Does
Tumor Extent on Needle Prostatic Biopsies Influence the Value of Perineural
Invasion to Predict Pathologic Stage > T2 in Radical Prostatectomies?
(
Download pdf )
Clinical Urology
doi: 10.1590/S1677-55382010000400007
Athanase Billis, Maisa M. de Quintal, Luciana Meirelles, Leandro L. L. Freitas, Luis A. Magna, Ubirajara Ferreira
Department of Anatomic Pathology (AB, MMQ, LM, LLLF), Department of Medical Genetics/Biostatistics (LAM), and Department of Urology (UF), School of Medicine, University of Campinas (Unicamp), Campinas, SP, Brazil
ABSTRACT
Purpose:
Perineural invasion (PNI) on needle prostatic biopsies (NPB) has been
controversial as a marker of extraprostatic extension and consequently
for planning of nerve-sparing radical prostatectomy (RP). The aim of this
study was to find whether tumor extent on NPB influences the value of
PNI to predict stage > pT2 on RP.
Materials and Methods: This retrospective
study was based on 264 consecutive patients submitted to radical retropubic
prostatectomy. Their NPB were matched with whole-mount processed and totally
embedded surgical specimens. Tumor extent on NPB was evaluated as the
percentage of linear tissue in mm containing carcinoma in all cores. Considering
the median value, patients were stratified into 2 groups: harboring less
or more extensive tumors on NPB. Univariate and multivariate logistic
regression analyses were used to relate stage > pT2 to PNI and other
clinical and pathological variables.
Results: In patients with more extensive
tumors, PNI was predictive of stage > pT2 in univariate analysis but
not in multivariate analysis. In less extensive tumors, PNI showed no
association between any clinical or pathological variables studied; no
difference in the time to biochemical progression-free status compared
to patients without PNI; and, no predictive value for pathological stage
> pT2 on both univariate and multivariate analyses.
Conclusion: Tumor extent on NPB influences
the predictive value of PNI for pathologic stage > pT2 on RP. With
a higher number of small tumors currently detected, there is no evidence
that perineural invasion should influence the decision on preservation
of the nerve during radical prostatectomy.
Key
words: prostate; prostatic neoplasms; biopsy; needle; prostatectomy;
prognosis
Int Braz J Urol. 2010; 36: 439-49
INTRODUCTION
Perineural invasion (PNI) on needle prostatic biopsies as a marker of extraprostatic extension has been controversial (1-15). In almost all studies, perineural invasion has been related to extraprostatic extension in univariate analysis but in only a few studies in multivariate analysis. The practical importance relates to the decision of whether to sacrifice part or all of the neurovascular bundle on the side of the biopsy with PNI when planning nerve-sparing radical prostatectomy. The aim of this study was to determine whether tumor extent on needle biopsies significantly influences the value of PNI to predict stage > pT2 (pT3a and/or pT3b) on radical prostatectomies.
MATERIALS AND METHODS
This
retrospective study was based on 264 consecutive patients submitted to
radical retropubic prostatectomy by one surgeon (UF) in the period 1997
to 2008 due to clinically localized (T1c or T2) prostate adenocarcinoma.
Their needle prostatic biopsies (mean 9 cores per biopsy) were matched
with whole-mount processed and totally embedded surgical specimens. A
mean of 32 paraffin blocks were processed, and 6µm sections from
each block were stained with hematoxylin and eosin. PNI was considered
as prostate cancer extension along the perineural sheath (Figure-1). The
presence of any PNI, regardless of amount, was recorded as positive for
PNI. Positive surgical margins were defined as cancer cells touching the
inked surface of the prostate. Extraprostatic extension (pT3a) was diagnosed
whenever cancer was seen in adipose tissue and, in case of desmoplastic
response, whenever a protuberance corresponding to extension of tumor
into the periprostatic tissue was seen (16). Seminal vesicle invasion
(pT3b) was defined as invasion of the muscular wall (17).
Extent on needle biopsy was evaluated as
the percentage of linear tissue in mm containing carcinoma. Considering
the median value of extent, biopsies were stratified into 2 equal groups:
132 biopsies with less extensive and 132 biopsies with more extensive
tumors. Tumor extent on radical prostatectomy was estimated by use of
a point-count method previously described (18,19). Grading was according
to the standard Gleason system (20,21). All pathological findings were
evaluated by one senior uropathologist (AB).
Clinical variables analyzed included preoperative serum prostate-specific
antigen (PSA), age and clinical stage (T1c or T2). Total serum PSA was
measured utilizing previous validated Immulite® PSA kit. Biochemical
progression was defined as PSA = 0.2 ng/mL according to recommendation
of the American Urological Association (22). After radical prostatectomy,
serum PSA was drawn every 3 months during the first year, every 6 months
during the second year, and annually thereafter. The mean and median follow-up
of the patients was 28 and 20 months, respectively. No patient of this
series was treated before or after surgery.
The data were analyzed using the Mann-Whitney
test for comparison of means and the Fishers exact test for comparison
of proportions. Time to biochemical (PSA) progression-free outcome was
studied using the Kaplan-Meier product-limit analysis; the comparison
between the groups was done using the Log-rank test. Univariate and multivariate
analyses using a logistic regression model was used to relate the outcomes
of prostatectomy stage > pT2 to several clinical and pathological variables.
Two-sided P value < 0.05 was considered statistically significant.
All statistical analyses were performed using the commercially available
SPSS 15.0.
RESULTS
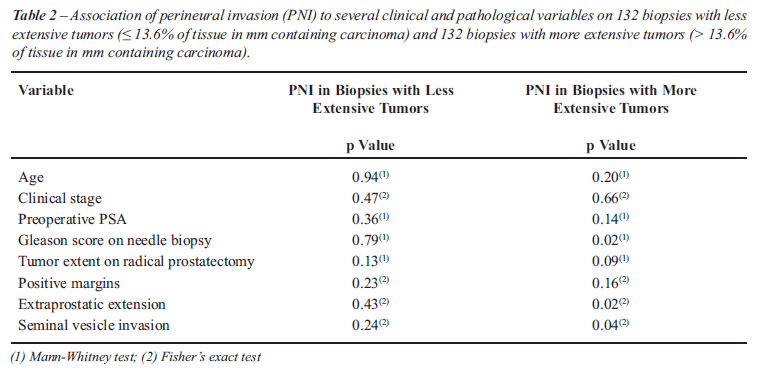
Table-1 shows the clinical and pathological findings of the 264 patients studied. Perineural invasion was present in 48/264 (18.2%) biopsies. The median value of tumor extent on biopsy was 13.6%. The frequency of PNI in the group with less extent tumors (= 13.6% of tissue in mm containing carcinoma) was 10.6%; in the group with more extensive tumors (> 13.6% of tissue in mm containing carcinoma) the frequency of PNI was 25.8%.

Table-2 shows the association of PNI to
several clinical and pathological variables comparing patients with less
extensive and more extensive tumors. On biopsies showing more extensive
tumors and PNI, patients showed statistically significant higher Gleason
score on biopsy (p = 0.02), more extraprostatic extension (p = 0.02),
and more seminal vesicle invasion (p = 0.04). On biopsies showing less
extensive tumors and PNI, there was no statistical significant association
to all variables studied.
Figures-2 and 3 show the Kaplan-Meier biochemical
progression-free survival curve following radical prostatectomy. In patients
with less extensive tumors on biopsy (Figure-2), at 5 years, 71% of the
patients without PNI were free of biochemical progression compared to
83% of the patients with PNI. There was no statistically significant difference
between the groups (log-rank, p = 0.24). In patients with more extensive
tumors on biopsy (Figure-3), at 5 years, 43% of the patients without PNI
were free of biochemical progression compared to 33% of the patients with
PNI. There was no statistically significant difference between the groups
(Log-rank, p = 0.26).
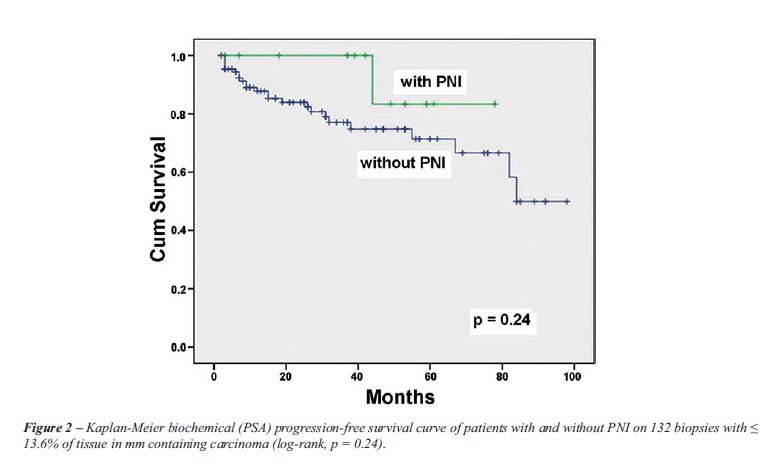
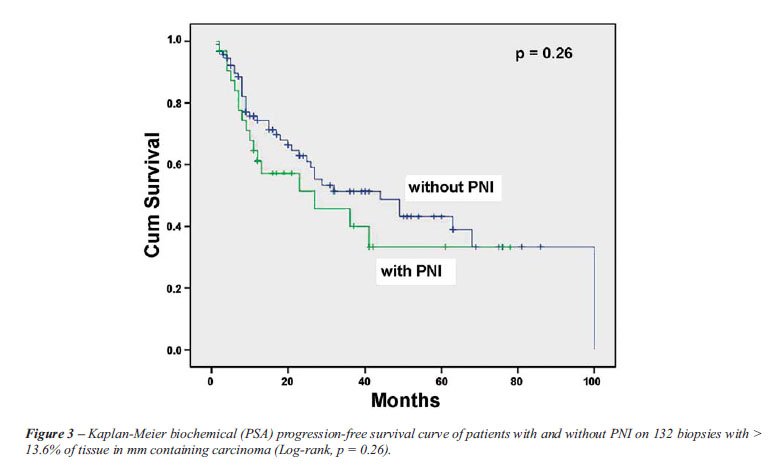
Table-3 shows the univariate and multivariate
logistic regression analyses of several clinical and pathological variables
predictive of pathological stage > T2 on radical prostatectomy in patients
with less extensive tumors on biopsy. PNI was not predictive of stage
> pT2 on both analyses. On univariate analysis, only extent of cancer
and Gleason score on biopsy were predictive. On multivariate analysis,
extent of tumor on biopsy was statistically significant and Gleason score
on the level of significance.
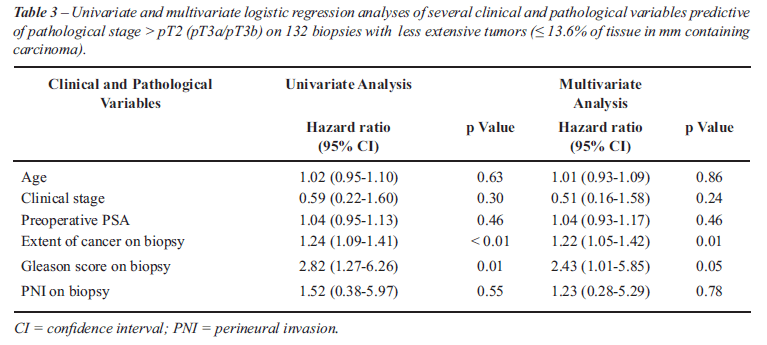
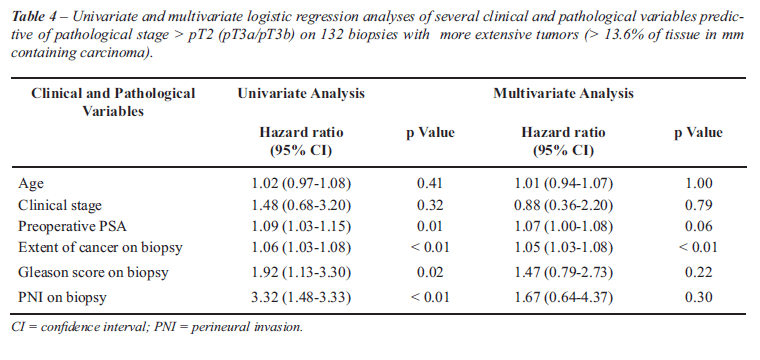
Table-4 shows the univariate and multivariate
logistic regression analyses of several clinical and pathological variables
predictive of pathological stage > T2 on radical prostatectomy in patients
with more extensive tumors on biopsy. PNI was predictive of stage >
pT2 only on univariate analysis. Other variables statistically significant
on univariate analysis were preoperative PSA, tumor extent on needle biopsy,
and Gleason score on biopsy. On multivariate analysis, only extent of
tumor on needle biopsy was an independent predictor of stage > pT2
(pT3a and/or pT3b) on radical prostatectomy.
COMMENTS
Our
study showed that extent of tumor influences the value of PNI to predict
pathological stage > T2 on radical prostatectomies. In patients with
less extensive tumors on biopsy (= 13.6% of tissue in mm containing carcinoma)
and PNI, there was no association to any one clinical or pathological
variable studied; no difference in the time to biochemical (PSA) progression-free
outcome compared to patients without PNI; and, no predictive value for
pathological stage > pT2 on both univariate and multivariate analysis.
The only study to mention the influence of extent of tumor to the predictive
value of PNI is Rubins’s et al. (6). In their study, PNI revealed
a significant association with stage pT3 on univariate analysis. However,
on multivariate analysis the association was significant only when the
greatest percentage of any single biopsy involved by prostate carcinoma
and the total percentage of cancer in all cores were not considered, due
to a significant interaction between these measures and PNI.
The findings with more extensive tumors
on biopsy are in accordance with most of the studies in the literature.
Egan and Bostwick (2) found on univariate analysis that PNI on needle
biopsy was significantly associated to extraprostatic extension and seminal
vesicle invasion. On multivariate analysis, however, only preoperative
PSA, proportion of the biopsy involved by cancer, and Gleason score were
significant. Ukimura et al. (4) found that PNI on biopsy was a good predictor
among others studied for extraprostatic extension on univariate analysis
but not on multivariate analysis. In the study by Vargas et al. (5) PNI
was not an independent predictor of extraprostatic extension when PSA
was included.
D’Amico et al. (7) evaluated the clinical
use of PNI on biopsy for predicting time to PSA failure following radical
prostatectomy of 750 men with clinically localized or PSA detected prostate
cancer. The presence of PNI on biopsy was not a significant predictor
of PSA outcome following RP for patients in the intermediate or high-risk
group. O’Malley et al. (9) compared 78 biopsies with PNI with 78
matched controls without PNI and were unable to show that PNI on needle
biopsy influences long-term tumor-free survival. Freedland et al. (10)
studied 190 men who underwent radical prostatectomy. Percent of tissue
with cancer on biopsy was the strongest predictor of biochemical recurrence
on multivariate analysis. PNI was not an independent predictor of either
adverse pathology or biochemical failure.
In Bismar’s et al. study (11) neither
presence nor absence of perineural nor number nor percentage of positive
nerves were related to pathologic stage on univariate or multivariate
analyses. In Tsuzuki’s et al. study (12) PSA, Gleason score, digital
rectal examination, percent of side specific cores with tumor and average
percent involvement of each positive core but not PNI were found to be
statistically significant independent predictors of extraprostatic extension
in the region of the neurovascular bundle. Studying 452 consecutive patients
undergoing radical retropubic prostatectomy by a single surgeon, Cannon
et al. (13) concluded that although biopsy PNI alone was associated with
a higher probability of extraprostatic extension, it was not predictive
of bilateral nerve-sparing technique or a positive surgical margin in
an individual patient.
In other studies, however, PNI was an independent
predictor of final pathologic stage. de la Taille et al. (3) found that
PNI, PSA and Gleason score on the biopsy independently predicted stage
pT3 disease. The authors concluded that PNI is an important preoperative
predictor of pathologic stage and should be reported when adenocarcinoma
is diagnosed on prostate needle biopsies. In the Sebo et al. study (8)
joint predictors of extraprostatic extension were the percent cores positive
for carcinoma, Gleason score of 7, Gleason score of 8 or 9, serum PSA
and PNI. In the Loeb et al. study (15), PNI was significantly associated
with aggressive pathology and biochemical progression. On multivariate
analysis, PNI was significantly associated with extraprostatic extension
and seminal vesicle invasion. Bastacky et al. (1) studied 302 needle prostatic
biopsies and found a sensitivity of 27% and a specificity of 96% for PNI
to predict extraprostatic extension. They concluded that measuring PNI
on needle biopsy helps identify extraprostatic extension and may help
in planning nerve-sparing radical prostatectomy in the decision of whether
to sacrifice part or all of the neurovascular bundle on the side of the
biopsy. These authors did not study the predictive value on multivariate
analysis.
Some factors may be responsible for the
discrepancies in the literature: the number of cores examined per case
may influence the rate of detection of PNI, the different methods of processing
and submitting tissue from radical prostatectomy specimens (completely
vs. partially embedded prostate glands) may contribute to different rates
of detection of extraprostatic carcinoma, different definitions of PNI
on needle biopsy tissue and extraprostatic extension, and different values
of PSA for evaluation of biochemical progression-free outcome following
surgery. In a search from January 1990 to December 2005 using MEDLINE,
Embase, and the Web of Knowledge, Harnden et al. (14) performed a systematic
review of studies that examined the association between perineural invasion
and prostate cancer recurrence. These authors concluded that a considerable
variation in study design, execution, and reporting precluded meta-analysis
and quantitative risk estimation.
The frequency of perineural invasion on
needle prostatic biopsies varies from 11% to 38% (1-6,8,10,11). Considering
all biopsies in our study the frequency was 18%. In the group with less
extensive tumors the frequency was 10.6% and in the group with more extensive
tumors 25.8%. It is worth noting that in less extensive tumors the frequency
of PNI is in the lower range of the frequency found in the literature.
Studying very small tumors on biopsies (less than 1 mm in length in only
one core), Thorson et al. (23) still found a frequency of 2% of PNI. We
have also seen PNI invasion in autopsied patients with very small incidentally
found histologic carcinomas. These findings probably suggest that PNI
may be an early event. This suggestion is shared by Byar and Mostofi (24).
The authors studied 208 total prostates removed using the step-section
technique for early carcinoma of the prostate. The high frequency of PNI
found (84.1%) suggested by the authors that the phenomenon occurs early
in the course of the disease. The similarity in the survival rates for
cases with and without PNI indicated little if any prognostic significance.
Our study may have some limitations. All
patients were submitted to radical prostatectomy, and therefore we were
unable to determine any different effect of PNI on biochemical progression-free
outcome with other forms of treatment, such as watchful waiting or radiotherapy.
Another limitation could be related to the fact that we only recorded
the presence or absence of PNI on needle biopsy but did not quantify the
extent. In addition, the mean follow-up is relatively short, and it is
possible that greater differences could occur with additional follow-up.
A strength of the study could be the homogeneity of the study population
which was comprised of consecutive patients treated by one expert surgeon
limiting any influence of variability in surgical technique and the pathological
evaluation also by one senior uropathologist.
In summary, tumor extent on needle biopsies
influences the predictive value of PNI for pathologic stage > pT2 (pT3a
and/or pT3b) on radical prostatectomies. In patients with more extensive
tumors on needle biopsy, PNI predicted pathologic stage > pT2 on radical
prostatectomy on univariate analysis but on multivariate analysis did
not show independent predictive value. This finding is in accordance to
most of the studies reported in the literature. In patients with less
extensive tumors on biopsy (= 13.6% of tissue in mm containing carcinoma)
and PNI, there was no association between any clinical or pathological
variables studied; no difference in the time to biochemical (PSA) progression-free
outcome comparing to patients without PNI; and, no predictive value for
pathological stage > pT2 on both univariate and multivariate analysis.
With a higher number of small tumors currently detected, there is no evidence
that perineural invasion should influence the decision on preservation
of the nerve during radical prostatectomy.
CONFLICT OF INTEREST
None declared.
REFERENCES
- Bastacky SI, Walsh PC, Epstein JI: Relationship between perineural tumor invasion on needle biopsy and radical prostatectomy capsular penetration in clinical stage B adenocarcinoma of the prostate. Am J Surg Pathol. 1993; 17: 336-41.
- Egan AJ, Bostwick DG: Prediction of extraprostatic extension of prostate cancer based on needle biopsy findings: perineural invasion lacks significance on multivariate analysis. Am J Surg Pathol. 1997; 21: 1496-500.
- de la Taille A, Katz A, Bagiella E, Olsson CA, O’Toole KM, Rubin MA: Perineural invasion on prostate needle biopsy: an independent predictor of final pathologic stage. Urology. 1999; 54: 1039-43.
- Ukimura O, Troncoso P, Ramirez EI, Babaian RJ: Prostate cancer staging: correlation between ultrasound determined tumor contact length and pathologically confirmed extraprostatic extension. J Urol. 1998; 159: 1251-9.
- Vargas SO, Jiroutek M, Welch WR, Nucci MR, D’Amico AV, Renshaw AA: Perineural invasion in prostate needle biopsy specimens. Correlation with extraprostatic extension at resection. Am J Clin Pathol. 1999; 111: 223-8.
- Rubin MA, Bassily N, Sanda M, Montie J, Strawderman MS, Wojno K: Relationship and significance of greatest percentage of tumor and perineural invasion on needle biopsy in prostatic adenocarcinoma. Am J Surg Pathol. 2000; 24: 183-9.
- D’Amico AV, Wu Y, Chen MH, Nash M, Renshaw AA, Richie JP: Perineural invasion as a predictor of biochemical outcome following radical prostatectomy for select men with clinically localized prostate cancer. J Urol. 2001; 165: 126-9.
- Sebo TJ, Cheville JC, Riehle DL, Lohse CM, Pankratz VS, Myers RP, et al.: Predicting prostate carcinoma volume and stage at radical prostatectomy by assessing needle biopsy specimens for percent surface area and cores positive for carcinoma, perineural invasion, Gleason score, DNA ploidy and proliferation, and preoperative serum prostate specific antigen: a report of 454 cases. Cancer. 2001; 91: 2196-204.
- O’Malley KJ, Pound CR, Walsh PC, Epstein JI, Partin AW: Influence of biopsy perineural invasion on long-term biochemical disease-free survival after radical prostatectomy. Urology. 2002; 59: 85-90.
- Freedland SJ, Csathy GS, Dorey F, Aronson WJ: Percent prostate needle biopsy tissue with cancer is more predictive of biochemical failure or adverse pathology after radical prostatectomy than prostate specific antigen or Gleason score. J Urol. 2002; 167: 516-20.
- Bismar TA, Lewis JS Jr, Vollmer RT, Humphrey PA: Multiple measures of carcinoma extent versus perineural invasion in prostate needle biopsy tissue in prediction of pathologic stage in a screening population. Am J Surg Pathol. 2003; 27: 432-40.
- Tsuzuki T, Hernandez DJ, Aydin H, Trock B, Walsh PC, Epstein JI: Prediction of extraprostatic extension in the neurovascular bundle based on prostate needle biopsy pathology, serum prostate specific antigen and digital rectal examination. J Urol. 2005; 173: 450-3.
- Cannon GM Jr, Pound CR, Landsittel DP, Bastacky SI, Dhir R, Becich MJ, et al.: Perineural invasion in prostate cancer biopsies is not associated with higher rates of positive surgical margins. Prostate. 2005; 63: 336-40.
- Harnden P, Shelley MD, Clements H, Coles B, Tyndale-Biscoe RS, Naylor B, et al.: The prognostic significance of perineural invasion in prostatic cancer biopsies: a systematic review. Cancer. 2007; 109: 13-24.
- Loeb S, Epstein JI, Humphreys EB, Walsh PC: Does perineural invasion on prostate biopsy predict adverse prostatectomy outcomes? BJU Int. 2009; 19. [Epub ahead of print]
- Bostwick DG, Montironi R: Evaluating radical prostatectomy specimens: therapeutic and prognostic importance. Virchows Arch. 1997; 430: 1-16.
- Epstein JI, Carmichael M, Walsh PC: Adenocarcinoma of the prostate invading the seminal vesicle: definition and relation of tumor volume, grade and margins of resection to prognosis. J Urol. 1993; 149: 1040-5.
- Billis A, Freitas LL, Magna LA, Samara AB, Ferreira U: Prostate cancer with bladder neck involvement: pathologic findings with application of a new practical method for tumor extent evaluation and recurrence-free survival after radical prostatectomy. Int Urol Nephrol. 2004; 36: 363-8.
- Billis A, Magna LA, Ferreira U: Correlation between tumor extent in radical prostatectomies and preoperative PSA, histological grade, surgical margins, and extraprostatic extension: application of a new practical method for tumor extent evaluation. Int Braz J Urol. 2003; 29: 113-9; discussion 120.
- Gleason DF, Mellinger GT: Prediction of prognosis for prostatic adenocarcinoma by combined histological grading and clinical staging. J Urol. 1974; 111: 58-64.
- Gleason DF: Histologic grading and clinical staging of prostatic carcinoma. In: Tannenbaum M (ed.), Urologic pathology: The prostate. Philadelphia, Lea & Febiger. 1977; pp. 171-98.
- Cookson MS, Aus G, Burnett AL, Canby-Hagino ED, D’Amico AV, Dmochowski RR, et al.: Variation in the definition of biochemical recurrence in patients treated for localized prostate cancer: the American Urological Association Prostate Guidelines for Localized Prostate Cancer Update Panel report and recommendations for a standard in the reporting of surgical outcomes. J Urol. 2007; 177: 540-5.
- Thorson P, Vollmer RT, Arcangeli C, Keetch DW, Humphrey PA: Minimal carcinoma in prostate needle biopsy specimens: diagnostic features and radical prostatectomy follow-up. Mod Pathol. 1998; 11: 543-51.
- Byar DP, Mostofi FK: Carcinoma of the prostate: prognostic evaluation of certain pathologic features in 208 radical prostatectomies. Examined by the step-section technique. Cancer. 1972; 30: 5-13.
____________________
Accepted
after revision:
March 3, 2010
_______________________
Correspondence
address:
Dr. Athanase Billis
Anatomia Patológica, FCM, Unicamp
Caixa Postal 6111
Campinas, SP, 13084-971, Brazil
E-mail: athanase@fcm.unicamp.br
EDITORIAL COMMENT
The importance of perineural invasion (PNI) in prostate biopsies remains controversial in the literature. The studies published since 1993 have shown an incidence of PNI variable from 11% to 38% significantly related to unfavorable histological features, specially extra-prostatic extension (EPE) and positive surgical margins (1,2). Some authors have shown a relationship between PNI and biochemical recurrence, and because of that, the description of the presence or absence of PNI in prostate biopsies plays a role in surgical pathology reports. The authors studied 264 patients, finding 18.2% of PNI related to EPE, higher Gleason score and seminal vesicles (SV) infiltration only for extensive tumors. However, they did not find any correlation between PNI and tumor progression. The most robust study was published recently by Loeb et al. (3) describing the Johns Hopkins experience with 1256 men submitted to radical prostatectomy, and they found a correlation between PNI with EPE and SV infiltration in multivariate analysis. In addition to the fact that tumor progression occurred in 10.5% of patients with PNI and in only 3.5% of patients without PNI, this aspect was not an independent risk factor for biochemical recurrence. Most importantly, it was not considered to decide nerve-sparing surgery. Since there is some agreement that PNI is related to aggressive pathologic features, we believe it is important that pathologists continue to describe the presence or absence of PNI to give the opportunity to surgeons, oncologists and radiotherapists to take this aspect in account to better plan patients’ treatment.
REFERENCES
- Rubin MA, Bassily N, Sanda M, Montie J, Strawderman MS, Wojno K: Relationship and significance of greatest percentage of tumor and perineural invasion on needle biopsy in prostatic adenocarcinoma. Am J Surg Pathol. 2000; 24: 183-9.
- Bismar TA, Lewis JS Jr, Vollmer RT, Humphrey PA: Multiple measures of carcinoma extent versus perineural invasion in prostate needle biopsy tissue in prediction of pathologic stage in a screening population. Am J Surg Pathol. 2003; 27: 432-40.
- Loeb S, Epstein JI, Humphreys EB, Walsh PC: Does perineural invasion on prostate biopsy predict adverse prostatectomy outcomes? BJU Int. 2009 Aug 19. [Epub ahead of print].
Dr.
Katia Ramos Moreira Leite
Laboratory of Medical Investigation
Divisio of Urology, School of Medicine
University of Sao Paulo, USP
São Paulo, SP, Brazil
E-mail: katiaramos@uol.com.br
EDITORIAL COMMENT
The
authors are to be commended on this thorough analysis of how transrectal
prostate needle biopsy specimens correlate with whole mount prostatectomy
specimens. This study is solid based on the large number of patients (264).
There is further strength in that a single surgeon performed each radical
retropubic prostatectomy, while all of the pathology was interpreted by
a single pathologist. Finally, the statistical methods employed were appropriate
for this type of analysis.
In the literature, there is great controversy
regarding the significance of perineural invasion (PNI). There are as
many reports to suggest significance of PNI as there are reports that
demonstrate no difference in patient outcomes. Specifically, can PNI predict
preoperatively the presence of extracapsular extension (ECE) at the time
of radical prostatectomy? Naturally, if this can be shown that a patient
is at risk preoperatively for ECE, the surgeon may electively alter the
technique to sacrifice part or all of the neurovascular bundle on the
ipsilateral side of the disease. Additionally, this information may be
useful in counseling patients pre-treatment regarding the pros and cons
of various treatment options.
This report by Billis et al., does in fact
demonstrate a subset of patients that have a significant risk of having
a higher grade Gleason score, ECE and seminal vesicle involvement at the
time of radical prostatectomy. Specifically, it was the group of patients
in that more than 13.6% of linear (mm) malignant involvement was identified
in the needle biopsy specimens. 25.8% of this cohort possessed PNI. Fortunately,
regarding overall freedom from biochemical progression, there was no significant
difference between those with and without PNI. Without question, better
pretreatment patient selection will ultimately improve treatment outcomes.
It is not uncommon to assume a patient has low risk criteria when in fact,
they may harbor more advanced disease. In the future, clinicians will
utilize more sophisticated biopsy techniques and apply known pathologic
risk factors to decrease the risk of understaging prostate cancer.
Dr.
Brian J. Moran
Medical Director, Radiation Oncology
Chicago Prostate Center
Westmont, Illinois, USA
E-mail: seeds@prostateimplant.com