LAPAROSCOPIC
NEPHROPEXY EXPOSES A POSSIBLE UNDERLYING PATHOGENIC MECHANISM AND ALLOWS
SUCCESSFUL TREATMENT WITH TISSUE GLUING OF THE KIDNEY AND FIXATION OF
THE COLON TO THE LATERAL ABDOMINAL WALL
(
Download pdf )
doi: 10.1590/S1677-55382010000100003
JONAS WADSTROM, MICHAEL HAGGMAN
Department of Surgical Sciences, Uppsala University Hospital, Uppsala, Sweden
ABSTRACT
Objectives:
Surgical treatment of “Ren Mobilis” has historically been
associated with poor results and fairly high morbidity. We have used a
transperitoneal laparoscopic approach in order to minimize morbidity.
The goal of this study was to evaluate the success rate and to discuss
the possible pathogenic mechanism, which has implications for the surgical
strategy.
Materials and Methods: Seven women with
a right mobile kidney were examined by intravenous pyelogram and CT scans.
Symptoms were judged to emanate from the mobile kidney. Transperitoneal
laparoscopic nephropexy was performed. The surgical treatment consisted
of fixing the kidney to the dorsal abdominal wall using tissue glue (Tisseel®)
after diathermy coagulation of the surfaces to induce fibrosis. The right
colon was fixed with clips to the lateral abdominal wall, trapping the
kidney in place.
Results: In 6 of the cases, there was an
incomplete rotation of the ascending colon to the right side, allowing
the kidney to move freely. In one case, the kidney moved into a retroperitoneal
pocket of the mesocolon. The 6 cases with a lateral passage for the kidney
were symptom-free at follow-up (30-80 months), but in the 7th case the
patient’s kidney quickly loosened and she underwent an open reoperation,
after which she was symptom-free.
Conclusion: Our series demonstrates that
good results can be achieved with a transperitoneal laparoscopic approach,
but also indicates that there is a common pathogenic mechanism with incomplete
rotation of the ascending colon that can be corrected during surgery,
which might contribute to the good results.
Key
words: kidney; ptosis; pathology; laparoscopic surgery
Int Braz J Urol. 2010; 36: 10-7
INTRODUCTION
The
general definition of “Ren Mobilis” (nephroptosis) is that
the kidney descends two vertebral bodies or 5 cm when the body posture
changes from supine to an upright position. Surgical treatment of nephroptosis
by fixation or suspension of the kidney has been performed since Hahn’s
first report in 1881 (1). Numerous surgical techniques have been described
since, but the success rate has been low and the procedures have been
associated with fairly significant morbidity. The procedures were therefore
almost abandoned for many years but with the introduction of refined diagnostic
tools and minimally invasive surgery the diagnosis and surgical treatment
of nephroptosis has received renewed interest. Indeed, there are several
reports today of successful laparoscopic nephropexy with low morbidity
(2-6).
These reports have all focused on modalities
for fixation of the kidney by sutures, vaginal tape or gluing, etc. In
the present report, we describe a new a possibly pathogenic mechanism
behind the condition and a causal therapy of a combination of fixation
of the kidney in its normal anatomical position and attachment of the
right colon to the lateral abdominal wall. The latter appears to be important
since we found that in all cases there was malrotation or a long mesocolon
that allowed the kidney to move freely in a medial and caudal direction.
MATERIALS AND METHODS
Patient Demographics
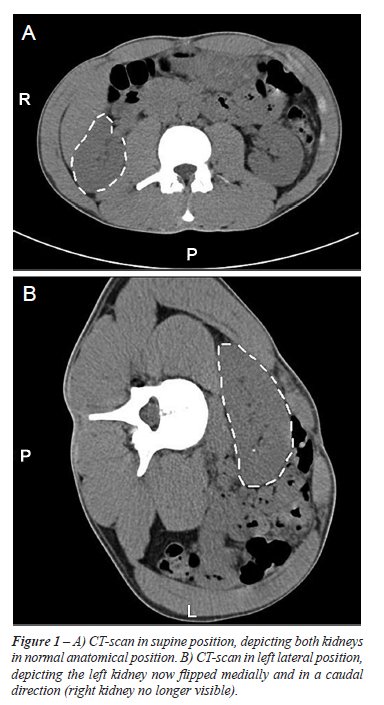
Between 1998 and 2004, 7 women, 19 to 57 years old (mean age 33), were referred to our department for surgery of a mobile kidney. All had right-sided symptoms and preoperative work-up with intravenous pyelography and CT scan showed a mobile right kidney, Figure-1. None of the patients had any previous history of urinary tract infections, hypertension or renal calculi. Patient characteristics are given in Table-1.
Laparoscopic Surgical
Technique
The
patients were positioned in a semi-lateral position with 45-degree rotation.
A transperitoneal approach was used for the procedure. Three 12 mm ports
were placed in the anterior axillary line and one in the flank.
With the patient rotated 45 degrees, it
was evident in all cases that there was an incomplete intestinal rotation
so that the right colon was flexed medially and the entire kidney was
immediately visible. The kidney was also freely movable in a medial and
caudal direction. The operation starts with incision of the posterior
peritoneum without having to incise the line of Toldt. The ureter is identified
and the perirenal fat dissected and the kidney completely mobilized.
Gerota’s fascia was in most cases
underdeveloped or absent. The posterior renal capsule was superficially
cauterized to induce postoperative scarring. The fatty tissue of the posterior
renal bed was treated in the same manner. A high viscosity fibrin sealant
(TisseelDuo Quick®, Baxter AG, Vienna, Austria) with prolonged setting
time was prepared by mixing the fibrinogen component with hyaluronic acid
(Healon®, Advanced Medical Optics, Inc., Santa Ana, CA) at a ratio
of 1:1. The thrombin component was diluted with saline to reach a concentration
of 40 U/mL. A final total volume of 4 mL was applied to the posterior
renal bed with an application catheter (Duplocath®, Baxter AG, Vienna,
Austria). The high viscosity combined with a prolonged setting time allowed
sufficient time to reposition the kidney without glue running off or instantaneous
setting of the glue. The kidney was then compressed to the posterior bed
for 5 minutes allowing the glue to set. After the kidney had been secured
in its anatomically correct position, the colon was attached to the lateral
abdominal wall and the right flexure to cover the kidney under the right
liver lobe. Fixation was achieved with clips (Endopath® EMS, Ethicon,
Cincinnati, USA).
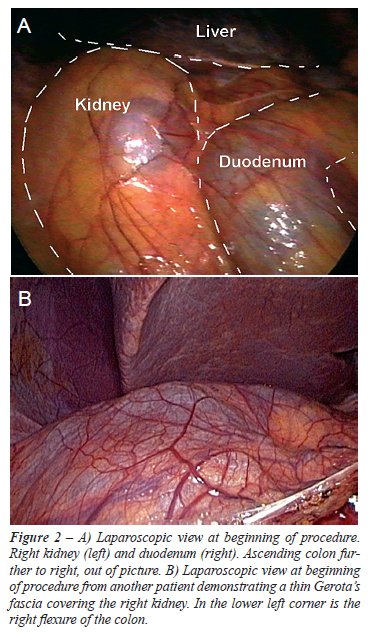
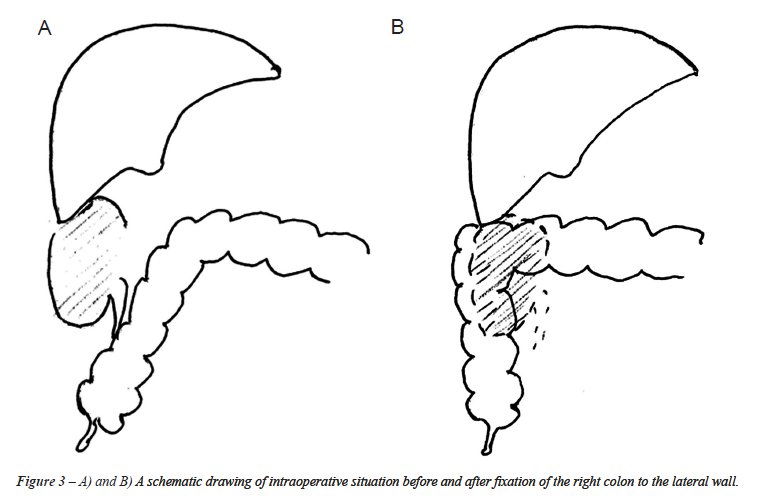
The intraoperative situs is depicted in
Figure-2A and B. A schematic drawing of intraoperative situs before and
after fixation of the right colon to the lateral wall is depicted in Figure-3.
Before allowing the patient to wake up after
surgery, a girdle was applied to the lower half of the abdomen to prevent
the kidney from sliding in a caudal direction.
Postoperatively the patients had bed rest for 24 hours to allow initial
scarring and fixation without subjecting the kidney to gravitational forces.
They were allowed oral feeding immediately.
Patients were followed up in the clinic 2-3 months postoperatively and
then by phone 1-4 years after their procedure.
RESULTS
Mean
operative time was 142 min. (100-145). In all cases, the immediate postoperative
course was uneventful, notably, no thromboembolic complications occurred
in spite of the 24 hours of bed rest in the dorsal position. Postoperative
hospitalization was 3.6 days (3-4), including 1 day of bed rest.
The 6 cases with a lateral passage for the
kidney were symptom-free at follow-up, but in the 7th case the patient’s
kidney quickly loosened and she underwent an open reoperation, this time
securing the kidney with traditional suturing in a retroperitoneal pocket,
after which no further symptoms occurred. Telephone follow-up at 30-80
months postoperatively revealed no further symptoms associated with the
kidney in the operated cases, though the patient who was reoperated with
an open procedure has had some scar-related problems in the flank incision.
Interestingly, one patient had complete relief of previous constipation
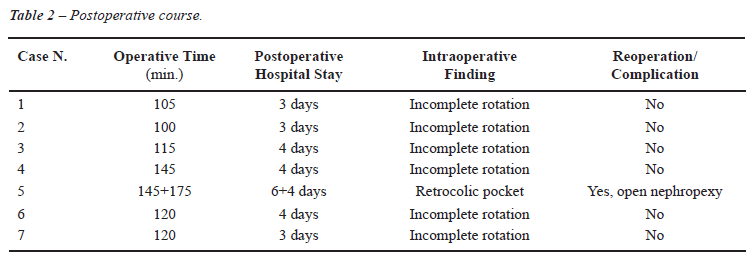
after her procedure. The postoperative course is summarized in Table-2.
COMMENTS
In
recent years, surgical treatment of nephoptosis has received renewed interest
and laparoscopic and retroperitoneoscopic techniques have been advocated
(2-13).
In these studies, treatment has in most
cases been successful with low surgical morbidity. Fixation of the kidney
has been achieved predominantly by suturing of the capsule to the posterior
abdominal wall. However, none of the studies have tried to analyze and
treat a pathogenic cause. Already in our first case, it was evident that
there was an incomplete rotation of the ascending colon. The colon was
not attached to the posterior-lateral abdominal wall. This allowed direct
visualization of the kidney without loosening the colon from the lateral
abdominal wall. There was a long and loose right mesocolon and the right
flexure was freely movable to the left side of the abdomen. In addition,
Gerota’s fascia was weak or missing. This allowed the kidney to
move freely in a caudal and medial direction (Figure-1). We therefore
decided intraoperatively not only to fix the kidney in its anatomical
location, but also to attach the colon to the lateral abdominal wall and
the right flexure so that it covered the kidney below the right lobe of
the liver (Figure-3). The same anatomical variant was then found in all
subsequent cases but one.
Instead of fixating the kidney with sutures,
we tried to induce scarring by treating the posterior capsule of the kidney
and the fatty tissue of the renal bed using cautery. To temporarily hold
the kidney in position, we used a viscosity-enhanced fibrin sealant with
a prolonged setting time. This allowed sufficient time to turn the kidney
back into its anatomical position before the glue ran off or started to
set. We believe the gluing is preferable to suturing since it is sometimes
difficult to suture without risking injury of the kidney or the ilio-hypogastric
or genitofemoral nerves. None of our patients had any neuralgia, which
is not an uncommon finding when sutures are used (14). An alternative
method could be to use polymer clips if the perirenal fat and tissue is
strong. This would lower costs (15).
Fibrin sealants are absorbed and lose their
strength in a fairly short time. The scarring induced by cauterization
of the renal capsule and the fatty tissue of the renal bed is unlikely
to be strong enough to hold the kidney in its anatomical position. We
therefore believe that attaching the right colon to the lateral abdominal
wall and covering the kidney with the right flexure of the colon is an
essential part of a successful treatment approach. This view is further
supported by the interoperative finding that insufficient rotation of
the colon appears to be an underlying pathogenic mechanism for nephroptosis.
Most previous open surgical techniques for
the treatment of nephroptosis and most of the minimally invasive techniques
have been performed by a retroperitoneal approach (7-13). The presently
proposed pathogenic mechanism is not revealed and cannot be corrected
with a retroperitoneal approach and could thus be an explanation for the
poor results in the past. None of the authors who have used a transperitoneal
technique have mentioned the finding of an incomplete rotation of the
colon (2-6). It should, however, be noted that Hübner and Plas (4,5)
offer the following description: “Owing to the absence of the fatty
capsular tissue, the kidney was easily identified”, a condition,
which is very similar to what we describe in Figure-1. In describing the
operative procedure, they do not mention mobilizing the colon but rather
add that, directly after creating the pneumoperitoneum: “The kidney
[is] easily identified. The peritoneum and Gerota’s fascia are opened
in a T-shaped incision.” Fornara et al. (6) give the following description:
“The line of Toldt was incised and the ureter was identified. The
perirenal fat was then dissected and the kidney was completely mobilized.”
The authors do not describe the necessity of mobilizing the ascending
colon. It is thus possible that the previous authors who used a transperitoneal
technique also encountered an incomplete rotation of the ascending colon
but did not realize that it could be a contributing pathogenic mechanism.
A further indication that there is a common pathogenic mechanism is the
intriguing fact that the condition is almost always on the right side.
It is also of interest to note that Curtis et al. (16) have demonstrated
that in cases of renal ectopia the distal ascending colon is visualized
medial to the anterior part of the right colonic flexure and proximal
to the transverse colon. These authors relate this finding to the fact
that during fetal development a normal ascent and fixation of the kidney
is necessary for the formation of the perirenal fascia and that this process
is intimately associated with the formation of extraperitoneal fascial
planes and colonic supporting structures. Cases are also described where
there is a malrotation of the left colon, which is associated with a left-sided
pelvic kidney (17). There are thus several pieces of circumstantial evidence
indicating that incomplete rotation of the colon plays a pathogenic role
in “Ren Mobilis” and that fixation of the colon in its correct
anatomical position is of advantage in order to achieve an optimal surgical
result.
CONCLUSION
Surgical treatment for nephroptosis should only be considered after a careful and cautious preoperative evaluation. The findings of our present study, however, indicate that a transperitoneal laparoscopic approach can give good results with a low morbidity and allow correction for a possible pathogenic mechanism underlying the condition. If the results can be confirmed in larger studies, this might lead to renewed interest in the condition and offer better treatment options for a number of patients currently dissuaded from surgical treatment.
ACKNOWLEDGEMENT
Funding for this study was received from Uppsala University.
CONFLICT OF INTEREST
None declared.
REFERENCES
- Barber NJ, Thompson PM: Nephroptosis and nephropexy--hung up on the past? Eur Urol. 2004; 46: 428-33.
- Urban DA, Clayman RV, Kerbl K, Figenshau RS, McDougall EM: Laparoscopic nephropexy for symptomatic nephroptosis: initial case report. J Endourol. 1993; 7: 27-30.
- Elashry OM, Nakada SY, McDougall EM, Clayman RV: Laparoscopic nephropexy: Washington University experience. J Urol. 1995; 154: 1655-9.
- Hübner WA, Schramek P, Pflüger H: Laparoscopic nephropexy. J Urol. 1994; 152: 1184-7.
- Plas E, Daha K, Riedl CR, Hübner WA, Pflüger H: Long-term followup after laparoscopic nephropexy for symptomatic nephroptosis. J Urol. 2001; 166: 449-52.
- Fornara P, Doehn C, Jocham D: Laparoscopic nephropexy: 3-year experience. J Urol. 1997; 158: 1679-83.
- El-Moula MG, Izaki H, Kishimoto T, Takahashi M, Fukumori T, Kanayama HO: Laparoscopic nephropexy. J Laparoendosc Adv Surg Tech A. 2008; 18: 230-6.
- Rassweiler JJ, Frede T, Recker F, Stock C, Seemann O, Alken P: Retroperitoneal laparoscopic nephropexy. Urol Clin North Am. 2001; 28: 137-44.
- Wyler SF, Sulser T, Casella R, Hauri D, Bachmann A: Retroperitoneoscopic nephropexy for symptomatic nephroptosis using a modified three-point fixation technique. Urology. 2005; 66: 644-8.
- Chueh SC, Hsieh JT, Chen J, Young YL, Chen SC, Tu YP: Retroperitoneoscopic nephropexy for symptomatic nephroptosis. Surg Endosc. 2002; 16: 1603-7.
- Ichikawa T, Yamada D, Takao A, Saegusa M, Aramaki K, Kumon H: Retroperitoneoscopic nephropexy for symptomatic nephroptosis. J Endourol. 2003; 17: 767-70.
- Matsui Y, Matsuta Y, Okubo K, Yoshimura K, Terai A, Arai Y: Laparoscopic nephropexy: treatment outcome and quality of life. Int J Urol. 2004; 11: 1-6.
- Gözen AS, Rassweiler JJ, Neuwinger F, Bross S, Teber D, Alken P, et al.: Long-term outcome of laparoscopic retroperitoneal nephropexy. J Endourol. 2008; 22: 2263-7.
- Hagmaier V, Heberer M, Leibundgut B, Ferstl A, Buser S, Schoenenberger GA, et al.: Long-term observations on different methods of nephropexy. Helv Chir Acta. 1979; 46: 351-5.
- Tunc L, Yesil S, Guneri C, Biri H, Ure I, Bozkirli I: The use of polymer clips in transperitoneal laparoscopic nephropexy. Surg Laparosc Endosc Percutan Tech. 2008; 18: 124-6.
- Curtis JA, Sadhu V, Steiner RM: Malposition of the colon in right renal agenesis, ectopia, and anterior nephrectomy. AJR Am J Roentgenol. 1977; 129: 845-50.
- Vanmuylder N, Rooze M, Louryan S: A case with undescended testis, left pelvic kidney and gut malrotation. Surg Radiol Anat. 2005; 27: 382-4.
____________________
Accepted after revision:
September 22, 2009
_______________________
Correspondence address:
Dr. Jonas Wadström
Surgical Department
Rikshospitalet University Hospital
0027, Oslo, Norway
Fax: + 47 2307-0510
E-mail: jonas.wadstrom@rikshospitalet.no
EDITORIAL COMMENT
Ever
since the existence of surgical therapy of nephroptosis it has always
been a subject of discussion. A partly uncritical jubilation for surgery
has led to the fact, that nephropexy was the most performed urological
operation in the beginning of the 20th Century with up to 200 different
operative variations. Laparoscopy has been reported recently as a minimally
invasive approach for nephropexy. The article of Wadström and Häggman
offers a new possible underlying pathogenic mechanism for the nephroptosis.
The explanation and the surgical solution seem to be very interesting
and impressive.
“For all those, who produce urinary
obstruction and those with a beginning dilation, nephropexy still has
an efficient justification and may - correctly performed - give much blessings”.
Nothing needs to be added to this statement of Professor Voelcker from
Halle in the year 1911.
Dr.
Martin Hatzinger
Klinik fur Urologie
Markushospital
Frankfurt, Germany
E-mail: martin.hatzinger@fdk.info
EDITORIAL COMMENT
“Nephroptosis is a phenomenon that
has been known about for centuries. After the first successful surgery
for nephroptosis performed by Eugen Hahn (1) surgical therapy of nephroptosis
has always been a subject of discussion with up to 200 different surgical
variations (2).
In one study, excessive kidney mobility is detected in almost 30 % of
healthy subjects without being able to directly connect the known pathologies
which were reported in the literature for this phenomenon (3). Can we
accept the neptroptosis as a normal variation of the kidney localization?
“An interesting paper published in
J Endourol has reported about laparoscopic nepropexy for autosomal dominant
polycystic kidney diseases for reducing the related pain and giving some
tips about the reason and possible pathology (4).
The main goal of the operation is to achieve
permanent fixation of the kidney to ensure that the urinary passage remains
unobstructed and patient stay pain free during the follow-up. This goal
can be achieved using different surgical methods, but the precise etiopathology
continues to be unclear, even when the fixation of the kidney due to fat,
muscle, fascia and tissue. One issue is clear, that we need long time
follow-up data and additional investigation about the newly described
techniques with excellent short term results.
One can justifiably describe laparoscopic
nephropexy as a suitable, established method for treating symptomatic
nephroptosis - and it is one that can be of great therapeutic value to
patients when the patients are selected carefully. In this way, laparoscopic
nephropexy can help patients become symptom free with an improved quality
of life and preserve the kidney from long-term damage.
REFERENCES
- Hahn E: Die operative Behandlung der beweglichen Niere durch Fixation. Zbl Chir. 1881; 8: 449-52.
- Hatzinger M, Langbein S, de la Rosette J, Sohn M, Alken P: Nephropexy in the course of time. Aspects of an historical surgical technique. Urologe A. 2007; 46: 166-9.
- Thomson WN, Innes JA, Munro JF, Geddes AM, Prescott RJ, Murdoch JM: Renal mobility in women attending a pyelonephritis clinic and in controls. Br J Urol. 1978; 50: 73-5.
- Casale P, Meyers K, Kaplan B: Follow-up for laparoscopic renal denervation and nephropexy for autosomal dominant polycystic kidney disease-related pain in pediatrics. J Endourol. 2008; 22: 991-3.
Dr.
Dogu Teber
Department of Urology
SLK-Kliniken Heilbronn
Heilbronn, Germany
E-mail: dogu.teber@slk-kliniken.de