CHROMOSOMAL
AND MOLECULAR ABNORMALITIES IN A GROUP OF BRAZILIAN INFERTILE MEN WITH
SEVERE OLIGOZOOSPERMIA OR NON-OBSTRUCTIVE AZOOSPERMIA ATTENDING AN INFERTILITY
SERVICE
(
Download pdf )
FERNANDA A. MAFRA, DENISE M. CHRISTOFOLINI, BIANCA BIANCO, MARCELLO M. GAVA, SIDNEY GLINA, SINTIA I. N. BELANGERO, CAIO P. BARBOSA
Division of Pathological Gynecology and Human Reproduction (FAM, DMC, BB, MMG, SINB, CPB), Department of Gynecology and Obstetrics and Division of Urology (MMG, SG), Department of Surgery, ABC School of Medicine, Santo Andre, Sao Paulo, Brazil
Clinical Urology
Vol. 37 (2): 244-251,
March - April, 2011
doi: 10.1590/S1677-55382011000200011
ABSTRACT
Purpose:
To determine the frequency of genetic alterations in a population of Brazilian
infertile men with severe oligozoospermia or non-obstructive azoospermia.
Materials and Methods: Retrospective study
of a group of 143 infertile men with severe oligozoospermia or non-obstructive
azoospermia from the Andrology Outpatient Clinic of the Human Reproduction
Service at the ABC School of Medicine. Of these patients, 100 had severe
oligozoospermia, and 43 non-obstructive azoospermia. All patients underwent
a genetic study which included karyotype analysis and Y-microdeletion
investigation.
Results: Genetic abnormalities were found
in 18.8% of the studied patients. Chromosomal abnormalities were found
in 6.2% of the patients, being more prevalent in the azoospermia group
(11.6%) than in the oligozoospermia group (4%). Chromosomal variants were
found in 8.3%, and Y-chromosome microdeletions in 4.2% of patients.
Conclusion: The high frequency of genetic
alterations (18.8%) in our series justified performing a genetic investigation
in a population with idiopathic infertility, as results may help determine
the prognosis, as well as the choice of an assisted reproduction technique.
Moreover, a genetic investigation could minimize the risk of transmitting
genetic abnormalities to future generations such as genetic male infertility,
mental retardation, genital ambiguity and/or birth defects.
Key
words: male infertility; chromosome abnormalities; Y chromosome;
microdeletions; oligozoospermia; azoospermia
Int Braz J Urol. 2011; 37: 244-51
INTRODUCTION
Infertility
is a very common health problem, affecting approximately 15-20% of couples
who attempt pregnancy. In almost 50% of infertile couples, the problem
is related to the male. About 15% of infertile men may carry a genetic
abnormality, including chromosomal aberrations and single-gene mutations
(1,2).
The frequency of chromosomal aberrations
in subfertile males is estimated to be 2-3%, and in infertile patients
with sperm counts below 10 x 106 spermatozoa/mL may reach 5-7%; among
patients with azoospermia, the percentage of individuals with cytogenetic
abnormalities increases by 10-15% (1,2).
Among the genetic abnormalities found in
infertile men, those involving chromosome anomalies amount to about 8%,
the most frequent one being the 47,XXY karyotype that characterizes the
Klinefelter Syndrome (1). In comparison, it is worth mentioning that,
in the general population, Robertsonian and reciprocal translocations
are present in 1/1000 and 0.9/1000 newborns, respectively, therefore the
incidence in the infertile male population is nine and seven times higher,
respectively (1).
Microdeletions of the Y chromosome are also
important causes of male infertility, and there are studies showing that
deletions of azoospermia factor (AZF) loci on the long arm of the Y chromosome
are associated with either reduced sperm count (oligozoospermia) or complete
absence of spermatozoa (azoospermia). Microdeletions in these regions
are a cause of severe testiculopathy and are present in 2% of infertile
men (3). In selected males affected by severe oligozoospermia or non-obstructive
azoospermia, the frequency of Y microdeletions can be increased to as
much as 15-20% (4).
The aim of this study was to determine the
frequency of chromosomal abnormalities and Y-chromosome microdeletions
in a group of Brazilian infertile men attending an infertility service.
MATERIALS AND METHODS
This
study made a retrospective assessment of the data of 143 infertile men
(age range 25 to 52 years, mean: 36.6 ± 5.6 years) with non-obstructive
sperm disorders, seen between June 2006 and September 2008 at the Andrology
Outpatient Clinic of the ABC School of Medicine, Santo André/SP,
Brazil. All patients included presented one or more years of primary infertility
and had at least two semen analyses showing either sperm counts under
5 million/mL or azoospermia, with no suspicion of an obstructive cause
(medical history, physical examination and complementary tests). All participants
gave informed consent, according to the protocol approved by the local
Ethics Committee (FMABC No. 237/2008).
Routine clinical and laboratory tests were
performed on all patients, including semen analysis, karyotype analysis
and investigation of Y chromosome microdeletions. Semen analysis was performed
according to the guidelines of the World Health Organization (5).
Karyotype analysis was performed using G-banding,
and at least 20 peripheral blood metaphases were analyzed for each patient.
The number of analyzed metaphases was increased to 100 whenever necessary.
The karyotype results were described according to the International System
for Human Cytogenetic Nomenclature (6).
Fluorescent in situ hybridization (FISH)
with X- and Y-chromosome centromeric probes and a sex-determining region
Y (SRY) probe (Cytocell®, Cambridge, UK) was performed according to
the protocol of Pinkel et al. (1988) (7). One hundred nuclei were analyzed
with the ISIS Software (MetaSystems GmbH, Altlussheim, Germany) and the
results were described according to the International System for Human
Cytogenetic Nomenclature (6).
For the molecular study, genomic DNA was
extracted from lymphocytes, using the Illustra™ Blood GenomicPrep
Mini Spin Kit (GE Healthcare Life Sciences, Buckinghamshire, UK), according
to the manufacturer’s instructions. Multiplex PCR was performed
for SRY (Yp), DBY (AZFa), RBMY (AZFb), DAZ (AZFc), AMELX (X-chromosome
control) and AMELY (Y-chromosome control), according to Umeno et al. (2006)
(8), with temperature modifications. A gene was considered absent only
after three amplification failures, in the presence of amplification of
the internal control (AMELX) and positive control.
RESULTS
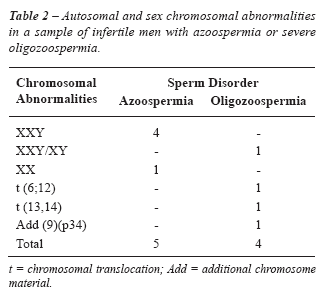
Genetic abnormalities were found in 18.8% of all infertile men studied, as shown in Table-1. The frequency of chromosomal abnormalities was 6.2% (9/143), and is shown in Table-2. Considering only the patients with severe oligozoospermia, the frequency of chromosomal abnormalities was 4.0% (4/100), while in those with azoospermia it was 11.6% (5/43). Chromosomal variants or polymorphisms were found in 8.3% of all cases (Table-3).
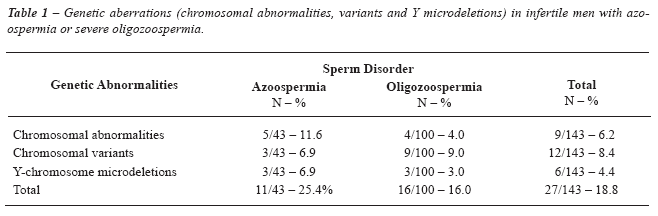

Y-chromosome microdeletions were found in 4.2% (6/143) of all the infertile men studied. In a separate analysis, we found 3.0% (3/100) in the oligozoospermia group and 6.9% (3/43) in the azoospermia group. Of these six patients, three carried microdeletions of the AZFc region and presented severe oligozoospermia, two carried microdeletions of the AZFb+c regions and presented non-obstructive azoospermia, and one had a deletion of AZFa and presented non-obstructive azoospermia.
COMMENTS
Genetic
causes, such as chromosomal aberrations, single-gene mutations and Y-chromosome
microdeletions, are found in a large proportion of infertile couples:
they are present in about 15% of infertile men and 10% of infertile women
(1).
In a previous study, Pina-Neto et al. (2006)
(9) found that 9.6% (16/165) of the patients had karyotype abnormalities.
In agreement with that study, we found a 6.2% (9/143) frequency of chromosomal
abnormalities, and their prevalence was higher in the patients with azoospermia
(11.6%) than in those with oligozoospermia (4.0%). In the group of azoospermic
patients, sex chromosome abnormalities such as 47,XXY, 46,XX/47,XXY and
46,XX were predominant. We found 1% of patients with Klinefelter syndrome
among those with severe oligozoospermia, and 9% among those with azoospermia.
The 46,XX sex differentiation disorder in
males represents the most common condition in which testicular development
occurs in the absence of a Y chromosome. This disorder occurs at a frequency
of 1/25.000 newborns (1). Phenotypically the adults are similar to patients
with Klinefelter syndrome, with normal male external genitalia, microrchidia
and sterility. Molecular analyses have shown that in approximately two
thirds of these cases there are Y-chromosome sequences present in the
genome, including the SRY gene (10). In our study population, there was
one patient who presented a 46,XX karyotype. FISH using a SRY probe confirmed
the presence of the SRY gene on the short arm of one X chromosome. This
is a common finding, resulting from an abnormal crossing-over of pseudoautosomal
regions of the X and Y chromosomes. This abnormal crossing-over can result
in a 46,XX male (SRY+) and/or a 46,XY female (SRY-).
On the other hand, in the group of oligozoospermic
patients, structural chromosomal abnormalities, such as balanced translocations,
Robertsonian translocations and additional chromosomal material, were
more common, as shown in Table-2. Balanced forms of reciprocal translocations
usually do not have any phenotypic effect on the carriers, except that
they may show variable sperm counts, ranging from normal to low (oligozoospermia)
or even total absence of spermatozoa (azoospermia) in the ejaculate (11).
However, the offspring of reciprocal translocation carriers, such as the
patient with a translocation between chromosomes 6 and 12 (12), may display
intellectual disabilities and/or birth defects, due to a partial monosomy
or trisomy. In addition, Robertsonian translocations, such as the rather
common translocation 14/21, can result in offspring with Down syndrome
or in gestational loss of a conceptus with monosomy of chromosome 14 or
21, or trisomy of chromosome 14, which are not compatible with life.
Assisted reproductive techniques have given
the chance of having a child to infertile males with azoospermia and oligozoospermia.
However, using the intracytoplasmic sperm injection (ICSI) in this group
with high genetic abnormality ratio may increase the inheritance of paternal
genetic disorders to offspring (13). It occurs because the presence of
the structural chromosomal abnormality predisposes to abnormal segregation
in meiosis. Alternate segregation in male gametes has been reported and
may result in in vitro fertilization IVF/ICSI failure (14). This may lead
to fertilization failure or poor embryonic development after fertilization.
Significant heterogeneity was observed in the rates of unbalanced gametes,
varying from 2.7% to 26.5% according to the translocations (15). In these
cases, sperm retrieval for IVF/ICSI procedures should be indicated with
restrictions, and the couple should be offered preimplantation diagnosis
whenever possible or sperm donation.
Polymorphic variants, particularly involving
the heterochromatic region of chromosomes 1, 9, 16, Y and the nucleolar
organizing region of acrocentric chromosomes, are known to occur in 2.7%
of the general population (16). However, much higher frequencies (12.2
- 38%) have been reported in infertile individuals (17,18). In the present
study, the overall occurrence of chromosomal variants was 8.3% and included
enlarged heterochromatin on the long arm of chromosome 9 (9qh+), pericentromeric
inversion of chromosome 9 (9ph), and enlarged satellites of acrocentric
chromosomes (14ps+, 15ps+, 21ps+, and 22ps+). Inversions (such as 9ph)
can disturb spermatogenesis and lead to the production of unbalanced gametes
through the formation of an inversion loop, even if the inverted region
includes only heterochromatin (19). Enlarged heterochromatin (9qh+) could
make synapse difficult and, as a consequence, may delay or prevent it.
One patient who presented oligoasthenozoospermia had both the 9ph and
the 9qh+ variants. This case was reported previously (20), and we believe
that the morphological difference between the homologous chromosomes 9
could have led to an error in crossing-over and, as a consequence, produced
aberrant gametes. Chromosomal variations associated with male infertility,
including structural or numerical chromosomal abnormalities and quantitative
or positional modifications of the constitutive heterochromatin, have
been shown to affect male gamete formation and function possibly due to
the silencing effect of these heterochromatic variations on otherwise
normally expressed genes (21). We can, however, not rule out the possibility
that the concomitant finding of azoospermia and chromosomal polymorphic
variants in two patients (46,XY,14ps+ and 46,XY,21ps+, Table-3) was coincidental.
Deletions in the AZFa region usually lead
to Sertoli-cell-only syndrome (SCOS). The AZFb region is involved in the
regulation of meiosis (22). Complete deletions of AZFb or AZFb+c lead
to azoospermia associated with SCOS or pre-meiotic spermatogenic arrest.
AZFc deletions lead to azoospermia or severe oligozoospermia, associated
with different spermatogenic phenotypes in the testis (1). In the present
study, 4.2% (6/143) of the infertile patients had Y-chromosome microdeletions,
while in previous studies on infertile men, Hassum-Filho et al. (2005)
(23) found 3.45% (1/29) of Y-chromosome microdeletions, and Pieri et al.
(2006) (24) found 8.8% (10/114) of microdeletions. In the present study,
the protocol used to investigate Y-chromosome microdeletions includes
one gene of each AZF region and can detect the three major deletions on
the Y-chromosome related to idiopathic spermatogenic failure. In the study
of Hassum-Filho et al. (2005) (23), 18 regions were investigated, while
Pieri et al. (2006) (24) started their analysis with 19 regions and then
decided to investigate the six most frequent ones. The number of investigated
genes may not influence the results of the deletion and prognosis, but
may allow a better characterization of the extent of the deletion. In
the literature, the frequency of Y-chromosome microdeletions ranges from
0 to 60%, according to patient selection and Y-chromosome regions studied
(25-27).
The phenotypes associated with microdeletions
in the different AZF regions are variable (26,28). As there is no precise
association between genotype and testicular histology, no well established
standard treatment or prognostic criterion exists to date for the different
chromosome anomalies found in infertile men. Testicular sperm extraction
and ICSI for the treatment of male-factor infertility have permitted the
use of sperm from oligozoospermic patients with AZFb and AZFc microdeletions,
to achieve successful fertilization and pregnancies (29). For AZFa microdeletions
the prognosis is poor, as in these cases there are no germ cells in the
testis (30,31).
It has to be considered, however, that testicular
sperm retrieval procedures performed on men with severe male infertility
of genetic origin might bear a potential risk of transmitting genetic
abnormalities to the offspring (7,28,32). If a man with an AZFc microdeletion
takes part in an ICSI procedure, he can pass the same deletion, or a larger
one, on to his male offspring, since deletions can generate chromosomal
instability and lead to deletions of the nearby regions, in further generations
(33). In view of the genetic risk for the subsequent generations, the
importance of a careful evaluation of karyotypes and AZF microdeletions
in men presenting idiopathic infertility with non-obstructive azoospermia
or severe oligozoospermia, prior to assisted reproduction by ICSI, is
evident.
In conclusion, our results support the existence
of a relationship between genetic abnormalities such as chromosomal aberrations/variants
and Y-chromosome microdeletions and non-obstructive azoospermia and severe
oligozoospermia. The high frequency of genetic alterations (18.8%) found
in the men affected by idiopathic infertility with severe oligozoospermia
and non-obstructive azoospermia strongly suggests that such patients should
be karyotyped and receive counseling before they are referred for assisted
reproduction techniques. Moreover, these investigations can minimize the
risk of transmitting genetic abnormalities to future generations, such
as genetic male infertility, intellectual disability, genital ambiguity
and/or birth defects.
ACKNOWLEDGEMENT
Supported by Center for Study of Genetics and Human Reproduction, ABC Medical School.
CONFLICT OF INTEREST
None declared.
REFERENCES
- Ferlin A, Arredi B, Foresta C: Genetic causes of male infertility. Reprod Toxicol. 2006; 22: 133-41.
- Shefi S, Turek PJ: Definition and current evaluation of subfertile men. Int Braz J Urol. 2006; 32: 385-97.
- Krausz C, Degl’Innocenti S: Y chromosome and male infertility: update, 2006. Front Biosci. 2006; 11: 3049-61.
- Foresta C, Ferlin A, Gianaroli L, Dallapiccola B: Guidelines for the appropriate use of genetic tests in infertile couples. Eur J Hum Genet. 2002; 10: 303-12.
- World Health Organization: WHO laboratory manual for the examination of human semen and semen-cervical mucus interaction. Cambridge: Cambridge University Press, 1999. 4th ed.
- Shaffer LG, Tommerup N: ISCN 2005 – An international system for human cytogenetic nomenclature. Basel: S. Karger; 2005; pp 37-84.
- Pinkel D, Landegent J, Collins C, Fuscoe J, Segraves R, Lucas J, et al.: Fluorescence in situ hybridization with human chromosome-specific libraries: detection of trisomy 21 and translocations of chromosome 4. Proc Natl Acad Sci U S A. 1988; 85: 9138-42.
- Umeno M, Shinka T, Sato Y, Yang XJ, Baba Y, Iwamoto T, et al.: A rapid and simple system of detecting deletions on the Y chromosome related with male infertility using multiplex PCR. J Med Invest. 2006; 53: 147-52.
- Pina-Neto JM, Carrara RC, Bisinella R, Mazzucatto LF, Martins MD, Sartoratto E, et al.: Somatic cytogenetic and azoospermia factor gene microdeletion studies in infertile men. Braz J Med Biol Res. 2006; 39: 555-61.
- Rajender S, Rajani V, Gupta NJ, Chakravarty B, Singh L, Thangaraj K: SRY-negative 46,XX male with normal genitals, complete masculinization and infertility. Mol Hum Reprod. 2006; 12: 341-6.
- Van Assche E, Bonduelle M, Tournaye H, Joris H, Verheyen G, Devroey P, et al.: Cytogenetics of infertile men. Hum Reprod. 1996; 11 (Suppl 4): 1-24; discussion 25-6.
- Bianco B, Christofolini D, Gava M, Mafra F, Moraes E, Barbosa C. Severe oligospermia associated with a unique balanced reciprocal translocation t(6;12)(q23;q24.3): male infertility related to t(6;12). Andrologia. 2011;43:145-8.
- Kosar PA, Ozçelik N, Kosar A: Cytogenetic abnormalities detected in patients with non-obstructive azoospermia and severe oligozoospermia. J Assist Reprod Genet. 2010; 27: 17-21.
- Scriven PN, Flinter FA, Braude PR, Ogilvie CM: Robertsonian translocations--reproductive risks and indications for preimplantation genetic diagnosis. Hum Reprod. 2001; 16: 2267-73.
- Moradkhani K, Puechberty J, Bhatt S, Lespinasse J, Vago P, Lefort G, et al.: Rare Robertsonian translocations and meiotic behaviour: sperm FISH analysis of t(13;15) and t(14;15) translocations: a case report. Hum Reprod. 2006; 21: 3193-8.
- Yakin K, Balaban B, Urman B: Is there a possible correlation between chromosomal variants and spermatogenesis? Int J Urol. 2005; 12: 984-9.
- Lissitsina J, Mikelsaar R, Punab M: Cytogenetic analyses in infertile men. Arch Androl. 2006; 52: 91-5.
- Madon PF, Athalye AS, Parikh FR: Polymorphic variants on chromosomes probably play a significant role in infertility. Reprod Biomed Online. 2005; 11: 726-32.
- Morel F, Laudier B, Guérif F, Couet ML, Royère D, Roux C, et al.: Meiotic segregation analysis in spermatozoa of pericentric inversion carriers using fluorescence in-situ hybridization. Hum Reprod. 2007; 22: 136-41.
- Belangero SI, Christofolini DM, Bianco B, Gava MM, Wroclawski ER, Barbosa CP: Male infertility related to an aberrant karyotype, 46,XY,9ph,9qh+. Fertil Steril. 2009; 91: 2732-3.
- Minocherhomji S, Athalye AS, Madon PF, Kulkarni D, Uttamchandani SA, Parikh FR: A case-control study identifying chromosomal polymorphic variations as forms of epigenetic alterations associated with the infertility phenotype. Fertil Steril. 2009; 92: 88-95.
- Ferlin A, Moro E, Rossi A, Dallapiccola B, Foresta C: The human Y chromosome’s azoospermia factor b (AZFb) region: sequence, structure, and deletion analysis in infertile men. J Med Genet. 2003; 40: 18-24.
- Hassun Filho PA, Cedenho AP, Lima SB, Ortiz V, Srougi M: Single nucleotide polymorphisms of the heat shock protein 90 gene in varicocele-associated infertility. Int Braz J Urol. 2005; 31: 236-42.
- Pieri Pde C, Pereira DH, Glina S, Hallak J, McElreavey K, Moreira-Filho CA: A cost-effective screening test for detecting AZF microdeletions on the human Y chromosome. Genet Test. 2002; 6: 185-94.
- Simoni M, Bakker E, Krausz C: EAA/EMQN best practice guidelines for molecular diagnosis of y-chromosomal microdeletions. State of the art 2004. Int J Androl. 2004; 27: 240-9.
- Foresta C, Moro E, Ferlin A: Y chromosome microdeletions and alterations of spermatogenesis. Endocr Rev. 2001; 22: 226-39.
- Simoni M, Tüttelmann F, Gromoll J, Nieschlag E: Clinical consequences of microdeletions of the Y chromosome: the extended Münster experience. Reprod Biomed Online. 2008; 16: 289-303.
- Huang WJ, Yen PH: Genetics of spermatogenic failure. Sex Dev. 2008; 2: 251-9.
- Gambera L, Governini L, De Leo V, Luddi A, Morgante G, Tallis V, Piomboni P. Successful multiple pregnancy achieved after transfer of frozen embryos obtained via intracytoplasmic sperm injection with testicular sperm from an AZFc-deleted man. Fertil Steril. 2010; 94: 2330.e1-3.
- Krausz C, Quintana-Murci L, McElreavey K: Prognostic value of Y deletion analysis: what is the clinical prognostic value of Y chromosome microdeletion analysis? Hum Reprod. 2000; 15: 1431-4.
- Foresta C, Moro E, Ferlin A: Y chromosome microdeletions and alterations of spermatogenesis. Endocr Rev. 2001; 22: 226-39.
- Cinar C, Yazici C, Ergünsu S, Beyazyürek C, Javadova D, Saglam Y, et al.: Genetic diagnosis in infertile men with numerical and constitutional sperm abnormalities. Genet Test. 2008; 12: 195-202.
- Lee SH,
Ahn SY, Lee KW, Kwack K, Jun HS, Cha KY: Intracytoplasmic sperm injection
may lead to vertical transmission, expansion, and de novo occurrence
of Y-chromosome microdeletions in male fetuses. Fertil Steril. 2006;
85: 1512-5.
____________________
Accepted
after revision:
September 2, 2010
_______________________
Correspondence
address:
Dr. Bianca Bianco
Faculdade de Medicina do ABC
Av. Príncipe de Gales, 821
Prédio CEPES / 2º andar
Santo André, SP, 09060-650, Brazil
Fax: + 55 11 4438-7299
E-mail: bianca.bianco@hotmail.com
EDITORIAL COMMENT
The
authors should be congratulated on reporting the incidence of genetic-related
infertility in a subset of Brazilian infertile patients presenting with
severe oligozoospermia or non-obstructive azoospermia. Clinically available
molecular testing may reveal microdeletions in the long arm of the Y chromosome.
This novel molecular diagnosis application reclassified about 10% of the
infertile male population previously misdiagnosed as idiopathic oligozoospermia
or non-obstructive azoospermia. The authors highlighted the importance
of screening men with non-obstructive azoospermia and severe oligozoospermia
for Y-chromosome infertility seeking infertility treatment, and reported
that about 5% of their patient population harbored Yq microdeletions.
Originally, 3 AZF regions were defined: AZFa, AZFb, and AZFc (azoospermia
factors a, b, and c), which map on the long arm (Yq) in order from the
centromere to the telomere, but AZFb and AZFc overlaps (1). A fourth region,
named AZFd, located between AZFb and AZFc was also reported. The relative
frequency of individual microdeletions are 60%, 5% and 16% for AZFc, AZFa
and AZFb regions, but combined deletions occur in about 15% of the cases
(2). Because the deletions tend to occur between large palindromic repeats,
Repping et al. (2002) proposed a more appropriate nomenclature using the
name of the flanking repeats for the types of recurrent deletions (1).
Not all men presenting with AZF microdeletions and azoospermia are sterile.
Sperm can be found in the testis with 70% of the cases in partial and
complete AZFc deletion azoospermic patients. In contrast, the chance of
finding sperm in azoospermic men with complete AZFa or AZFb deletions
is unlikely (3). It seems that the presence of a deletion of the Yq chromosome
has no apparent negative effect on fertilization or pregnancy rates and
it does not increase the risk for birth defects in children conceived
via assisted reproduction (3). Y chromosome infertility is inherited in
a Y-linked manner. Because males with deletion of the AZF regions of the
long arm of the Y chromosome are infertile, the deletions are usually
de novo and therefore not present in the father of the proband. However,
the male offspring have the same deletion as their father, with a high
risk of male infertility. The authors have highlighted the importance
of adequate counseling for this group of patients, and added to the discussion
since deletions can generate chromosomal instability and lead to deletions
of the nearby regions.
It was also interesting to note that about
7% of their oligozoospermic population presented with chromosomal abnormalities,
which can be easily detected by karyotyping. Due to the low cost, availability
and clinical implications of genetic testing for azoospermic and severe
oligozoospermic males, it now clear that such screening should be offered
to males who meet these criteria and are seeking infertility treatment.
REFERENCES
- Repping S, Skaletsky H, Lange J, Silber S, Van Der Veen F, Oates RD, et al.: Recombination between palindromes P5 and P1 on the human Y chromosome causes massive deletions and spermatogenic failure. Am J Hum Genet. 2002; 71: 906-22.
- Shefi S, Turek PJ: Definition and current evaluation of subfertile men. Int Braz J Urol. 2006; 32: 385-97.
- Hopps CV, Mielnik A, Goldstein M, Palermo GD, Rosenwaks Z, Schlegel PN: Detection of sperm in men with Y chromosome microdeletions of the AZFa, AZFb and AZFc regions. Hum Reprod. 2003; 18: 1660-5.
Dr. Sandro
Esteves
ANDROFERT, Center for Male Reproduction
Campinas, Sao Paulo, Brazil
Fax: + 55 19 3295-8877
E-mail: s.esteves@androfert.com.br