GLEASON
SCORE AS PREDICTOR OF CLINICOPATHOLOGIC FINDINGS AND BIOCHEMICAL (PSA)
PROGRESSION FOLLOWING RADICAL PROSTATECTOMY
(
Download pdf )
MARBELE S. GUIMARAES, MAISA M. QUINTAL, LUCIANA R. MEIRELLES, LUIS A. MAGNA, UBIRAJARA FERREIRA, ATHANASE BILLIS
Departments of Anatomic Pathology (MSG, MMQ, LRM, AB), Medical Genetics and Biostatistics (LAM), and Urology (UF), School of Medicine, State University of Campinas (Unicamp), Campinas, SP, Brazil
ABSTRACT
Objective:
There is evidence showing that Gleason grading of prostatic adenocarcinoma
is one of the most powerful predictors of biological behavior and one
of the most influential factors used to determine treatment for prostate
cancer. The aim of the current study was to compare the Gleason score
for needle biopsy to the Gleason score for the correspondent surgical
specimen, find any possible difference in the biochemical (PSA) progression
following surgery in upgraded cases, correlate Gleason score in the specimens
to several clinicopathologic variables, and compare outcomes between patients
with low-grade vs. high-grade Gleason and Gleason scores 3+4 vs. 4+3.
Materials and Methods: The study population
consisted of 200 consecutive patients submitted to radical prostatectomy.
Biochemical progression was defined as PSA ≥ 0.2 ng/mL. Time to
PSA progression was studied using the Kaplan-Meier product-limit analysis.
Results: In 47.1% of the cases, there was
an exact correlation and 40.6% of cases were underestimated in the biopsies.
Half of the tumors graded Gleason 6 at biopsy were Gleason score 7 at
surgery. These upgraded tumors had outcomes similar to tumors with Gleason
score 7 in both biopsy and surgery. There was a positive correlation of
high-grade Gleason score in the surgical specimens to higher preoperative
PSA, more extensive tumors, positive margins and more advanced pathologic
staging. Tumors with a Gleason score ≥ 7 have lower PSA progression-free
survival vs. Gleason scores < 7. In this series, there was no significant
difference when comparing Gleason scores of 3+4 vs. 4+3.
Conclusions: The findings support the importance
of Gleason grading for nomograms, which are used by clinicians to counsel
individual patients and help them make important decisions regarding their
disease.
Key
words: prostatic neoplasms; prostatectomy; Gleason grading system
Int Braz J Urol. 2008; 34: 23-9
INTRODUCTION
There
is evidence showing that a Gleason grading of prostatic adenocarcinoma
is one of the most powerful predictors of biological behavior and one
of the most influential factors used to determine treatment for prostate
cancer. The Gleason grading system is the most commonly used grading system
for prostate cancer in the United States and is gaining worldwide acceptance.
There are several unique aspects of the Gleason grading system including
the fact that is solely based on the architectural pattern and that cytologic
features are not factored in (1-3).
The aim of the current study report was:
a) to compare the Gleason score on needle biopsy to the Gleason score
in the correspondent surgical specimen; b) to find any possible difference
in the biochemical (PSA) progression following surgery in upgraded cases;
c) to correlate the Gleason score in the surgical specimen with the variables:
preoperative serum PSA, tumor extent, positive surgical margins, and extraprostatic
extension (pT3a and/or pT3b); and, d) to compare biochemical (PSA) progression
following surgery between patients with Gleason low-grade vs. high-grade,
and Gleason score 3+4 vs. 4+3.
MATERIAL AND METHODS
This
retrospective study was based on 200 consecutive patients submitted to
retropubic radical prostatectomy from January 1997 to July 2004 at our
Institution. The Gleason score on all needle biopsies (58.9% sextant biopsies
and 41.1% extended biopsies) was compared to the Gleason score on the
correspondent surgical specimen by one senior pathologist (AB). The mean
and median cores for the extended biopsies were 9.3 and 8.5 respectively.
The Gleason score in the specimen was evaluated to find any association
with the variables: preoperative serum PSA, tumor extent, positive margins,
extraprostatic extension (pT3a and / or pT3b), and biochemical (PSA) progression
following surgery.
The previously fixed surgical specimen was
weighed, measured and the entire surface inked. The bladder neck and apical
margins were amputated. From each cone-shaped amputated margin, 8 fragments
were processed through perpendicular sections relative to the margins.
The rest of the prostate was serially cut in transverse sections at 3
to 5 mm intervals. The prostate slices were subdivided into quadrants
and labeled to allow for reconstruction as whole-mount sections. Blocks
were embedded in paraffin, cut at 6mm, and one section from each block
was stained with hematoxylin and eosin.
Presence of adenocarcinoma was diagnosed
according to the criteria of Mostofi & Price (1). The diagnosis was
based on invasion or architectural disturbance. Histological grading was
performed according to the Gleason system (2). Prostatic carcinomas with
final score < 7 were considered low-grade; and, with final score ≥
7 were considered high-grade (3). Extraprostatic extension was diagnosed
according to Bostwick & Montironi (4), whenever cancer was seen in
adipose tissue, and corresponds to pT3a in the 2002 TNM staging system
(5). Seminal vesicle invasion (pT3b) was defined as an invasion of the
muscular wall, as described by Epstein et al. (6). Tumor extent was estimated
by use of a point-count method (7). Drawn on a sheet of paper, each quadrant
of the whole mount sections contained 8 equidistant points. During the
microscopic examination of the slides, the tumor area was drawn on the
correspondent quadrant seen on the paper. At the end of the examination,
the amount of positive points represented an estimate of the tumor extent.
The data were analyzed using the Mann-Whitney
test for comparison of independent samples and Spearman’s coefficient
of rank correlation to evaluate the association of Gleason score with
preoperative serum PSA and tumor extent. Time to biochemical (PSA) progression-free
survival was studied using the Kaplan-Meier product limit analysis; the
comparison between the groups was done using the log-rank test. Biochemical
progression was defined as PSA ≥ 0.2 ng/mL according to recent recommendation
of the American Urological Association (8). P value < 0.05 was considered
statistically significant. All statistical analyses were performed using
Statistica 5.5 (StatSoft, Inc., Tulsa, OK, USA).
RESULTS
We
compared the Gleason score on 187 needle biopsies with matched whole mounted
radical retropubic prostatectomy specimens. Twelve patients without information
about Gleason grade on needle biopsy were excluded, as well as one patient
without carcinoma in the prostatectomy specimen. There was exact correlation
in 47.1% of cases. The Gleason grade was higher (underestimated in the
biopsies) in 40.6% of cases and lower (overestimated in the biopsies)
in 12.3% of cases. The distribution in biopsies was 2.1%, 68.1%, 24.5%
and 5.3% of cases for Gleason score 2-5, 6, 7, and 8-10 respectively;
in the surgical specimens, distribution was 8.5%, 27.6%, 57.3%, and 6.5%
for the same Gleason scores respectively. From a total of 128 tumors with
a Gleason score 6 in the biopsy, 65/128 (50.8%) tumors were Gleason score
7 in the surgical specimen. From a total of 46 tumors with Gleason score
7 in the biopsy, 36/46 (78.3%) tumors were also 7 in the surgical specimen.
The data were analyzed using Spearman’s
coefficient of rank correlation to evaluate the association of Gleason
score in the surgical specimen with preoperative serum PSA in 199 patients.
One case showing no carcinoma in the prostatectomy specimen was excluded.
The median value of preoperative PSA was 10.46 ng/mL (range: 0.28 to 50
ng/mL). There was a positive correlation, albeit weak, of high-grade Gleason
score in the surgical specimens to higher preoperative serum PSA (p =
0.032; r = 0.152).
The tumor extent in the surgical specimens
ranged from 1 to 225 positive points (mean 37.5 positive points). Twenty-eight
cases were excluded from the analysis because the point count method was
not conducted, including one patient without carcinoma in the prostatectomy
specimen. More extensive tumors correlated with high-grade Gleason score
in the surgical specimens using Spearman’s coefficient of rank correlation
(p < 0.001; r = 0.524).
A total of 82 (41.2%) surgical specimens
showed positive margins and 54 (27.1%) extraprostatic extension (pT3a
and/or pT3b). There was statistically significant correlation between
high-grade Gleason score and positive margins (p < 0.001) as well as
extraprostatic extension (p < 0.001) using the Mann Whitney test. Only
one case was excluded for this analysis. The higher percentage of positive
margins (41.2%) versus extraprostatic extension (27.1%) may have occurred
because in surgical specimens with positive margins but no extraprostatic
extension the resulting stage is pT2+ or pT2x according to Epstein (9).
This stage denotes that elsewhere the tumor is organ confined, yet one
cannot determine whether there is extraprostatic extension in the region
of incision into the prostate because the edge of the prostate has been
left in the patient.
For the analysis of time to biochemical
progression, 14 patients without tests for serum PSA level following radical
prostatectomy were excluded. The mean and median follow-up of our study
(111 censored and 75 uncensored patients) was 30.3 and 25.5 months, respectively
(range 1 to 99 months). During the follow-up 75/186 (40.3%) patients developed
biochemical (PSA) progression.
Time of biochemical (PSA) progression-free
survival comparing 68 patients with Gleason score < 7 (low-grade) vs.
118 patients with Gleason score ≥ 7 (high-grade) in the surgical
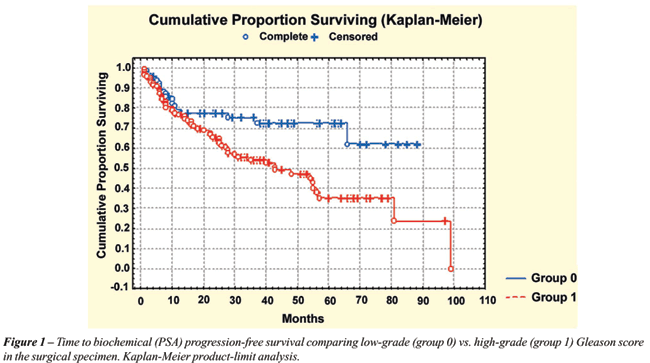
specimens was statistically significant (log-rank, p = 0.005) (Figure-1).
At 5 years, the PSA progression-free survival rates were 73% and 35% for
patients with low-grade and high-grade Gleason score in the surgical specimens,
respectively.
Time of biochemical (PSA) progression-free
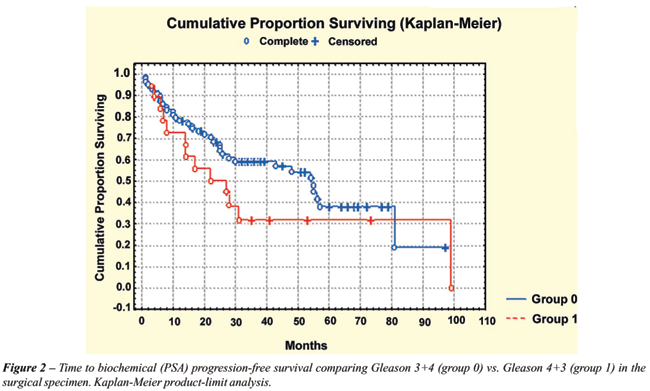
survival comparing 87 patients with Gleason score 3+4=7 vs. 19 patients
with Gleason score 4+3=7 was not statistically significant (log-rank,
p = 0.193) (Figure-2). At 5 years, the PSA progression-free survival rates
were 37% and 32% for patients with Gleason score 3+4=7 and Gleason score
4+3=7, respectively.
COMMENTS
There
have been several studies correlating core biopsy and radical prostatectomy
Gleason grade (10-13). Bostwick (12) found for Gleason score an exact
correlation in 35% of cases; in 40% of the cases the score was higher;
and, in 25% was lower in the surgical specimen. Spires et al. (11) found
an exact agreement between biopsy and excision in 58%, higher grade in
37.5% and lower grade in 4.5% of the cases. Steinberg et al. (10) at the
Johns Hopkins Hospital found an exact agreement of 58%. In our study the
exact agreement of Gleason score was 47.1%. Gleason score was higher (underestimated
on the biopsies) in 40.6% of cases and lower (overestimated on the biopsies)
in 12.3% of cases. In our series, half of tumors graded Gleason score
6 at biopsy were Gleason score 7 at surgery. This finding is similar to
the study by Pinthus et al. (13). We also compared the PSA progression-free
survival of tumors that were scored 6 on prostate biopsy but were 7 on
subsequent radical prostatectomy to those in tumors with a consistent
rating of Gleason score 7 at biopsy and surgery. Upgraded Gleason score
6 to 7 tumors had outcome similar to those of Gleason score 7 in both
biopsy and surgical specimen (log-rank, p = 0.359). This finding is also
similar to the study by Pinthus et al. (13).
For a low-grade Gleason score in biopsies,
urologists should consider the likelihood of tumor upgrading when contemplating
treatments. High-grade Gleason score in biopsies have a high predictive
value, which is enhanced when combined with other clinical parameters
(14). Nomograms typically include the Gleason score in the pretreatment
variables. Based on statistical modeling of cumulative, prospectively
accrued data from a large consecutive series of patients, the nomograms
have a reasonable discriminatory ability to counsel the urologist for
pretreatment bone scans, magnetic resonance imaging for seminal vesicle
involvement, and for lymph node dissection.
In general, serum PSA levels correlate with
larger tumor volume, advanced pathologic stages, and higher grades. Although
higher grade cancer produces less PSA per cell as compared to lower grade
tumors, overall, poorly differentiated tumors are associated with higher
PSA levels as these tumors tend to be larger and of more advanced stage
(15). In our study, there was a positive correlation, albeit weak, of
high-grade Gleason score in the surgical specimens to higher preoperative
serum PSA.
Many studies have been published that consistently
demonstrate the independent value of biopsy Gleason score in predicting
pathologic stage at radical prostatectomy (16,17). Based on these studies
of needle biopsies and radical prostatectomy specimens, several groups
have developed nomograms for predicting pathologic stage based on clinical
stage, serum prostate-specific antigen (PSA), and needle biopsy Gleason
score (18). The power of Gleason score in predicting outcome after radical
prostatectomy has been repeatedly demonstrated in numerous reports (19,20).
Nomograms using the radical prostatectomy Gleason score and other variables
are widely available. In patients with organ-confined, margin-negative
tumors, the data are particularly compelling. Patients with Gleason score
6 tumors infrequently fail (21). Clinicians can use these nomograms to
counsel individual patients and help them make important decisions regarding
their disease. The findings of our study support the importance of Gleason
grading in these nomograms. There was a statistically significant correlation
between high-grade Gleason score and positive margins (p < 0.001) as
well as extraprostatic extension (p < 0.001) using the Mann-Whitney
test.
During the follow-up 75/186 (40.3%) patients
developed biochemical (PSA) progression. This relatively high biochemical
recurrence rate for our series may be due to considering a prostate specific
antigen of > or = 0.2 ng/mL as a biochemical recurrence, based on recent
recommendations from the American Urological Association Prostate Guidelines
(8). According to the Panel, strict definitions for biochemical recurrence
are necessary to identify men at risk, but not necessarily to identify
disease progression. The purpose for establishing this standard is for
data reporting purposes. It is not intended to represent threshold value
for initiating treatment. The Panel acknowledges that the clinical decision
to initiate treatment will be dependent on multiple factors.
Time of biochemical (PSA) progression-free
survival comparing patients with Gleason score< 7 (low-grade) vs. Gleason
score ≥ 7 (high-grade) was statistically significant (log-rank,
p = 0.005). At 5 years, the PSA progression-free survival rates were 73%
and 35% for patients with low-grade and high-grade Gleason score, respectively.
These PSA progression-free survival rates were independent of margin status.
Considering only patients with negative margins, the biochemical progression-free
survival was 41% and 81% for high-grade and low-grade Gleason score, respectively
(log-rank, p = 0.019). For patients with prostate cancer, the Gleason
score remains one of the most important parameters, affecting all aspects
of their disease to include, potentially, the therapeutic stratification
or approach. The importance placed by clinicians and patients on the many
nomograms available cannot be overemphasized.
Tumors with a Gleason score of 7 have a
significantly worse prognosis than those with a Gleason score of 6. Given
the adverse prognosis associated with Gleason pattern 4, one would expect
that whether a tumor is Gleason score 3+4 or 4+3 would influence prognosis
(9). This issue has been controversial in the literature. No significant
survival advantage was reported for Gleason pattern 3+4 over 4+3 by Oefelein
et al. (22). Other investigators have shown that Gleason score 4+3 has
a worse prognosis than Gleason score 3+4 (23,24). Chan et al. (24) found
that the 5-year actuarial risk of progression was 15% and 40% for Gleason
score 3+4 and 4+3 tumors respectively. In our study, time of biochemical
(PSA) progression-free survival comparing patients with Gleason score
3+4=7 vs. Gleason score 4+3=7 was not statistically significant (log-rank,
p = 0.193). At 5 years, the PSA progression-free survival rates were 37%
and 32% for patients with Gleason score 3+4=7 and Gleason score 4+3=7,
respectively. We must consider, however, that this series has small numbers
and short follow-up for any definitive conclusion to be drawn.
CONCLUSIONS
High-grade Gleason score tumors in the surgical specimens show a positive correlation with higher preoperative serum PSA, more extensive tumors, positive margins, and more advanced pathologic staging. Most of the patients show either an exact Gleason score or a higher score in the surgical specimen in comparison to the biopsy. Half of tumors graded Gleason score 6 at biopsy were Gleason score 7 at surgery. Time of biochemical (PSA) progression-free survival following surgery is significantly shorter for patients with high-grade Gleason score in the surgical specimens. Upgraded Gleason score 6 tumors in the biopsy to 7 in surgery, have outcomes similar to those of tumors with Gleason score 7 in both biopsy and surgery. In our series, time of biochemical (PSA) progression-free survival between patients with Gleason score 3+4 vs. 4+3 was not statistically significant. The findings support the importance of Gleason grading for nomograms, which are used by clinicians to counsel individual patients and help them make important decisions regarding their disease. Thus, accurate Gleason grading is essential information in the pathology report.
CONFLICT OF INTEREST
None declared.
REFERENCES
- Mostofi, FK, Price EB Jr: Tumors of the Male Genital System, Atlas of Tumor Pathology, Second Series, Fascicle 8. Washington DC, Armed Forces Institute of Pathology. 1973; pp. 202-17.
- Gleason DF, Mellinger GT: Prediction of prognosis for prostatic adenocarcinoma by combined histological grading and clinical staging. J Urol. 1974; 111: 58-64.
- Gleason DF: Histologic grading of prostate cancer: a perspective. Hum Pathol. 1992; 23: 273-9.
- Bostwick DG, Montironi R: Evaluating radical prostatectomy specimens: therapeutic and prognostic importance. Virchows Arch. 1997; 430: 1-16.
- International Union Against Cancer: Prostate. In: Sobin LH, Wittekind Ch. (ed.), TNM Classification of Malignant Tumours, 6th ed. New York, Wiley-Liss. 2002; pp.184-7.
- Epstein JI, Carmichael M, Walsh PC: Adenocarcinoma of the prostate invading the seminal vesicle: definition and relation of tumor volume, grade and margins of resection to prognosis. J Urol. 1993; 149: 1040-5.
- Billis A, Magna LA, Ferreira U: Correlation between tumor extent in radical prostatectomies and preoperative PSA, histological grade, surgical margins, and extraprostatic extension: application of a new practical method for tumor extent evaluation. Int Braz J Urol. 2003; 29: 113-9; discussion 120.
- Cookson MS, Aus G, Burnett AL, Canby-Hagino ED, D’Amico AV, Dmochowski RR: Variation in the definition of biochemical recurrence in patients treated for localized prostate cancer: the American Urological Association Prostate Guidelines for Localized Prostate Cancer Update Panel report and recommendations for a standard in the reporting of surgical outcomes. J Urol. 2007; 177: 540-5.
- Epstein JI, Amin M, Boccon-Gibod L, Egevad L, Humphrey PA, Mikuz G, et al.: Prognostic factors and reporting of prostate carcinoma in radical prostatectomy and pelvic lymphadenectomy specimens. Scand J Urol Nephrol Suppl. 2005; 216: 34-63.
- Steinberg DM, Sauvageot J, Piantadosi S, Epstein JI: Correlation of prostate needle biopsy and radical prostatectomy Gleason grade in academic and community settings. Am J Surg Pathol. 1997; 21: 566-76.
- Spires SE, Cibull ML, Wood DP Jr, Miller S, Spires SM, Banks ER: Gleason histologic grading in prostatic carcinoma. Correlation of 18-gauge core biopsy with prostatectomy. Arch Pathol Lab Med. 1994; 118: 705-8.
- Bostwick DG: Gleason grading of prostatic needle biopsies. Correlation with grade in 316 matched prostatectomies. Am J Surg Pathol. 1994; 18: 796-803.
- Pinthus JH, Witkos M, Fleshner NE, Sweet J, Evans A, Jewett MA, et al.: Prostate cancers scored as Gleason 6 on prostate biopsy are frequently Gleason 7 tumors at radical prostatectomy: implication on outcome. J Urol. 2006; 176: 979-84; discussion 984.
- Amin M, Boccon-Gibod L, Egevad L, Epstein JI, Humphrey PA, Mikuz G, et al.: Prognostic and predictive factors and reporting of prostate carcinoma in prostate needle biopsy specimens. Scand J Urol Nephrol Suppl. 2005; 216: 20-33.
- Partin AW, Carter HB, Chan DW, Epstein JI, Oesterling JE, Rock RC, et al.: Prostate specific antigen in the staging of localized prostate cancer: influence of tumor differentiation, tumor volume and benign hyperplasia. J Urol. 1990; 143: 747-52.
- Partin AW, Kattan MW, Subong EN, Walsh PC, Wojno KJ, Oesterling JE, et al.: Combination of prostate-specific antigen, clinical stage, and Gleason score to predict pathological stage of localized prostate cancer. A multi-institutional update. JAMA. 1997; 277: 1445-51. Erratum in: JAMA 1997; 278: 118.
- Partin AW, Mangold LA, Lamm DM, Walsh PC, Epstein JI, Pearson JD: Contemporary update of prostate cancer staging nomograms (Partin Tables) for the new millennium. Urology. 2001; 58: 843-8.
- Han M, Partin AW: Nomograms for clinically localized prostate cancer. Part I: radical prostatectomy. Semin Urol Oncol. 2002; 20: 123-30.
- D’Amico AV, Whittington R, Malkowicz SB, Fondurulia J, Chen MH, Tomaszewski JE, et al.: The combination of preoperative prostate specific antigen and postoperative pathological findings to predict prostate specific antigen outcome in clinically localized prostate cancer. J Urol. 1998; 160: 2096-101.
- Kattan MW, Wheeler TM, Scardino PT: Postoperative nomogram for disease recurrence after radical prostatectomy for prostate cancer. J Clin Oncol. 1999; 17: 1499-507.
- Palisaar RJ, Graefen M, Karakiewicz PI, Hammerer PG, Huland E, Haese A, et al.: Assessment of clinical and pathologic characteristics predisposing to disease recurrence following radical prostatectomy in men with pathologically organ-confined prostate cancer. Eur Urol. 2002; 41: 155-61.
- Oefelein MG, Smith ND, Grayhack JT, Schaeffer AJ, McVary KT: Long-term results of radical retropubic prostatectomy in men with high grade carcinoma of the prostate. J Urol. 1997; 158: 1460-5.
- Han M, Partin AW, Pound CR, Epstein JI, Walsh PC: Long-term biochemical disease-free and cancer-specific survival following anatomic radical retropubic prostatectomy. The 15-year Johns Hopkins experience. Urol Clin North Am. 2001; 28: 555-65.
- Chan TY, Partin AW, Walsh PC, Epstein JI: Prognostic significance of Gleason score 3+4 versus Gleason score 4+3 tumor at radical prostatectomy. Urology. 2000; 56: 823-7.
____________________
Accepted
after revision:
September 1, 2007
_______________________
Correspondence address:
Dr. Athanase Billis
Dep. de Anatomia Patológica, FCM
UNICAMP, Caixa Postal 6111
Campinas, SP, 13084-971, Brazil
Fax: + 55 19 3289-3897
E-mail: athanase@fcm.unicamp.br