PERTINENT
ISSUES RELATED TO LAPAROSCOPIC RADICAL PROSTATECTOMY
(
Download pdf )
SIDNEY C. ABREU, INDERBIR S. GILL
Section of Laparoscopic and Minimally Invasive Surgery, Glickman Urological Institute, Cleveland Clinic Foundation, Cleveland, Ohio, USA
ABSTRACT
Purpose:
We describe the critical steps of the laparoscopic radical prostatectomy
(LRP) technique and discuss how they impact upon the pertinent issues
regarding prostate cancer surgery: blood loss, potency and continence.
Results: A major advantage of LRP is the
reduced operative blood loss. The precise placement of the dorsal vein
complex stitch associated with the tamponading effect of the CO2 pneumoperitoneum
significantly decrease venous bleeding, which is the main source of blood
loss during radical prostatectomy. At the Cleveland Clinic, the average
blood loss of our first 100 patients was 322.5 ml, resulting in low transfusion
rates.
The continuous venous bleeding narrowed pelvic surgical field and poor
visibility can adversely impact on nerve preservation during open radical
prostatectomy. Laparoscopy, with its enhanced and magnified vision in
a relatively bloodless field allows for excellent identification and handling
of the neurovascular bundles.
During open retropubic radical prostatectomy, the pubic bone may impair
visibility and access to the urethral stump, and the surgeon must tie
the knots relying on tactile sensation alone. Consequently, open prostatectomy
is associated with a prolonged catheterization period of 2 – 3 weeks.
Comparatively, during laparoscopic radical prostatectomy all sutures are
meticulously placed and each is tied under complete visual control, resulting
in a precise mucosa-to-mucosa approximation.
Conclusion: The laparoscopic approach may
represent a reliable less invasive alternative to the conventional open
approach. Despite the encouraging preliminary anatomical and functional
outcomes, prospective randomized comparative trials are required to critically
evaluate the role of laparoscopy for this sophisticated and delicate operation.
Key
words: prostate; carcinoma; prostatic neoplasms; prostatectomy;
laparoscopy
Int Braz J Urol. 2003; 29: 489-496
INTRODUCTION
Once
considered an unpopular operation with significant morbidity, radical
retropubic prostatectomy has evolved into a refined, anatomically precise
operation (1). In view of the already satisfactory oncological and functional
outcomes achieved with the traditional open retropubic approach and encouraged
by the initial inroads of laparoscopy in renal oncology, efforts have
been recently focused towards developing a laparoscopic approach to radical
prostatectomy, aiming to decrease the morbidity related to the open operation.
However, the advantages of laparoscopic renal surgery, wherein avoidance
of a flank incision markedly reduces morbidity, are not so evident for
laparoscopic radical prostatectomy (LRP). In fact, the avoidance of a
low midline incision used for the open retropubic approach likely will
not have the same implications as regards reduction of morbidity (2).
Due to the enhanced visualization and magnification during LRP, and the
tamponading effect of the CO2 pneumoperitoneum, the laparoscopic approach
has the potential to favorably impact upon the functional sequelae related
to this intricate operation. The decreased blood loss, adequate preservation
of the neurovascular bundles and superior watertight urethrovesical anastomosis
may ultimately further improve potency and continence rates (3,4).
In this article, we describe the critical
steps of laparoscopic radical prostatectomy and discuss how they impact
upon the important issues regarding prostate cancer surgery: blood loss,
positive surgical margins, and postoperative potency and continence rates.
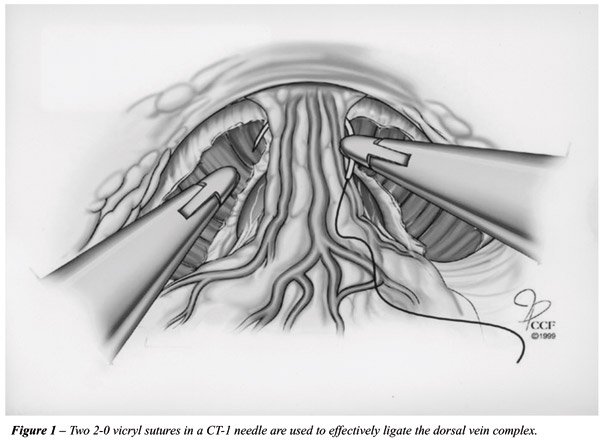
DORSAL VEIN COMPLEX LIGATION AND TRANSECTION - IMPACT OVER BLOOD LOSS
Similar
to the open counterpart, the dorsal vein complex is suture ligated in
order to decrease blood loss upon it transection. After the Foley catheter
is replaced by an 18F metallic urethral sound, a 2 - 0 vicryl stitch with
a CT-1 needle is employed to ligate the dorsal vein. This stitch is placed
in a back-hand manner from the right to the left side, distal to the apex
of the prostate, between the dorsal vein complex and the urethra (Figure-1).
In order to avoid inadvertent transgression of the urethra by the suture,
the assistant pushes down the metallic sound, displacing the urethra posteriorly.
We routinely place two stitches across the dorsal vein complex in an attempt
to achieve a safe ligation. Also, we anchor the dorsal vein stitch to
the pubic periosteum, aiming to achieve a retropubic urethropexy suspension
in order to possibly enhance continence outcomes. A back-bleeding stitch
is placed across the anterior surface of the base of the prostate. Following
bladder neck transection, the prostate base is grasped and tractioned
cephalad, which places the urethra and the dorsal vein complex on stretch.
Subsequently, an electrosurgical J-hook or the harmonic scalpel is used
to slowly divide the dorsal vein complex at the apex of the prostate achieving
meticulous hemostasis. Occasionally the dorsal vein stitch may become
loosened, leading to venous hemorrhage. However due to the tamponading
effect of the CO2 pneumoperitoneum this hemorrhage is usually not intense,
and can be controlled by placing another stitch around the transected
dorsal vein.
High operative blood loss and transfusion
is a common problem of open prostate surgery. A major advantage of LRP
is the reduced operative blood loss. The precise placement of the dorsal
vein complex stitch under laparoscopic visualization associated with the
tamponading effect of the 15 mmHg pressure CO2 pneumoperitoneum significantly
decrease venous bleeding, which is the main source of blood loss during
radical prostatectomy. Conversely, during retropubic open surgery, a right
angle clamp is blindly passed underneath the dorsal vein complex in order
to control it with a tie. This maneuver is mainly based on the palpation
of the Foley catheter to identify the anterior surface of the urethra
and may lead to clamp misplacement and significant bleeding.
In a review of 1228 LRP at six European
centers, average blood loss was 488 ml with a transfusion rate of 3.5%
(5). The French team of Guillonneau et al. also reported a mean intraoperative
blood loss of 354 ml with a transfusion rate of 5.7% (20 patients) in
a series of 350 LRP (6). At the Cleveland Clinic, the average blood loss
of our first 100 patients was 322.5 ml, resulting in low transfusion rates
(2%) (7). This is in contrast to open prostatectomy series with reported
blood losses between 500 ml to 1000 ml.
BLADDER NECK TRANSECTION AND APICAL DISSECTION – IMPACT OVER POSITIVE SURGICAL MARGINS
The
precise anatomic location of the junction between the prostate and the
bladder neck is not well defined under laparoscopic visualization. A combination
of maneuvers can be performed to overcome the absence of precise landmarks
for identifying the bladder neck: Close laparoscopic visualization usually
identifies the area where the prevesical fat ends, signifying the prostatovesical
junction. Gentle blunt palpation with the elbow of the J-hook eletrocautery
also aids in defining this junctional area. Repeated in and out movements
of the metallic urethral dilator, with its curved tip pointing anteriorly,
provide another indication where the prostate ends and where the bladder
begins. At the presumed prostato-vesical junction, a horizontal incision
is created using J-hook eletrocautery (Figure-2). The anterior bladder
neck is divided in the midline, and the tip of the urethral dilator is
delivered through the cystotomy into the space of Retzius. We do not make
a major effort to spare the bladder neck in an attempt to minimize positive
surgical margin at this location. However, a carefully dissected bladder
neck can avoid the extra step of bladder neck reconstruction. Due to the
precise mucosa-to-mucosa laparoscopic urethrovesical anastomosis, bladder
neck evertion is not necessary.
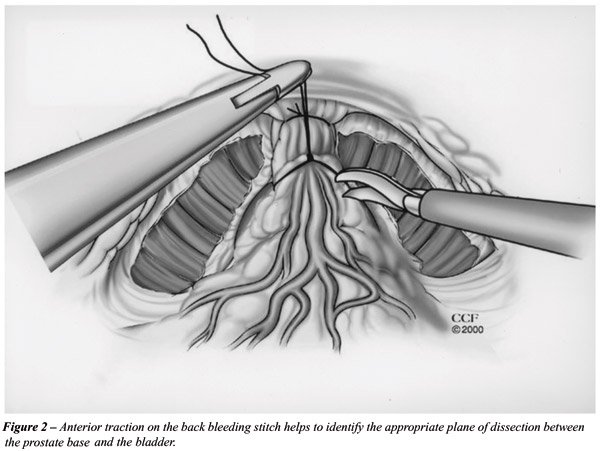
Meticulous apical dissection is probably
the most challenging step of LRP. During this step technical shortcomings
can lead to incontinence (compromised urethral stump length), impotency
(damage to the neurovascular bundle), and positive surgical margins (iatrogenic
entry into the prostate apex) (8). After the dorsal venous complex is
divided, the anterior urethral wall is identified with the aid of the
metallic urethral sound. The neurovascular bundles, located posterolateral
to the urethra, are mobilized laterally from the prostato-urethral junction
using the fine gently curved tip of the harmonic scalpel without activation.
At this point, cold Endoshears is used to transect the anterior urethral
wall close to the concave notch of the prostate, which assures preservation
of an excellent urethral stump. The tip of the intraurethral metallic
sound is delivered through the urethral opening. The posterior urethral
wall and the rectourethralis muscles are divided, completely detaching
the prostate. At this point, care must be taken to avoid inadvertent entry
into the rectum.
Concerns about bladder neck and apical positive
surgical margins after laparoscopic radical prostatectomy have been raised
(9). Laparoscopy is likely to be comparable to open surgery as regards
positive surgical margins. In our experience, a significant decline on
the positive surgical margin rate occurred from our first 50 cases to
our third 50 cases (7). Technique refinements, especially during the apical
dissection, were inherently related to this reduction of our positive
surgical margins rate. In addition to this, we believe that a reasonable
number of positive surgical margins present in our series may be “false-positive”,
related to the laparoscopic manipulation of the prostate with traumatic
instruments. In fact, the laparoscopic Allis forceps used to tautly retract
the prostate cephalad and laterally during neurovascular bundle dissection
has a propensity to routinely remove divots from the gland. In an attempt
to overcome this, we are now using an additional clamp through a 5 mm
port placed at the supra-pubic region to anteriorly retract the seminal
vesicles and vas deferens and maneuver the prostate, thus potentially
avoiding these presumed “false positive” surgical margins.
Although the available data are sparse,
it is likely that LRP will emerge as a sound oncologic alternative. Recently,
Guillonneau et al. reported an oncological mid-term evaluation of 1000
patients that underwent LRP (10). In this study, the incidence of positive
surgical margins for the stages pT2a, pT2b was 6.9%, 18.6%, which is comparable
to the series of Lepor et al. who reported 19.9% positive margin rate
in 1000 open retropubic radical prostatectomies (11). Similarly Katz et
al. reported an 18.9% incidence of positive surgical margins in 169 pT2
cases (12). These authors highlight that a constant decrease was noted
in the overall incidence of positive margins over time, and that avoiding
bladder neck preservation eliminated positive bladder neck margins. Moreover,
the overall incidence of positive lateral surgical margins in pT2 cases
treated with a nerve sparing procedure was 8.4%. Rassweiller et al. reported
a 16% incidence of positive margins in 180 patients, almost half of who
had pT3 disease (13).
NEUROVASCULAR BUNDLE DISSECTION – IMPACT OVER POSTOPERATIVE POTENCY
The
optimal laparoscopic technique of nerve sparing continues to evolve. We
currently employ a combined antegrade-retrograde technique. A few technical
maneuvers in this regard include: 1) Upon opening the endopelvic fascia,
while releasing the levator muscles from the apex and lateral aspect of
the prostate, no thermal or electrical energy is used near the NVB. A
laparoscopic kittner or “cold” cut scissors is used to complete
the dissection; 2) Following dorsal vein ligation, the lateral fascia
over the prostate is bilaterally incised superficially with “cold”
Endoshears in order to release the tethering of the NVB; 3) Aiming to
minimize eletrocautery trauma to the NVB near the tip of the seminal vesicles,
the vesicular artery is secured with hemostatic locking clips (Weck Systems,
Triangle Park, NC).
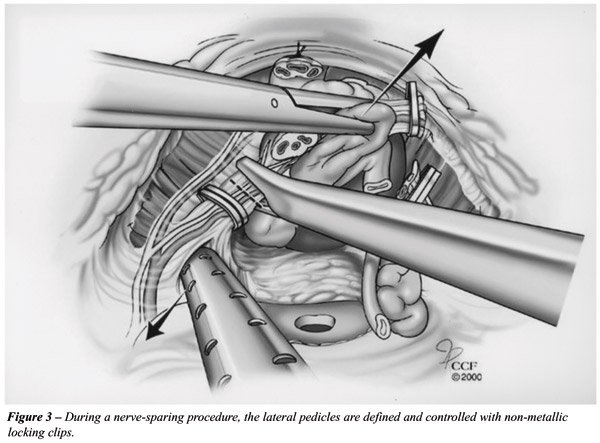
Initially, the lateral pedicles of the prostate
are controlled with one or two 10 mm Hemolock clips (Weck Systems, Triangle
Park, NC) (Figure-3). The postero-lateral edge of the prostate base is
identified. Once this anatomical landmark is encountered, the magnified
laparoscopic vision allows the surgeon to identify the neurovascular bundle.
Staying approximately 2 mm away from the prostate, the harmonic scalpel
is employed to develop a plane between the gland and the NVB. The harmonic
scalpel is preferred because of its limited spread of thermal energy (1
– 2 mm). Furthermore, no electric energy (monopolar or bipolar)
is involved, which could potentially disrupt nerve integrity and conduction.
Anatomic studies have demonstrated the precise
location of the neurovascular bundle, which is familiar to most urologists.
However, the continuous venous bleeding narrowed pelvic surgical field
and poor visibility can adversely impact on nerve preservation during
open radical prostatectomy. Laparoscopy, with its enhanced and magnified
vision in a relatively bloodless field allows for excellent identification
and handling of the neurovascular bundles (14). Therefore, the potential
exits to match the best potency rates obtained with open surgery. Guillonneau
et al. reviewed 73 of their patients who had either bilateral (46 patients)
or unilateral (27 patients) nerve-sparing LRP. A remarkable 74% spontaneous
erection rate was reported in the bilateral nerve sparing group and 51%
in the unilateral group with a follow-up ranging from 2 to 12 months (15).
More recently, Katz et al. followed-up 143 patients that underwent LRP
which were potent preoperatively. Of these patients, 100, 80, 48 and 26
responded to a sexual function questionnaire at 1, 3, 6 and 12 months
after surgery, respectively. Of the unilateral and bilateral nerve-sparing
groups 50% and 87.5% reported spontaneous erections at 12 months after
surgery, respectively (16). These authors found the overall rate of patients
who had erections preoperatively and maintained erections after surgery
(53.8%) to be comparable to the results for open surgery.
URETHRO-VESICAL ANASTOMOSIS – IMPACT OVER POSTOPERATIVE CONTINENCE
During
open retropubic radical prostatectomy, the pubic bone may impair visibility
and access to the urethral stump, and the surgeon must tie 4 to 8 knots
in a blind field, relying on tactile sensation alone. Consequently, open
prostatectomy is associated with a prolonged catheterization period of
2 – 3 weeks. Comparatively, during laparoscopic radical prostatectomy
all sutures are meticulously placed and each is tied under complete visual
control, resulting in a precise mucosa-to-mucosa approximation (17).
We employ a continuous running suture in
an attempt to decrease postoperative urinary leak (Figure-4). A double-armed
stitch is prepared by tying two 2 - 0 sutures on a UR-6 needle, one dyed
Monocryl and the other undyed Caprosyn, each 10 inches in length (18).
Both needles are initially passed outside in at 6 o’clock on the
posterior bladder neck, thus placing the knot outside the bladder and
anchoring the stitch at this position. The first stitch is run up in a
clockwise direction from 6 o’clock to 9 o’clock. The second
stitch is run up in a counter-clockwise direction from 6 o’clock
to 3 o’clock. At this point, both stitches are placed on traction.
Due to the low friction characteristics of the Monocryl and Caprosyn,
the sutures glide smoothly under traction, thus tautly anchoring the entire
posterior half of the bladder neck to the urethral stump. Upon creation
of this posterior plate, a 22F urethral Foley catheter is easily advanced
into the bladder. Anastomosis is completed by running both stitches to
the 12 o’clock where they are tied together. Our preliminary results
with this technique indicate a low rate of urinary leak on postoperative
day 3 cystogram, allowing catheter removal at that time in the majority
of the patients. Moreover, only one intracorporeal knot is tied.
Adequate preservation of a long urethral
stump and an intact urinary sphincter mechanism with the laparoscopic
technique may potentially result in improved postoperative continence
rates. Guillonneau et al. reported on their first 133 patients with at
least a one year follow-up and found 85.5% were totally continent (no
protection needed during day or night). Five patients (3.8%) were classified
as severely incontinent (15). Nadu et al. have reported continence rates
greater then 93% in a median follow-up of 7 months (range from 1 to 15).
In this particular study, the authors reported that only 15.1% of the
patients had anastomotic leak on postoperative day 2 to 4 cystography
(19). Despite using a single circular running stitch technique, no anastomotic
stricture, pelvic abscess or urinoma were noticed in this series. At the
Cleveland Clinic, we have not documented any case of anastomotic stricture
in an overall experience that exceeds 300 cases of LRP. (unpublished data).
CONCLUSION
We believe that radical prostatectomy is the gold standard for the definitive treatment of localized prostate cancer in the appropriate patient. The laparoscopic approach may represent a reliable less invasive alternative to the conventional open approach. Despite the encouraging preliminary anatomical and functional outcomes above discussed, prospective randomized comparative trials are required to critically evaluate the role of laparoscopy for this sophisticated and delicate operation.
REFERENCES
- Walsh PC, Donker PJ: Impotency following radical prostatectomy: insight into etiology and prevention. J Urol. 1982; 128: 492-7.
- Andriole GL: Laparoscopic radical prostatectomy: CON. Urology. 2001; 58: 507-8.
- Steinberg A, Gill I: Laparoscopic radical prostatectomy. Contemp Urol. 2002; 14: 34–49.
- Gill I, Zippe C: Laparoscopic radical prostatectomy: technique. Urol Clin North Am. 2001; 28: 423-36.
- Susler T, Guillenneau B, Vallancien G, Gaston R., Piechaud T, Turk I, et al.: Complications and initial experience with 1228 laparoscopic radical prostatectomy at 6 European centers. J Urol. 2001; 165: (Abst# 605).
- Guillonneau B, Cathelineau X, Doublet JD, Vallancien G: Laparoscopic radical prostatectomy: the lessons learned. J Endourol. 2001; 15: 441-5.
- Farouk A, Gill I, Kaouk J, Matin S, Meraney A, Abreu SC, et al.: 150 laparoscopic radical prostatectomy: learning curve in the United the States. J Endourol. 2002; 16: A33.
- Steinberg AP, Abreu SC, Gill, IS: Laparoscopic Radical Prostatectomy. In: Smith, A. (ed.), Smith’s Textbook of Endourology, New York, Quality Medical Publishing (In Press – 2004).
- Walsh PC: Minimally invasive treatment of prostate cancer. J Endourol. 2001; 15: 447-8.
- Guillonneau B, El Fettouh H, Baumert HC: Laparoscopic radical prostatectomy: Oncological miditerm evaluation about 1000 patients at Montsouris Institute. J Urol. 2002; 169: 1261-6.
- Lepor H, Ferrandino M, Nieder AM: The intraoperative and postoperative complications following radical retropubic prostatectomy in a consecutive series of 1000 cases. J Urol. 2001; 165: 1347A.
- Katz R, Salomon L, Hoznek A, Taille A, Antiphon P, Abbou CC: Surgical margins in laparoscopic radical prostatectomy: the impact of apical dissection, bladder neck remodling and nerve preservation. J Urol. 2003; 169: 2049-52.
- Rassweiler J, Sentker L, Seemann O, Hatzinger M, Rumpelt HJ: Laparoscopic radical prostatectomy with the Heilbronn technique: an analysis of the first 180 cases. J Urol. 2001; 166: 2101-8.
- Kaouk HJ, Desai MM, Abreu SC, Papay F, Gill IS: Robotic assisted laparoscopic sural nerve grafting during radical prostatectomy: initial experience. J Urol. 2003; 170: 909-12.
- Guillonneau B, Cathelineau X, Doublet JD: Prospective assessment of functional results after laparoscopic radical prostatectomy. J Urol. 2001; 165: 614A.
- Katz R, Salomon L, Hoznek A: Patient reported sexual function following laparoscopic radical prostatectomy. J Urol. 2002; 168: 2078-82.
- Hoznek A, Salomon L, Rabii R, Slama MB, Cicco A, Antiphon P, et al.: Vesicourethral anastomosis during laparoscopic radical prostatectomy: the running suture method. J Endourol. 2000; 14: 749-53.
- Von Vethoven RF, Ahlering TE, Peltier A, Skarecky DW, Clayman RV: Technique for laparoscopic running urethrovesical anastomosis: the single knot method. Urology. 2003; 61: 699-702.
- Nadu A, Salomon L, Hoznek A, Olsson LE, Saint F, Taile A, et al.: Early removal of the catheter after laparoscopic radical prostatectomy. J Urol. 2001; 166: 1662-4.
_________________________
Received:
September 9, 2003
Accepted: October 14, 2003
________________________
Correspondence address:
Dr. Sidney C. Abreu
Section of Laparoscopy and Endourology,
São Carlos Hospital
Av. Pontes Vieira, 2551
Fortaleza, CE, 60135-420, Brazil
Fax: + 55 85 227-6393
E-mail: sidneyabreu@hotmail.com