IMPACT
OF INFECTION ON THE SECRETORY CAPACITY OF THE MALE ACCESSORY GLANDS
(
Download pdf )
M. MARCONI, A. PILATZ, F. WAGENLEHNER, T. DIEMER, W. WEIDNER
Department of Urology and Pediatric Urology, University of Giessen, Giessen, Germany
ABSTRACT
Introduction:
Studies that compare the impact of different infectious entities of the
male reproductive tract (MRT) on the male accessory gland function are
controversial.
Materials and Methods: Semen analyses of
71 patients with proven infections of the MRT were compared with the results
of 40 healthy non-infected volunteers. Patients were divided into 3 groups
according to their diagnosis: chronic prostatitis NIH type II (n = 38),
chronic epididymitis (n = 12), and chronic urethritis (n = 21).
Results: The bacteriological analysis revealed
9 different types of microorganisms, considered to be the etiological
agents, isolated in different secretions, including: urine, expressed
prostatic secretions, semen and urethral smears: E. Coli (n = 20), Klebsiella
(n = 2), Proteus spp. (n = 1), Enterococcus (n = 20), Staphylococcus spp.
(n = 1), M. tuberculosis (n = 2), N. gonorrhea (n = 8), Chlamydia tr.
(n = 16) and, Ureaplasma urealyticum (n = 1). The infection group had
significantly (p < 0.05) lower: semen volume, alpha-glucosidase, fructose,
and zinc in seminal plasma and, higher pH than the control group. None
of these parameters was sufficiently accurate in the ROC analysis to discriminate
between infected and non-infected men.
Conclusion: Proven bacterial infections
of the MRT impact negatively on all the accessory gland function parameters
evaluated in semen, suggesting impairment of the secretory capacity of
the epididymis, seminal vesicles and prostate. These findings were associated
with an infectious related significant increase of semen pH. None of the
semen parameters evaluated can be suggested as a diagnostic tool for infection.
Key
words: accessory sex organs, male; infection; semen; alpha-glucosidases;
fructose; zinc
Int Braz J Urol. 2009; 35: 299-309
INTRODUCTION
Infection
of the male reproductive tract (MRT) is a common disease that can deteriorate
the quality of spermatozoa and impair the function of the male accessory
glands; for this reason it is considered one of the potentially correctable
causes of male infertility (1,2). However, the physiopathology and epidemiology
regarding the impact of infection on the male accessory gland function
is still a matter of debate, and the power of different exocrine function
markers of the male accessory glands to discriminate between infected/inflammated
vs. non-infected/inflammated patients have been reported with controversial
results (3-6).
Male
accessory glands secrete several factors such as alpha-glucosidase, fructose,
prostaglandins, bicarbonate and citric acid amongst others, which are
crucial for sperm physiology. Inflammation “per se” (7) and
secondary obstruction (8) have been proposed as possible mechanisms through
which different infectious agents may impair their function. Under normal
conditions the epididymis secretory factors are involved in the maturation
of sperm; its function can be evaluated by the measurement of L-carnitine,
glycerylphosphoryl choline and alpha-glucosidase in seminal plasma. The
secretion of alpha-glucosidase is used to reliably evaluate epididymal
function; however, there is no consensus regarding the impact of chronic
epididymitis on the level of this marker (7,9,10). The seminal vesicles
produce: fructose, ascorbic acid, ergothioneine, prostaglandins and bicarbonate.
These factors act as reducing agents and in preventing sperm agglutination
(11). A deleterious effect of infection on the secretory function of the
seminal vesicles, evaluated by fructose levels has been previously reported
(4); however, these findings were not confirmed by other authors (10,12).
The secretory function of the prostate gland has been widely investigated:
seminal plasma pH, citric acid, gamma-glutamyl transpeptidase and zinc
have been proposed as markers of its exocrine function, their concentrations
are usually altered in response to bacterial infection and inflammation
(1). However, they are currently not recommended as diagnostic tools to
detect inflammation or infection in the MRT (5).
Since
studies that evaluate the impact of infection on the male accessory gland
function still remain controversial, we decided to evaluate the secretory
function of the epididymis, seminal vesicles and the prostate, using alpha-glucosidase,
fructose and zinc as parameters, in patients with chronic epididymitis,
chronic bacterial prostatitis (CBP) and, chronic urethritis.
MATERIALS AND METHODS
Seventy-one
symptomatic consecutive patients (age ranges 23-62) with proven chronic
infections of the MRT, attending our special outpatient Department for
Urological Andrology were recruited as a study group and 40 age-matched
healthy volunteers (age range 20-62), with no previous medical history
or evidence of andrological or urological disease and with sterile urine
and semen, were enrolled as a control group. The inclusion criteria for
the patients enrolled in the study were genital pain or discomfort secondary
to infection of the MRT lasting for more than 6 months, no antibiotic
uptake for at least six weeks prior the first bacteriological evaluation
and, a positive bacteriological finding in the Meares-Stamey 4-glass test.
Patients with severe chronic systemic illnesses (i.e. HIV, chemotherapy),
previous chronic non-infectious genitourinary diseases under treatment
(i.e. benign prostatic hyperplasia under treatment with alpha-blockers
or 5 alpha-reductase inhibitors) and, history of prostate biopsy, were
excluded from the study. Patients were included in the study irrespective
of their fertility status and classified into three groups according to
our diagnostic schedule (Table-1).
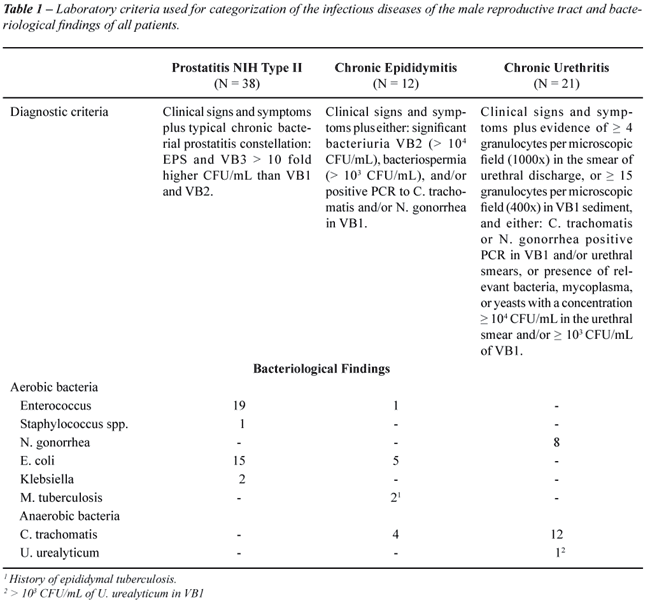
The
diagnosis of CBP was made clinically and based on microbiological tests
following the consensus criteria of the NIH (13,14). The diagnosis of
chronic epididymitis was performed clinically, sonographically and microbiologically
according to consensus statements including search for sexually transmitted
disease microorganisms and inflammatory parameters in the ejaculate (peroxidase
positive leukocytes, elastase) (15,16). The diagnosis of chronic urethritis
was done clinically and microbiologically including the search for sexually
transmitted microorganisms and leukocyte counts of the first voided urine
and in the ejaculate (peroxidase positive leukocytes, elastase) (16) (Table-1).
Categorization of CBP, Chronic Epididymitis and Chronic Urethritis
Evaluation
of patients with CBP included (17): the NIH chronic prostatitis symptom
index German version (18), physical examination including digital rectal
examination of the prostate, transrectal ultrasound (TRUS), the 4-glass
test with search for common urinary bacteria, mycoplasma and yeasts in
all urine fractions (first voided urine: VB1, midstream urine: VB2, and
post-prostatic massage urine: VB3) and expressed prostatic secretions
(EPS), ejaculate analysis (19), polymerase chain reaction (PCR) for Chlamydia
(C.) trachomatis in VB1 (Abbott, Wiesbaden, Germany), and microscopic
examination of VB3 for detection of Trichomonas vaginalis (20).
In
men with chronic epididymitis, the evaluation included scrotal ultrasound
and duplex according to consensus suggestions for evaluation of patients
with epididymitis (15). The microbiological evaluation included PCR in
VB1 for C. trachomatis and Neisseria (N.) gonorrhea, and search for common
relevant bacteria in VB2 and in the ejaculate (16) (Table-1).
In
patients with chronic urethritis, microbiological evaluation included
microscopic evaluation of VB1 with cytological analysis for leukocytes
(Papanicolaou-staining), semi quantitative culture methods for relevant
bacteria (N. gonorrhea, mycoplasmas and Candida spp.) in urethral discharge
and in VB1, and PCR for C. trachomatis and N. gonorrhea (Abbott, Wiesbaden,
Germany) in urethral smears and in VB1. Evidence of ≥ 4 granulocytes
per microscopic field (X1000) in the urethral discharge smear, or ≥
15 granulocytes per microscopic field (X400) in VB1 sediment, and either:
C. trachomatis or N. gonorrhea positive PCR in VB1 and/or urethral smears,
or presence of common bacteria, mycoplasmas, or yeasts with a concentration
≥ 104 CFU/mL in the urethral discharge and/or ≥
103 CFU/mL in VB1, were considered criteria for diagnosis (16)
(Table-1).
Detection of Microorganisms
The bacteriological analysis of the patients revealed 9 different types of infectious agents isolated in different MRT secretions including urine, EPS, urethral smears and semen (Table-1). The most common isolated microorganisms in all patients were Escherichia (E.) coli (n = 20) and Enterococcus spp. (n = 20). When the different diseases were analyzed separately, Enterococcus spp. was the most common agent isolated in patients with CBP and, in patients with chronic epididymitis, E. coli was the most prevalent. Infections due to sexually transmitted microorganisms were only detected in men suffering from chronic epididymitis and chronic urethritis. Two patients had a previous history of epididymitis due to Mycobacterium (M.) tuberculosis previously treated with antibiotics.
Ejaculate Analysis
Complete
ejaculate analysis according to the WHO standards (19) including semen
volume, pH, elastase and peroxidase positive leukocytes (PPL) determination
was performed in all men (21). Levels of a-glucosidase and fructose (total
enzymatic activity) at neutral pH were determined by spectrophotometrical
methods described elsewhere (5). Zinc was assessed using a commercially
available kit (Zinc Kit, Bako, Germany).
The
impact of inflammation on the levels of semen volume, pH, α-glucosidase,
fructose and zinc was analyzed stratifying the patients as having an inflammatory
or non-inflammatory spermiogram according to two well accepted criteria:
PPL ≥ 1x106/mL (19) and/or elastase ≥ 230 ng/mL
(21).
Statistical Analysis
Data were analyzed by the Prisma program for Windows version 5.0. Mann-Whitney U, Kruskal-Wallis and Dunn’s multiple comparison test were used to analyze the results of the ejaculate and, Receiver Operating Characteristic Curves (ROCC-analysis) was applied to assess the normal ranges of the seminal plasma parameters in the cases where statistical difference was found. Statistical significance was achieved at p < 0.05, all reported p values are two-sided.
RESULTS
Cytomorphological
Analysis of the Ejaculate
Compared
with the controls, the patients had statistically significantly (p <
0.05) lower sperm concentration, % of sperm with progressive motility
(a+b), % of sperm with normal morphology; and higher: % of immotile sperm,
% of sperm with head deformity, and % of sperm with tail deformity. No
significant differences were observed in the levels of PPL, elastase and
sperm vitality between the two groups (Table-2).

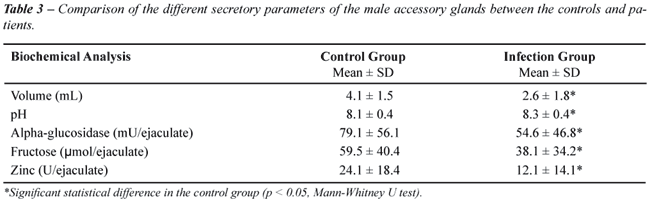
Secretory Parameters of the Male Accessory Glands
Compared
to the controls, the patients had statistically significantly (p <
0.05) lower (mean ± SD): semen volume (4.1 ± 1.5 vs. 2.6
± 1.8 mL), levels of a-glucosidase (79.1 ± 56.1 vs. 54.6
± 46.8 mU/ejaculate), levels of fructose (59.5 ± 40.4 vs.
38.1 ± 34.2 µmol/ejaculate), levels of zinc (24.1 ±
18.4 vs. 12.1 ± 14.1 U/ejaculate) and, higher pH (8.1 ±
0.4 vs. 8.3 ± 0.4) (Table-3).
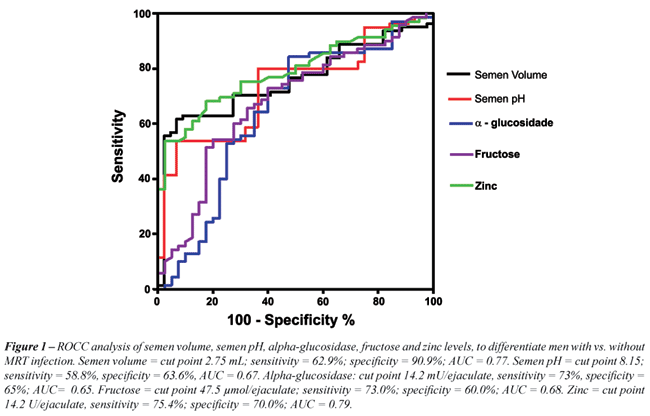
We
applied a ROCC analysis to quantify a cut point to discriminate men with
versus without infection using the parameters of male accessory gland
function that were significantly different in the infection group (n =
71). A semen volume of 2.75 mL was found to discriminate patients from
controls with a sensitivity of 62.9% and specificity of 90.9%, the Area
Under the Curve (AUC) was 0.77, a level a-glucosidase of 56.1 mU/ejaculate
discriminated men with versus without infection with a sensitivity of
73% and specificity of 60% (AUC: 0.65), a level of fructose of 47.5 µmol/ejaculate
discriminated men with versus without infection with a sensitivity of
73% and specificity of 60% (AUC: 0.68) and, a level of zinc of 14.2 U/ejaculate
discriminated men with versus without infection with a sensitivity of
75.4% and specificity of 70% (AUC: 0.79). A semen pH of 8.15 was found
to discriminate patients from controls with a sensitivity of 58.8% and
specificity of 63.6% (AUC: 0.67) (Figure-1).
The
multiple comparison analysis between the different diagnoses, microorganisms
and control group, revealed that patients with CBP had statistically significant
(p < 0.05) lower: semen volume (4.1 ± 1.5 vs. 2.5 ± 1.7
mL), levels of fructose (59.5 ± 40.4 vs. 37.2 ± 32.9 µmol/ejaculate),
and levels of zinc (24.1 ± 18.4 vs. 10.9 ± 13.7 U/ejaculate)
than the controls. Patients with chronic urethritis had statistically
significant (p < 0.05) lower: semen volume (4.1 ± 1.5 vs. 2.1
± 1.1 mL) and levels of zinc (24.1 ± 18.4 vs. 13.7 ±
15.1 U/ejaculate) than the controls. Patients with chronic epididymitis
had statistically significant (p < 0.05) lower levels of zinc (24.1
± 18.4 vs. 13.1 ± 14.2 U/ejaculate) than the controls. Patients
infected with E. coli had statistically significant (p < 0.05) lower
levels of fructose (59.5 ± 40.4 vs. 23.9 ± 18.4 µmol/ejaculate),
lower levels of zinc (24.6 ± 18.4 vs. 6.3 ± 7.2 U/ejaculate),
reduced semen volume (4.1 ± 1.5 vs. 2.2 ± 1.4 mL), and higher
pH (8.1 ± 0.4 vs. 8.5 ± 0.4) compared to the control group.
Patients infected with N. gonorrhea presented statistically significant
(p < 0.05) lower semen volume (4.1 ± 1.5 vs. 1.8 ± 1.2
mL) and lower levels of zinc (24.6 ± 18.4 vs. 8.0 ± 8.5
U/ejaculate) than the control group. No other significant differences
between the patients and the controls were observed considering all the
other specific diagnoses and microorganisms evaluated.
The
subgroup of patients with inflammatory signs in the spermiogram according
to two criteria: PPL ≥ 1x106/mL (19) and/or elastase
≥ 230 ng/mL (21) (n = 36), demonstrated also a significant decrease
in the levels of semen volume, alpha-glucosidase, fructose and zinc compared
to the controls and, higher values of semen pH (data not shown). The sensitivity
and specificity of these parameters to diagnose MRT infection in patients
with inflammatory signs in the ejaculate were; semen volume: cut point
2.75 mL, sensitivity 63.2%, specificity 90.9%, AUC 0.79; semen pH: cut
point 8.15, sensitivity 61.1%, specificity 63.6%, AUC 0.70; alpha-glucosidase
cut point 56.1 mU/ejaculate, sensitivity of 73% specificity of 60%, AUC:
0.65; fructose: cut point: 47.5 µmol/ejaculate, sensitivity of 73%,
specificity of 60%, AUC: 0.68; zinc: cut point 10.7 U/ejaculate, sensitivity
83%, specificity 80%, AUC 0.88.
COMMENTS
In
the clinical setting MRT infection has a questionable effect on the fertility
prognosis. What seems to be clear is that the different factors secreted
by the male accessory glands are crucial for normal conception, since
sperm retrieved directly from the epididymis or the testis are unable
to fertilize the egg without the assistance of artificial reproduction
techniques (22). Taking this fact into account, it is interesting to analyze
the effect of different infectious entities of MRT and their etiologic
agents on the secretory function of the male accessory glands. Mainly
two questions need to be answered, first if the different diseases and
microorganisms can cause obstruction of the seminal pathways and by that
means deteriorate the normal composition of the ejaculate; and second
if they are related to any intrinsic impairment of the secretory function
of the epididymis, seminal vesicles and prostate.
The
fact that all patients included in the study group had proven MRT infectious
diseases defined according to well accepted consensus definitions, allowed
us to accurately identify the origin of the MRT infection. Moreover, in
previous reports semen cultures identify significant bacteriospermia in
only 50% of semen specimens from men with CBP, thus ejaculate culture
is not yet recommended for the first line of diagnostic management in
men with suspected CBP (23). The microbiological findings in our study
group are in agreement with previous reports (1); N. gonorrhea and C.
trachomatis are the most frequent infectious agents isolated patients
with chronic urethritis. In patients with CBP, Enterococcus and E. coli
were, as expected, the more frequent microbiological agents isolated.
Finally, it is interesting that in patients consulting for chronic epididymitis,
the microbiological findings gave similar results to those found in cases
of acute epididymitis (15), suggesting that under special conditions the
epididymis may act as a reservoir of bacteria in the male reproductive
tract.
In
our series of patients, we did not find any azoospermic patients, although,
the levels of alpha-glucosidase, fructose, zinc and sperm concentration
were significantly lower in the infection group compared to the control
group. Also, there were no conclusive findings indicating a total obstruction
of the MRT at any level. This fact confirms that obstruction is not an
important cause of impairment of the male accessory gland function in
an infectious setting and although infection has been previously mentioned
in the literature as common cause of obstruction of the MRT (2), our findings
and more recent studies (6,20) seem to confirm that it is a rare occurrence
in patients with demonstrated MRT infection and inflammation.
Analyzing
the secretory function of the epididymis we found that men with MRT infectious
diseases (n = 71) had significantly lower concentration of a-glucosidase.
However, in the multivariate analysis no significant differences were
found in the levels of this marker between any specific diagnosis and
the controls, even in patients with chronic epididymitis. Moreover, no
specific bacteria had a significant impact on its level. Although Cooper
and co-workers (10) found a significant decrease in the level of alpha-glucosidase
in patients with acute epididymitis; in patients with chronic epididymitis
previous reports suggest that the impact on secretory function of the
epididymis is not significant (9). We propose that our series of patients
reflect that the secretion of alpha-glucosidase is significantly decreased
in cases of chronic infections of the MRT; however, this decrease is not
as pronounced compared to the decrease observed in the levels of others
markers, i.e. zinc.
The
function of the seminal vesicles can be accurately determined by the level
of fructose in seminal plasma, questionable since these glands produce
the majority of the semen volume, this parameter may also be used as indirect
indicator of its function. When discussing the literature, the impact
of infection on the secretory function of the seminal vesicles remains
controversial. Comhaire and co-workers (4), in agreement with our results,
found a negative impact of infection on the fructose level, but concluded
that the measurement of its concentration was not useful for discriminating
between infected and non-infected patients due to its low sensitivity
and specificity. Vicari and co-workers (24) demonstrated that patients
with prostate-vesiculo-epididymitis had significantly lower seminal fructose
levels than those patients with prostatitis alone. On the other hand,
Bezold and co-workers (12) did not find a significant decrease in the
concentration of fructose in seminal plasma in a population of infertile
men with sexually transmitted diseases. The controversial results regarding
the impact of MRT infection on fructose levels may be explained by the
different clinical and microbiological criteria used to include patients
in the studies, we believe that the evaluation of patients with suspected
MRT infection should follow consensus criteria and guidelines e.g. NIH
criteria for patients with prostatitis (13,14) or the European guidelines
for the management of epididymo-orchitis for patients with epididymitis
(15). Using these diagnostic procedures and criteria to manage our patients,
we found a negative impact of infection for the MRT on the fructose level.
In contrast, if we had considered the fact that the seminal vesicles produce
approximately 80% of the ejaculate volume, a decrease in this parameter
would indicate at least in part, that the secretory functions of these
glands were impaired, most probably secondary to an intrinsic damage rather
than to obstruction, since azoospermia was not found in any patient indicating
a normal passage of seminal fluid to the colliculus. The negative effect
of infection and inflammation on semen volume has been previously described
(25,26). Although semen volume was significantly lower in our patients,
the low sensitivity and specificity in the ROCC analysis prevents us from
suggesting it as a diagnostic tool to detect MRT infection.
Secretory
dysfunction of the prostate gland is a common finding in patients with
documented prostatitis. Our previously reported research (5) found, that
the levels of gamma-GT were significantly decreased in patients with inflammatory
chronic prostatitis/chronic pelvic pain syndrome (CP/CPPS); however, it
cannot be recommended as a diagnostic tool for inflammation due to its
low sensitivity and specificity. In this new study group, including only
patients with documented infection, the levels of zinc, identified as
prostatic secretory markers were significantly lower. These findings agree
with previous reports demonstrating lower levels of zinc in the seminal
plasma of patients with MRT infection (27). However, again the power of
this marker to discriminate infected from non-infected patients (sensitivity
43.6%, specificity 75%) was low.
Although,
a zinc-rich diet can slightly increase the concentration of this element
in the prostate gland (28); there is no conclusive evidence that this
therapy can increase the zinc concentration in semen, making the rationale
of this therapy unclear.
Semen
pH is determined by the acid secretions of the prostate and the alkaline
secretions of the seminal vesicles. In vitro, external pH is an important
factor in the regulation of sperm physiology. An acid pH contributes to
maintain a non-capacitated state preventing premature acrosomal reaction
(29). Higher levels of semen pH in patients with MRT infection have been
reported in the literature and could reflect, at least in part, a secretory
dysfunction of the prostate due to lower levels of citric acid in semen
(30). The importance of semen pH in fertility prognosis of these patients
is not clear. However, from a diagnostic point of view semen pH can not
be recommended as a tool to discriminate infected from non-infected patients,
due to its low sensitivity and specificity.
Nevertheless,
all the evaluated parameters of the secretory function of the accessory
glands were significantly altered in patients with concomitant infection
and inflammation of the MRT (n = 36) compared to controls. In the ROCC
analysis the sensitivity and specificity of these factors to detect infection
was not significantly increased, when compared to the whole infection
group analysis (n = 71) and were not significant when inflammatory activity
was considered.
Finally,
a constellation that includes inflammatory signs in the spermiogram (PPL
≥ 1x106/mL and/or elastase ≥ 230 ng/mL), of low
semen volume, elevated semen pH and low levels of alpha-glucosidase, fructose
and zinc could indicate the presence of an infection; however, the sensitivity
and specificity of these parameters, prevents their application as a diagnostic
tool for the detection of MRT infection.
CONCLUSIONS
Infection of the male reproductive tract significantly decreases the levels of semen volume, alpha-glucosidase, fructose and zinc in seminal plasma suggesting impairment of the secretory function of the epididymis, seminal vesicles and prostate. Due to their low sensitivity and specificity these parameters can not be recommended as a diagnostic tool to detect MRT infection. Although semen pH is significantly increased in patients with infection of the male reproductive tract, its sensitivity and specificity to detect infection are low.
ACKNOWLEDGEMENTS
M. Marconi, M.D. is a fellow in “Clinical Andrology” and received a scholarship from MIDEPLAN, Chile.
CONFLICT OF INTEREST
None declared.
REFERENCES
- Weidner W, Krause W, Ludwig M: Relevance of male accessory gland infection for subsequent fertility with special focus on prostatitis. Hum Reprod Update. 1999; 5: 421-32.
- World Health Organization: WHO Manual for the Standardized Investigation, Diagnosis and Management of the Infertile Male. Cambridge University Press. 2000; 2nd Ed.
- Eggert-Kruse W, Zwick EM, Batschulat K, Rohr G, Armbruster FP, Petzoldt D, et al.: Are zinc levels in seminal plasma associated with seminal leukocytes and other determinants of semen quality? Fertil Steril. 2002; 77: 260-9.
- Comhaire FH, Vermeulen L, Pieters O: Study of the accuracy of physical and biochemical markers in semen to detect infectious dysfunction of the accessory sex glands. J Androl. 1989; 10: 50-3.
- Ludwig M, Vidal A, Diemer T, Pabst W, Failing K, Weidner W: Seminal secretory capacity of the male accessory sex glands in chronic pelvic pain syndrome (CPPS)/chronic prostatitis with special focus on the new prostatitis classification. Eur Urol. 2002; 42: 24-8.
- Weidner W, Wagenlehner FM, Marconi M, Pilatz A, Pantke KH, Diemer T: Acute bacterial prostatitis and chronic prostatitis/chronic pelvic pain syndrome: andrological implications. Andrologia. 2008; 40: 105-12.
- Mahmoud AM, Geslevich J, Kint J, Depuydt C, Huysse L, Zalata A, et al.: Seminal plasma alpha-glucosidase activity and male infertility. Hum Reprod. 1998; 13: 591-5.
- Dohle GR: Inflammatory-associated obstructions of the male reproductive tract. Andrologia. 2003; 35: 321-4.
- Wolff H, Bezold G, Zebhauser M, Meurer M: Impact of clinically silent inflammation on male genital tract organs as reflected by biochemical markers in semen. J Androl. 1991; 12: 331-4.
- Cooper TG, Weidner W, Nieschlag E: The influence of inflammation of the human male genital tract on secretion of the seminal markers alpha-glucosidase, glycerophosphocholine, carnitine, fructose and citric acid. Int J Androl. 1990; 13: 329-36.
- Okamura N, Tajima Y, Ishikawa H, Yoshii S, Koiso K, Sugita Y: Lowered levels of bicarbonate in seminal plasma cause the poor sperm motility in human infertile patients. Fertil Steril. 1986; 45: 265-72.
- Bezold G, Politch JA, Kiviat NB, Kuypers JM, Wolff H, Anderson DJ: Prevalence of sexually transmissible pathogens in semen from asymptomatic male infertility patients with and without leukocytospermia. Fertil Steril. 2007; 87: 1087-97.
- Krieger JN, Nyberg L Jr, Nickel JC: NIH consensus definition and classification of prostatitis. JAMA. 1999; 282: 236-7.
- Schaeffer AJ, Anderson RU, Krieger JN, Lobel B, Naber K, Nakagawa M, Nickel JC, Nyberg L, Weidner W: The Assessment And Management Of Male Pelvic Pain Syndrome Including Prostatitis. In: Mc Connel J, Abrams P, Denis L, Khoury S, Roehrsom C (eds.), Male Lower Urinary Tract Dysfunction. Evaluation and Management6th International Consultation in Prostate Cancer and Prostate Diseases. Health Publications, Paris. 2006; pp. 343-385.
- Horner PJ; European Branch of the International Union against Sexually Transmitted Infection and the European Office of the World Health Organization: European guideline for the management of epididymo-orchitis and syndromic management of acute scrotal swelling. Int J STD AIDS. 2001; (12 Suppl 3): 88-93.
- Schiefer HG: Microbiology of male urethroadnexitis: diagnostic procedures and criteria for aetiologic classification. Andrologia. 1998; (30 Suppl 1): 7-13.
- Weidner W, Anderson RU: Evaluation of acute and chronic bacterial prostatitis and diagnostic management of chronic prostatitis/chronic pelvic pain syndrome with special reference to infection/inflammation. Int J Antimicrob Agents. 2008; (31 Suppl 1): S91-5.
- Schneider H, Brähler E, Ludwig M, Hochreiter W, Collins MF, Eremenco S, et al.: Two-year experience with the German-translated version of the NIH-CPSI in patients with CP/CPPS. Urology. 2004; 63: 1027-30.
- World Health Organization Laboratory Manual for the Examination of Human Semen and Semen-Cervical Mucus Interaction. Cambridge University Press, Cambridge. 1999; 4th ed.
- Ludwig M, Vidal A, Diemer T, Pabst W, Failing K, Weidner W: Chronic prostatitis/chronic pelvic pain pyndrome): seminal markers of inflammation. World J Urol. 2003; 21: 82-5.
- Ludwig M, Kümmel C, Schroeder-Printzen I, Ringert RH, Weidner W: Evaluation of seminal plasma parameters in patients with chronic prostatitis or leukocytospermia. Andrologia. 1998; (30 Suppl 1): 41-7.
- Diemer T, Schroeder-Printzen I, Weidner W: Surgical sperm retrieval. Urologe A. 2007; 46: 789-98.
- Weidner W, Wagenlehner FM, Marconi M, Pilatz A, Pantke KH, Diemer T: Acute bacterial prostatitis and chronic prostatitis/chronic pelvic pain syndrome: andrological implications. Andrologia. 2008; 40: 105-12.
- Vicari E, La Vignera S, Castiglione R, Calogero AE: Sperm parameter abnormalities, low seminal fructose and reactive oxygen species overproduction do not discriminate patients with unilateral or bilateral post-infectious inflammatory prostato-vesiculo-epididymitis. J Endocrinol Invest. 2006; 29: 18-25.
- Sanocka-Maciejewska D, Ciupinska M, Kurpisz M: Bacterial infection and semen quality. J Reprod Immunol. 2005; 67: 51-6.
- Engeler DS, Hauri D, John H: Impact of prostatitis NIH IIIB (prostatodynia) on ejaculate parameters. Eur Urol. 2003; 44: 546-8.
- Wang Y, Kang L, Hou Y, Wu X, Chen J, Han X: Microelements in seminal plasma of infertile men infected with Ureaplasma urealyticum. Biol Trace Elem Res. 2005; 105: 11-8.
- Cortesi M, Fridman E, Volkov A, Shilstein SSh, Chechik R, Breskin A, et al.: Clinical assessment of the cancer diagnostic value of prostatic zinc: a comprehensive needle-biopsy study. Prostate. 2008; 68: 994-1006.
- Parrish JJ, Susko-Parrish JL, First NL: Capacitation of bovine sperm by heparin: inhibitory effect of glucose and role of intracellular pH. Biol Reprod. 1989; 41: 683-99.
- Comhaire FH, Vermeulen L, Pieters O: Study of the accuracy of physical and biochemical markers in semen to detect infectious dysfunction of the accessory sex glands. J Androl. 1989; 10: 50-3.
____________________
Accepted after revision:
January 20, 2009
________________________
Correspondence address:
Dr. Wolfgang Weidner
Dept of Urology & Pediatric Urology
Justus-Liebig-Universität Giessen
Rudolf-Buchheim-Strasse 7
Giessen, 35385, Germany
Fax: + 49 0641 99-44509
E-mail: wolfgang.weidner@chiru.med.uni-giessen.de
EDITORIAL COMMENT
Male
accessory gland infection (MAGI) is a syndrome which includes clinical
symptoms of inflammation of the prostate gland, the seminal vesicles,
the ductus deferens and the epididymis. There is not a clear discrimination
against the term prostatis, glandulitis vesicalis or epididymitis. There
is also not a clear classification of MAGI as formulated for prostatitis,
although it is likely that different types of MAGI in terms of infectious
and non-infectious causes also exist.
Usually MAGI is caused by infectious agents spreading
from the urethra via prostate gland, seminal vesicles, ductus deferens,
and epididymis. The frequency of a changeover from urethritis to MAGI
is unknown. Infections by viral agents are to date hypothetical.
MAGI may lead to obstruction of the ductus epididymidis,
impairment of spermatogenesis in epididymo-orchitis, to impairment of
sperm function and to the induction of sperm auto-antibodies, as well
as to dysfunctions of the male accessory glands, which leads to decreased
seminal concentrations of citric acid, phosphatase, fructose concentration,
zinc and alpha-glutamyltransferase.
In general, however, the impact of MAGI on semen
composition and sperm function, possibly relevant for male fertility,
is low and vice versa, none of the dysfunctions can be considered suitable
for the diagnosis of MAGI. The paper clearly shows that the diagnosis
of MAGI on the basis of semen analysis is difficult and that the diagnosis
of prostatitis and epididymitis is more reliably performed by means of
an examination of prostatic fluid and imaging procedures.
Dr.
Walter Krause
Dept. Dermatology & Allergology
University Hospital, Philipp University
Marburg, Germany
E-mail: krause@med.uni-marburg.de