MALNUTRITION
AS AN ADDITIONAL RISK FACTOR TO GENTAMICIN NEPHROTOXICITY
(
Download pdf )
G. NASCIMENTO GOMES, H. SATO, A.Y. AIHARA, M.F. CAVANAL, F. ZALADEK GIL
Section of Renal Physiology and Thermometabolism, Paulista School of Medicine, Federal University of São Paulo (UNIFESP), São Paulo, SP, Brazil
ABSTRACT
The association between malnutrition and infectious diseases has long been described. Gentamicin is an aminoglycoside antibiotic that is widely used for the treatment of severe gram-negative infections, despite its nephrotoxicity. Clinical and experimental studies have also shown important alterations in renal function during malnutrition. In this way, the aim of the present study was to verify if the use of gentamicin (G) in rats subjected to food restriction (R) could interfere with the development of gentamicin nephrotoxicity. Two month-old Male Wistar rats were submitted to food restriction (50%) during 30 days. In the last 10 days, they were treated with saline or gentamicin (40 mg/kg/day ip). The studied groups were: C)- rats with ad libitum food intake + saline, G)- ad libitum food intake + gentamicin, R)- food-restricted + saline, RG)- food-restricted + gentamicin. A significant fall in glomerular filtration rate (GFR) was observed in groups R (5.69 ± 0.22) and G (5.31 ± 0.27) when compared to group C (7.17 ± 0.42 ml.min-1.kg-1). In RG group, the impairment in GFR was more evident than in groups G or R, (4.42 ± 0.24 ml.min-1.kg-1). In all the experimental groups, the decrease in GFR occurred in parallel to the decrease in renal plasma flow (RPF) so that the filtration fraction (FF%) was maintained. A decline in urine/plasma inulin ratio was observed in both groups treated with gentamicin and also in food restricted group. Although G and R “per se” caused an increased excretion of Na+, the highest values were obtained in RG group. Although our study has been performed in an experimental model, if extrapolated to human protocols, it can be suggested that the use of aminoglycoside antibiotics in malnourished patients should be performed with caution, specially concerning renal function and considering the potential increased risk exhibited by these patients.
Key words:
kidney; gentamicin, nephrotoxicity; rats; malnutrition; renal function
Braz J Urol, 28: 265-270, 2002
INTRODUCTION
In
undeveloped countries, malnutrition (MN) is still the most important public-health
problem which underscores the high rates of mobility and mortality. The
prevalence of MN, according to the World Health Organization (WHO), is
more than 30% of the world’s infant population (1). MN is usually
the reflex of synergistic factors such as low family income, low educational
levels and poor environment and housing conditions, which facilitate the
contact with infectious agents. In fact, the association between MN and
infectious diseases has long been described in the history (2). Impairment
in both humoral and cellular immunity present in cases of malnutrition
can aggravate the prognostic in face of infectious episodes.
Gentamicin is an aminoglycoside antibiotic
that has been widely used in the treatment of infections caused by gram-negative
bacteria, due to its efficacy and low cost (3). The antibacterial activity
is mediated by an irreversible inhibition of the bacteria protein synthesis
at 30S ribosomal level (4). Gentamicin is excreted by glomerular filtration
and is partially reabsorbed by renal proximal tubules. The tubules also
accumulate the antibiotic and are the primary site of nephrotoxicity which
is an important limiting factor in its clinical use (5,6). Several clinical
and experimental studies have been performed concerning drug dose, patient’s
age, and administration schedule, in order to investigate factors that
can aggravate or attenuate aminoglycoside nephrotoxicity. Among these
factors, different therapeutic regimens are being increasingly explored
because of the recent interest in the once-a-day schedule, whose current
evidence suggests to be as safe and effective as multiple daily doses
(7-9).
Few studies have investigated the possible
influence of nutritional status on the development of aminoglicosyde nephrotoxicity.
Since aminoglicosyde therapy is frequently indicated to malnourished patients,
this study was designed to investigate the influence of nutritional status
on the development of gentamicin nephrotoxicity.
MATERIALS AND METHODS
Male
Wistar rats weighing around 200 g were obtained from Paulista School of
Medicine, São Paulo, Brazil. They were housed individually in plastic
cages with sawdust on the floor and maintained inside a temperature controlled
room (25°C ± 0.5) with dark-light cycle of 12:12 hours (lights
on at 8:00 am). Water was provided ad libitum. The animals received care
in accordance with the guidelines of the institutional review board and
animal use committees.
The animals were divided into 4 groups.
During 30 days, 2 groups received ad libitum food intake, while the other
2 groups received only 50% of the intake recorded from the ad libitum
groups. During the last 10 days (from the 21st until 30th) of the experiment,
gentamicin (40 mg/kg) or saline was injected intraperitoneally in the
animals once a day. The following groups were, thus, studied: C)- Control,
with ad libitum food intake, that received saline, G)- ad libitum food
intake that received gentamicin, R)- Food-restricted rats, that received
saline, RG)- Food-restricted that received gentamicin.
On the day 31st, animals were anesthetized
with sodium thiopental 100 mg/kg (ip) and prepared for clearance measurements.
In brief, the jugular vein, carotid artery and urinary bladder were cannulated
for infusions and blood or urine withdrawal, respectively. Tracheostomy
was also performed.
Clearance measurements - The glomerular
filtration rate (GFR) was evaluated by the inulin clearance (10) and the
renal plasma flow (RPF) by the sodium p-aminohippurate (PAH) clearance
(11). Animals were primed with 1 ml of saline containing inulin (90 mg)
and sodium p-aminohippurate (PAH; 2 mg) and then submitted to a continuous
infusion of a solution containing inulin (15 mg/ml) and PAH (4 mg/ml)
at a rate of 0.05 ml/min. After a stabilization period of 30 min, 4 clearance
periods of 30 min were obtained. Blood samples were drawn at the midpoint
of each clearance period. Urine was collected quantitatively in pre-weighed
containers, and urine volume was determined gravimetrically. Inulin and
PAH concentration in plasma and urine were measured colorimetrically (10,11).
The inulin and PAH clearances (Cl) were calculated using the following
formula: Cl = U/P.V, where U and P are the concentration of inulin or
PAH in urine and plasma samples respectively, and V is the urinary flow.
U/P inulin ratio is also used as an index of urinary concentration capacity.
Filtration fraction (FF%) was calculated using the formula: GFR/RPF.100.
Blood and urinary Na+ and K+ were measured
using an Na+ / K+ analyzer, model 248 (Ciba-Corning Diagnostics Ltd.,
Essex, England). Fractional excretion (FE%) of sodium, potassium or bicarbonate
were calculated using the formula: EA/FA.100, where: EA is the excreted
amount and FA is the filtrated amount. Urine and blood pH, pCO2 and bicarbonate
values were obtained with a Ciba-Corning, model 614 blood gas analyzer.
Data are means + standard error (SE); n
is the number of measurements. Differences between experimental groups
were evaluated by analysis of variance followed by the Tuckey test or
Kruskal Wallis test when appropriate. For all analysis the null hypothesis
rejection level was set at 0.05.
RESULTS
At
the end of the experimental period, the animals subjected to food restriction
presented, as expected, a lower body weight (214 ± 3.19 g) than
that observed in animals with ad libitum food intake (296 ± 5.62
g), p < 0.05. All the groups presented values of acid-base status in
the normal range.
The parameters of renal homodynamic are
shown in Table-1. In R and G groups, a similar fall in GFR, RPF and U/P
inulin ratio were observed, when compared to those from control values.
When both, food restriction and gentamicin treatment were present as in
RG, GFR and U/P decreased even more, suggesting that food restriction
can worsen the nephrotoxicity of gentamicin.

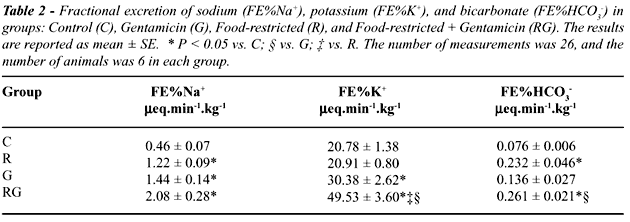
Values for fractional excretion (FE%) of
sodium, potassium and bicarbonate are shown in Table-2. Food restriction
and gentamicin treatment caused a similar increase in FE%Na (1.22 ±
0.09 for R and 1.44 ± 0.14 for G, vs. 0.46 ± 0.07 in C,
p < 0.05). Gentamicin treatment led to a significant increase in FE%K,
when compared to values from C group, 30.38 ± 2.62 vs. 20.78 ±
1.38% (p < 0.05), respectively. When both conditions were present as
in RG group, the increase in FE%K was more accentuated, even compared
to G, (49.53 ± 3.60 vs. 30.38 ± 2.62%, p < 0.05). Although
gentamicin treatment tended to increase the FE%HCO3 in control rats, food
restriction led to a significant impairment in tubular reabsorption of
bicarbonate as shown by the increased values for this parameter in both
R and RG groups when compared to C or G (p < 0.05).
DISCUSSION
Nephrotoxicity
is an important factor which can limit the use of aminoglycosides. Among
them, gentamicin, has been widely used, due to its efficacy and low cost
(3). In a recent study by Caksen et al., (12) it was shown that most part
of urinary tract infections were caused by Escherichia coli, whose strains
were resistant to co-trimoxazone (82.3%), ceftriaxone (17.6%), cefotaxime
(17.6%), and ciprofloxacin (17.6%); none of the studied strains were resistant
to gentamicin, reinforcing the importance of this kind of antibiotic in
the treatment of gram-negative infections.
In the present work, it was observed that
gentamicin affected, more intensely, the glomerular function, the urinary
concentration ability and also the fractional excretion of potassium in
the group submitted to food restriction than the observed in the ad libitum
group. Previous work from this laboratory showed that rats treated with
several types of aminoglycosides exhibited a significant decrease in GFR,
RPF and in sodium and potassium tubular reabsorption, (5,6). On the other
hand, clinical and experimental studies of MN have shown changes in renal
functional parameters such as impairment in renal homodynamic, concentration
ability, and acid excretion (13,14). Klahr et al. (14) studied the effects
of malnutrition on renal concentrating ability and suggested that a diminished
medullary urea concentration could reduce the effective tubular to interstitium
gradient, leading to impairment in the concentration capacity. Another
possibility could be the interference with the Na+/K+ ATPase caused by
malnutrition (15). Studies in children with protein-caloric malnutrition
have consistently demonstrated reductions in GFR and effective renal plasma
flow (ERPF), which were reverted after protein repletion (16,17). Micropuncture
studies performed in protein deprived rats have shown that glomerular
capillary ultrafiltration coefficient (Kf) was reduced in rats fed low
protein diet; moreover, isolated glomeruli from low-protein-fed rats showed
a smaller planar surface area than those from pair-fed isocaloric normal
protein diets. Increased production of angiotensin II was held accountable
for the decrease in glomerular filtration surface area, the fall in Kf,
and the reduction in snGFR and whole kidney GFR (18). Changes in Kf, tubular
Na+/K+ ATPase and antidiuretic hormone (ADH) action were also described
during gentamicin nephrotoxicity. (19,20). Our data show that the use
of gentamicin impaired the reabsorption of Na+, which could result in
extracellular volume depletion and consequent stimulus to renin-angiotensin
system. In food-restricted rats, a model that has already alterations
in extracellular volume homeostasis, gentamicin treatment could impose
an additional factor which could aggravate both glomerular and tubular
function.
Bicarbonate excretion was observed to be
increased in the groups submitted to food restriction, R and RG. Bicarbonate
reabsorption in the proximal tubule is mediated mainly by Na+/H+ antiporter
localized in the brush border membrane (21). Studies by Harris et al.
(22) have shown a decrease in the Na+/H+ antiporter in renal brush border
membrane vesicles from animals kept on a low protein diet. It is possible
that this mechanism is responsible for the bicarbonate wasting observed
in R and in RG. A tendency to elevate the bicarbonate excretion, although
not significant, was observed in G; previous work from this laboratory
showed that during metabolic acidosis, aminoglycoside treatment caused
impairment in proximal bicarbonate reabsorption. However, the capacity
of distal bicarbonate reabsorption was maintained (5,6). Our present data
suggest that restricted animals are able to keep an adequate acid-base
equilibrium even in the presence of gentamicin treatment, suggesting that
the intact distal tubular function can compensate the impairment in proximal
Na+/H+ antiporter.
Although our study has been performed in
an experimental model, if extrapolated to human protocols, the present
results deserve some comments. The use of aminoglycoside antibiotics in
malnourished patients is not unusual, since this condition is frequently
seen in severely ill patients. In this way, the possibility of an enhancement
of nephrotoxic effects must be taken into account, considering the potential
increased risk that may be present in these circumstances.
REFERENCES
- Onis M, Monteiro C, Akre J, Clugston G: The worldwide magnitude of protein-energy malnutrition: an overview from the WHO global database on child growth. Bull World Health Organ, 71: 703-712, 1993.
- Davidson S, Passmore R, Brock JF, Truswell, AS: Nutrition and Infection. In: Human Nutrition and Dietetics. Edinburg, Churchill Livingstone, 7th Ed, Chapt 59, p. 505, 1979.
- Hitt CM, Klepser ME, Nightingale CH, Quintiliani R, Nicolau DP: Pharmacoeconomic impact of once-daily aminoglycoside administration, Pharmacother, 17: 810-814, 1997.
- Kadurugamuwa JL, Clarke AJ, Beveridge TJ: Surface action of gentamicin on Pseudomonas aeruginosa. J Bacteriol, 175: 5798-5805, 1993.
- Costa Silva VL, Gil FZ, Nascimento G, Cavanal MF: Evaluation of distal tubular function in aminoglycoside-induced nephropaty. Braz J Med Biol Res, 20: 833-836, 1987.
- Silva VL, Gil FZ, Nascimento G, Cavanal MF: Effect of gentamicin on urinary acidification in the rat. Ren Physiol, 9: 204-212, 1986.
- Gilbert DN, Lee BL, Dworkin RJ, Leggett JL, Chambers HF, Modin G et al.: A randomized comparison of the safety and efficacy of once-daily gentamicin or thrice-daily gentamicin in combination with ticarcillin-clavulanate. Am J Med, 105: 182-191, 1998.
- Beauchamp D, Collin P, Grenier L, LeBrun M, Couture M, Thibault L et al.: Effects of fasting on temporal variations in nephrotoxicity of gentamicin in rats. Antimicrob Agents Chemother, 40: 670-676, 1996.
- Uijtendaal EV, Rademaker CM, Schobben AF, Fleer A, Kramer WL, van Vught AJ: Once-daily versus multiple-daily gentamicin in infants and children. Ther Drug Monit, 23: 506-513, 2001.
- Flores-Mendoza I, Mello Aires M, Malnic G: Effect of furosemide on urinary acidification during alterations of acid-base equilibrium in the rat. J Bras Nefrol, 1: 51-58, 1979.
- Smith HW, Finkelstein N, Aliminosa L, Gralford B, Graber M: The renal clearances of substituted hippuric acid derivatives and other aromatic acids in dog and man. J Clin Invest, 24: 388, 1945.
- Caksen H, Cesur Y, Uner A, Arslan S, Sar S, Celebi V et al.: Urinary tract infection and antibiotic susceptibility. Int Urol Nephrol, 32: 245-247, 2000.
- Benabe JE, Martinez Maldonado M: The impact of malnutrition on kidney function. Miner Electrolyte Metab, 24, 20-26, 1998.
- Klarhr S, Tripathy K, Garcia FT, Mayoral LG, Ghitis J, Bolanos O: On the nature of the renal concentration defect in malnutrition. Am J Med, 43: 84-96, 1967.
- Kudo LH, Shimizu MH, Seguro AC, Rocha AS: Renal concentrating defect in protein malnutrition: The role of thick ascending limb of Henle and inner medullary collecting duct. Nephron, 57: 156-163, 1991.
- Gordillo G, Soto RA, Medcoff J, Lopez E, Antillon LG: Intracellular composition and homeostatic mechanisms in severe chronic infantile malnutrition. III Renal adjustments. Pediatrics, 20: 303-316, 1957.
- Arroyave G, Wilson D, Behar M, Scrimshaw NS: Serum and urinary creatinine in children with severe protein malnutrition. Am J Clin Nutr, 9: 176-179, 1956.
- Ichikawa I, Purkerson ML, Klahr S, Troy JL, Martinez-Maldonado M, Brenner BM: Mechanism of reduced glomerular filtration rate in chronic malnutrition. J Clin Invest, 65: 982-988, 1980.
- Humes HD, Weinberg JM: The effect of gentamicin on antidiuretic hormone – stimulated osmotic water flow in the toad urinary bladder. J Lab Clin Med, 101: 472-478, 1983.
- Fukuda Y, Malmborg AS, Aperia A: Gentamicin inhibition of Na+/K+ ATPase in rat kidney cells. Acta Physiol Scand, 141: 27-34, 1991.
- Preisig PA, Ives HE, Cragoe EJ Jr, Alpern RJ, Rector FC Jr: Role of the Na+/H+ antiporter in rat proximal tubule bicarbonate absorption. J Clin Invest, 80: 970-978, 1987.
- Harris RC, Seifter JL, Brenner BM: Adaptation of Na+/H+ exchange in renal microvillus membrane vesicles: Role of dietary protein and uninephrectomy. J Clin Invest, 74: 1979-1987, 1984.
_________________________
Received: December 12, 2001
Accepted after revision: March 19, 2002
_______________________
Correspondence address:
Dr. Frida Zaladek Gil
Disciplina de Fisiologia Renal e Termometabologia
Universidade Federal de São Paulo
Rua Botucatu, 862, 5o. andar
São Paulo, SP, 04023-900, Brazil
E-mail: frida@ecb.epm.br