
October 25, 2006
_______________________
Correspondence address:
Dr. Lucio Flavio Gonzaga Silva
Rua Dr. José Lino, 141 / 1002, Varjota
Fortaleza, CE, 60165-270, Brazil
E-mail: gonzagalf@secrel.com.br
EDITORIAL COMMENT
In
1994, we started with sentinel node biopsy for penile cancer patients.
While acknowledging the pioneering work of Cabanas we deliberately labeled
our procedure as a dynamic sentinel node procedure to underscore the static
nature of the original description and the dynamic nature of the new one.
The dynamism is readily seen on the lymphoscintigrams, giving an almost
real-time picture of the individual drainage pattern. Too many individual
variations led to false negative findings, explaining the lack of enthusiasm
of the original description of the procedure.
There has been a long controversy between
proponents of an early lymph node dissection and proponents of a wait
and see strategy for clinically node negative patients. An analysis from
our institute showed an increased survival for patients who underwent
an early lymph node dissection based on tumor positive sentinel node finings.
In these series no unnecessary lymph node dissections were done. All patients
harbored pathologically proven clinically occult metastases. Lymph node
dissection was done at the earliest possible moment (1).
The main problem remained to improve the
accuracy of the sentinel node biopsy. Accurate sentinel node biopsies
rely on collaboration of nuclear medicine physicians, surgeons and pathologists.
Moreover, it relies on the use of all possible methods to exclude false
negative findings. In our practice this means preoperative ultrasound
with or without fine needle aspiration biopsy, a preoperative lymphoscintigram,
marking of the sentinel nodes on the skin, discussing the lymphoscintigram
with the nuclear medicine physician, injecting patent blue around the
tumor, using a gamma detector and having a protocol for measurements before
removal and after removal, palpation of the wound after removal and a
strict pathology protocol. With all these refinements, we were able to
bring down the false negative rate of the initial series of 22% to an
acceptable 4.8% (in press European Urology). In our hands sentinel node
biopsy for penis cancer has evolved as an important clinical staging tool
just as reliable as in melanoma and breast cancer patients. One should
realize however, that the procedure was initiated in a specialized cancer
center together with surgical oncologists, who pioneered the procedure
for breast cancer and melanoma.
Another aspect not often cited is the small
size of the Netherlands, making regular outpatient-clinic visits easy,
follow-up very reliable, with a 100% literacy within the population and
the increasing tendency to centralize management of penile cancer patients.
All these aspects have been helpful in developing, analyzing and improving
the dynamic sentinel node biopsy.
How different is the situation in Brazil
and many other countries. The procedure should be viewed against the above-mentioned
elements. A rational choice should be made between the introduction of
a sentinel node biopsy program, with all the logistics around it or a
straightforward lymph node dissection in a patient without any follow
up after initial surgery.
What is clear from this paper is that introducing
only some aspects of the procedure is not benefiting the patient and should
not be done.
REFERENCE
1. Ravi R: Correlation between the extent of nodal involvement and survival following groin dissection for carcinoma of the penis. Br J Urol. 1993; 72: 817-9.
Dr. Simon
Horenblas
Professor of Urologic Oncology
Head, Departmen of Urology
Netherlands Cancer
Inst-Antoni van Leeuwenhoek Hosp
Amsterdam, The Netherlands
E-mail: s.horenblas@nki.nl
EDITORIAL COMMENT
Defining
the presence (or absence) of microscopic metastases within the inguinal
lymph nodes in patients with squamous penile cancer and no clinical lymphadenopathy
remains a challenge. Selective lymphadenectomy based upon the stage, grade
or the presence of vascular invasion within the primary tumor decreases
the incidence of unnecessary lymphadenectomy but is clearly imprecise.
Based upon the evolving experience initially published by colleagues at
the Netherlands Cancer Institute (ref. 10, 13, 14 in the article) Dynamic
Sentinel Lymph Node Biopsy with preoperative lymphoscintigraphy with subsequent
intraoperative detection of emitted gamma radioactivity using a hand held
probe has been shown to be a minimally morbid procedure.
In the current study, the authors studied
a cohort of patients using only intraoperative detection, as preoperative
lymphoscintigraphy was unavailable. Four patients were eventually found
to exhibit lymph node metastasis but only one of four was discovered at
surgery leading to a sensitivity of only 25%. One of the three died due
to cancer, one was lost to follow-up at 15 months, and the other is alive
and presumed cured at 52 months.
Based upon their experience the authors
correctly conclude that gamma detection alone was not sensitive and they
have now moved on to performing both lymphoscintigraphy and intraoperative
mapping.
This study points to some of the difficulties
in establishing a new surgical technique in the setting of a rare disease
with a relatively narrow “window of curability”. First in
their study and those from the Netherland’s Cancer Institute (NCI)
the patients with false negative findings often present later with incurable
disease and die. Second, in the largest series reported the NCI group
(using optimal technique) reported a false negative rate of 18% that is
almost the same as that reported using physical examination. This has
lead to subsequent technical modifications that could improve test performance
but this is not guaranteed. Finally the learning curve is thought to be
about 30 cases to gain proficiency (ref. 17 in the article) and thus is
not practical for most urologists that are not in a referral setting.
Alternatively superficial inguinal lymphadenectomy
detects all the first echelon lymph nodes at risk, is less morbid in contemporary
reports, and in several reports was not associated with false negative
findings (1-3). Thus I believe superficial inguinal lymphadenectomy in
selected high risk patients to be the “standard” and Dynamic
Sentinel Node Biopsy as reported here, by the NCI group, and in our own
experience (ref. 3 below) to be a technique in evolution that is still
experimental and requires further refinement in high volume centers.
REFERENCES
- Bevan-Thomas R, Slaton JW, Pettaway CA: Contemporary morbidity from lymphadenectomy for penile squamous cell carcinoma: The M.D. Anderson Cancer Center experience. J Urol. 2002; 167: 1638-42.
- Colberg JW, Andriole GL, Catalona WJ: Long-term follow-up of men undergoing modified inguinal lymphadenectomy for carcinoma of the penis. Brit J Urol. 1997; 79: 54-57.
- Spiess PE, Bassett R, Izawa J, Kedar D, Busby JE, Wong F, et al.: Utility of pre-operative lymphoscintigraphy and intra-operative sentinel lymph node mapping in penile cancer: a different viewpoint. J Urol. 2006 (Suppl) 175: #600.
Dr. Curtis A. Pettaway
Professor of Urology
The University of Texas
M.D. Anderson Cancer Center
Houston, Texas, USA
E-mail: cpettawa@mdanderson.org
EDITORIAL COMMENT
The
presence of nodal metastases is the single most important factor in penile
cancer. However, the timing of lymphadenectomy in patients presenting
without clinical signs of node disease is still controversial. In the
last years, there has been a growing awareness on the need of adopting
better staging procedures in order to decrease the number of unnecessary
node dissections and to improve detection of occult metastases.
The current European Association of Urology
Guidelines recommend a risk-based approach for lymph node staging, including
a wait and see strategy and surgical groin exploration using a low, intermediate
and high risk stratification (1). In this setting, a new concept for lymph
node staging, the so called dynamic sentinel biopsy (DSNB), has been introduced
(2). Also in our experience, this technique offered similar results to
those of radical lymphadenectomy but significantly lower morbidity (3).
Thus, the present report by Gonzaga-Silva
et al. is interesting as it deals with a timely topic in uro-oncology.
Of course, the major limitation of this prospective single institution
study is the fact that the authors adopt an incomplete technique, as they
clearly admit. This choice is questionable as they offer an already obsolete
procedure to their 27 patients, with an expected limited clinical benefit.
As a matter of fact, they recognize from the beginning that the real DSNB
technique consists of different parts (i.e. preoperative lymphoscintigraphy,
blue dye injection, intraoperative gamma ray detection), each one with
a specific role in the reliability of the entire diagnostic procedure.
They obviously found that the isolated gamma probe use has a very low
sensibility and high false negative rate. Of note is that the authors
also considered T1 tumors in their series, as recently suggested by Leijte
et al. (4), as a risk of metastasis should be considered for these patients.
Finally, we agree with the authors when
they state that DSNB procedure is difficult to master and it should be
performed in centers with a minimum number of cases. We look forward to
have the results from the same group of investigators after the introduction
of the complete DSNB technique in their clinical practice as this should
minimize false negative rates and optimize its diagnostic accuracy. In
order to achieve this goal, a recent report by the group with the most
extensive experience worldwide highlight some modifications to the original
DSNB technique: fine needle aspiration cytology before lymphoscintigraphy;
serial sectioning when analyzing the nodes; intraoperative palpation after
injection of patent blue and surgical exploration in case of non-visualized
groin (4). Further clinical research in this area is needed to better
define the place of the sentinel node biopsy in the management of penile
cancer patients. In this respect a very interesting approach has been
proposed by Tobias-Machado et al, who presented their encouraging results
on 10 patients submitted to video endoscopic inguinal lymphadenectomy
(5).
REFERENCES
- Solsona E, Algaba F, Horenblas S, Pizzocaro G, Windahl T; European Association of Urology: EAU guidelines on penile cancer. Eur Urol. 2004; 46: 1-8.
- Horenblas S, Jansen L, Meinhardt W, Hoefnagel CA, de Jong D, Nieweg OE, et al.: Detection of occult metastasis in squamous cell carcinoma of the penis using a dynamic sentinel node procedure. J Urol. 2000, 163: 100-4.
- Perdona S, Autorino R, De Sio M, Di Lorenzo G, Gallo L, Damiano R, et al.: Dynamic sentinel biopsy in clinically node-negative penile cancer versus radical inguinal lymphadenectomy: a comparative study. Urology. 2005, 66: 1282-6.
- Leijte JAP, Kroon BK, Valdes Olmos RA, Nieweg OE, Horenblas S: Reliability and safety of current dynamic sentinel node biopsy for penile carcinoma. Eur Urol 2007, in press.
- Tobias-Machado M, Tavares A, Ornellas AA, Molina WR Jr, Juliano RV, Wroclawski ER: Video endoscopic inguinal lymphadenectomy: a new minimally invasive procedure for radical management of inguinal nodes in patients with penile squamous cell carcinoma. J Urol. 2007, 177: 953-58.
Dr. Riccardo Autorino
Clinica Urologica, AOU Policlinico
Seconda Università
Napoli, Italy
E-mail: ricautor@tin.it
Dr. Sisto Perdonà
UO Urologia, Istituto Nazionale Tumori
Fondazione G. Pascale
Napoli, Italy