UROTHELIAL-TYPE
ADENOCARCINOMA OF THE PROSTATE MIMICKING METASTATIC COLORECTAL ADENOCARCINOMA
(
Download pdf )
BRIAN P. ADLEY, KELLY MAXWELL, DANIEL P. DALTON, XIMING J. YANG
Departments of Pathology (BPA, XJY) and Urology (KM, DPD, XJY) Northwestern University, Feinberg School of Medicine, Chicago, Illinois, USA
ABSTRACT
Adenocarcinoma arising in urinary bladder or prostatic urethra is uncommon. When they occur, the tumor can be mistaken for metastatic lesions, especially from the colon. Here we report the fifth case of a primary urothelial-type adenocarcinoma arising in the prostate which showed enteric differentiation. The patient was a 55 year-old male whose prostatic needle core biopsy showed a high grade adenocarcinoma which was initially thought to be metastatic colon cancer. A follow-up colonoscopy was unremarkable. Subsequent prostatectomy revealed a high grade adenocarcinoma which was positive for cytokeratins 7 and 20, carcinoembryonic antigen, CDX2, and high molecular weight cytokeratin, and negative for prostate specific antigen, prostate specific acid phosphatase and AMACR. A diagnosis of urothelial-type adenocarcinoma of the prostate was rendered. We review the literature regarding this entity, and discuss the differential diagnosis, emphasizing utility of immunohistochemistry in making the diagnosis. Finally, we speculate on the behavior of these rare tumors.
Key
words: prostate; urothelial-type adenocarcinoma; pathology
Int Braz J Urol. 2006; 32: 681-8
INTRODUCTION
Urothelial-type
adenocarcinomas with mucinous/enteric differentiation arising in the prostatic
gland is exceedingly rare, with only 4 case reports found in the literature.
Tran & Epstein (1) described 2 cases of mucinous adenocarcinoma of
urethral type which occurred in the setting of urethritis glandularis
and associated adenocarcinoma in situ. Curtis et al. (2) described 2 additional
cases in which both were centered around the prostatic urethra but were
not associated with urethritis glandularis. The authors discussed the
utility of immunohistochemistry in distinguishing these tumors from other
histologic mimickers including conventional prostatic adenocarcinoma and
metastatic colorectal adenocarcinoma (2).
Here we report a case of rare urothelial-type
adenocarcinoma with enteric differentiation arising in the prostate. The
initial biopsy was suggestive of metastatic colorectal carcinoma to the
prostate. We describe the clinicopathologic features and discuss the importance
of immunohistochemistry in differentiating these two entities.
CASE REPORT
A 55 year old male presented to the urology clinic at our institution with a serum prostate-specific antigen (PSA) of 10.0 ng/mL and abnormal digital rectal examination. Past medical history included hypertension and gout. The patient denied alcohol intake and quit smoking 10 years prior. The patient subsequently underwent a prostatic needle core biopsy which showed prostatic acinar adenocarcinoma, Gleason’s grade 3+3 in 2 of 4 cores (30% of examined tissue) from the right lobe. In addition, the presence of high grade adenocarcinoma morphologically similar to colonic adenocarcinoma was observed in the left lobe. Based on the pathologic suspicion of metastatic colon cancer, a complete work-up was conducted to locate a primary colon cancer. An MRI of the pelvis showed diffuse involvement of the peripheral zone of left prostate gland (apex to base) with extension across the midline and possible extension to the left seminal vesicle. The patient subsequently had an unremarkable colonoscopy and negative colonic biopsy. A bone scan failed to reveal any metastatic tumor at that time. The patient underwent a retropubic radical prostatectomy with pelvic lymph node dissection for the prostatic acinar adenocarcinoma.
PATHOLOGIC FINDINGS
Gross
The radical prostatectomy specimen weighed
92 grams and measured 8.0 x 7.0 x 4.5 cm. The right seminal vesicle measured
2.5 x 2.0 x 1.4 cm, and the left seminal vesicle measured 1.0 x 1.0 x
0.7 cm. The external surface of the prostate was tan with a shaggy appearance.
Serially sectioning from apex to base revealed a large, tan-white, firm
and focally necrotic, infiltrative mass lesion measuring 3.5 x 3.0 x 2.0
cm, replacing almost the entire left lobe. This mass grossly extended
into the capsule and the left seminal vesicle. The remaining cut surface
of the prostate had a tan nodular appearance without additional discernable
tumors.
Microscopic
Hematoxylin and eosin (HE) stained sections
from both the left needle core biopsy and prostatectomy specimens showed
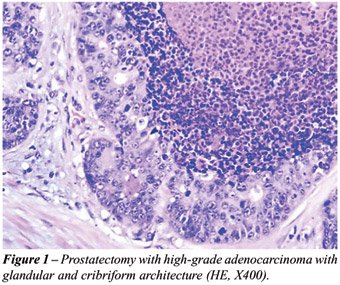
a high grade adenocarcinoma with glandular and cribriform architecture
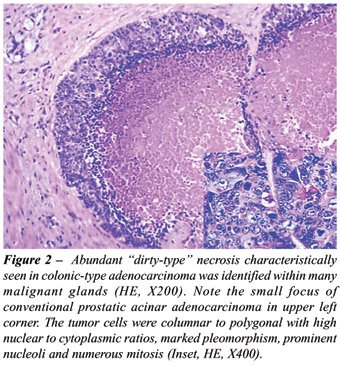
infiltrating the prostatic parenchyma (Figure-1). Abundant “dirty-type”
necrosis within malignant glandular structures, characteristically seen
in colonic-type adenocarcinoma, was identified (Figure-2). The tumors
cells were columnar to polygonal with high nuclear to cytoplasmic ratios,
marked pleomorphism, and prominent nucleoli (Figure-2, inset). They displayed
abundant mitotic activity and apoptosis. No intracellular or extracellular
mucin was identified. The morphology seen on the H&E stains was very
reminiscent of a high grade enteric-type adenocarcinoma. The tumor was
predominantly present on the left side, with extensive extraprostatic
extension and focally positive margins. Perineural as well as vascular
invasion was also identified. The tumor involved approximately 90% of
the left lobe and 10% of the right lobe of the gland. It also extended
into both seminal vesicles microscopically. Both left and right pelvic
lymph nodes sampled were negative for metastatic tumor.
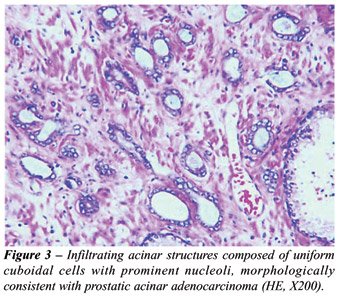
A second population of tumor cells was present
in both the right side core biopsy and prostatectomy specimens, mostly
in the right lobe. It consisted of infiltrating acinar structures composed
of uniform cuboidal cells with prominent nucleoli, morphologically characteristic
of conventional prostatic acinar adenocarcinoma, Gleason’s score
4+3 (Figure-3). The tumor was a minor component in the prostatectomy specimen
occupying less than 5% of the total gland volume and was in close proximity
to the enteric type adenocarcinoma.
Immunohistochemistry for cytokeratin 20
(CK20), prostate-specific antigen (PSA), and CDX2 were performed on the
right-sided core biopsy containing the high grade adenocarcinoma reminiscent
of colonic adenocarcinoma. The tumor was diffusely and strongly positive
for CK20 and CDX2 and negative for PSA. This led to the suspicion of metastatic
poorly differentiated adenocarcinoma consistent with colonic primary on
prostatic biopsy specimen.
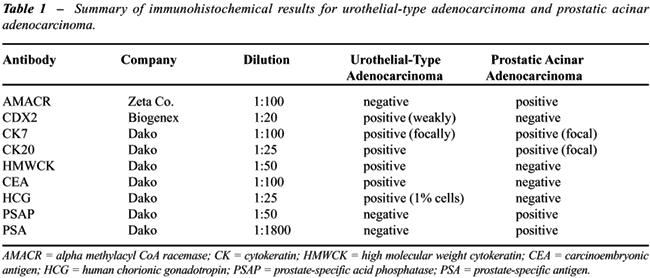
Furthermore, a panel of immunohistochemical
stains for cytokeratin 7 and 20, PSA, PSAP, thrombomodulin, alpha methylacyl
CoA racemase (AMACR), CDX2, monoclonal carcinoembryonic antigen (mCEA),
CD31, human chorionic gonadotropin (HCG), alpha-fetoprotein (AFP), placental
alkaline phosphatase (PLAP), and high-molecular weight keratin (HMWK)
were performed on sections from the prostatectomy specimen. The high grade
adenocarcinoma showed strong diffuse expression of CK20, CDX2, and HMWK
as well as focal expression of CK7 and mCEA. The tumor was completely
negative for PSA, PSAP, thrombomodulin, AMACR, CD31, AFP, and PLAP. Less
than 1% of cells were positive for HCG. The immunostaining results are
summarized in Table-1 and partially illustrated in Figure-4 and 5.
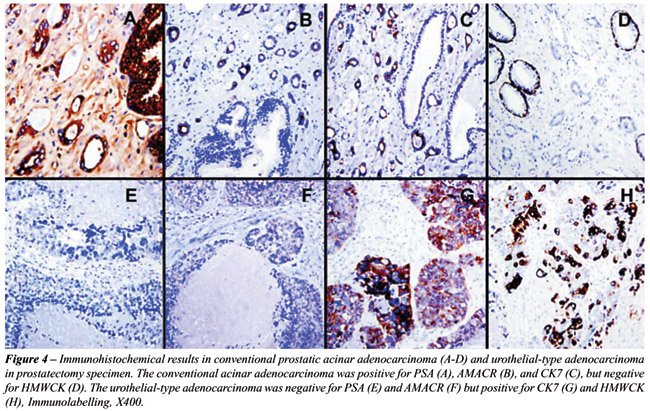
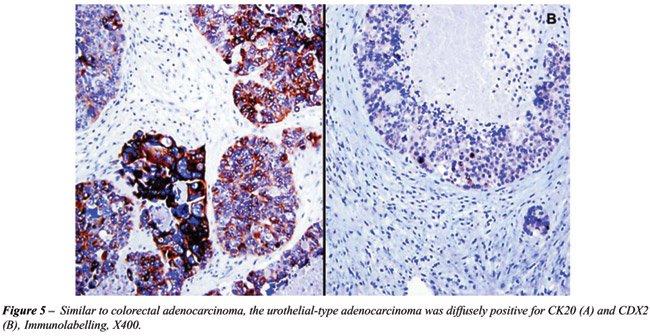
COMMENTS
Within
the prostate gland, urothelium lines primarily the prostatic urethra but
may extend to portions of major prostatic ducts. Even prostatic acini
can undergo urothelial (transitional) metaplasia. Theoretically, any lesion
which is known to occur elsewhere in the urothelial tract may occur in
the prostate gland, and urothelial lesions such as inverted papilloma
and urothelial carcinoma have all been described in the prostate (3-5).
Adenocarcinoma, a tumor usually occurring in the bladder or urethra, can
assume patterns similar to colonic adenocarcinoma including the typical
enteric pattern, signet-ring pattern, and mucinous pattern (6,7). It is
suggested that adenocarcinoma of bladder or urethra usually arises in
precursor lesions like urethritis glandularis (1) or periurethral structures
such as Cowper’s glands. Urothelial-type adenocarcinoma arising
in the prostate is exceedingly rare, with only four previous cases reported
in the literature.
The differential diagnosis for urothelial-type
adenocarcinoma of the prostate includes prostatic mucinous adenocarcinoma,
prostatic ductal adenocarcinoma, and metastatic colonic adenocarcinoma.
Mucinous adenocarcinoma of the prostate typically has cribriform glands
with bland cytology characteristic of conventional prostatic acinar adenocarcinoma.
These tumors are typically intermingled with conventional acinar adenocarcinoma,
and usually classified as Gleason score 3+4. The epithelium may float
in mucin and the cells express PSA and PSAP (8). Ductal adenocarcinomas
of the prostate may very closely resemble urothelial type adenocarcinoma
but are also positive for PSA and PSAP (9).
Perhaps the most challenging differential
diagnosis is that of metastatic colorectal adenocarcinoma, which can look
almost identical to urothelial-type adenocarcinoma. Immunohistochemistry
can be helpful in these situations as we previously reported (10). Cytokeratin
7 (CK7) is typically positive in urothelial carcinoma but negative in
colorectal carcinoma (10), although both tumors are positive for cytokeratin
20. In this case, tumor cells were focally positive for CK7. Villin, reported
to be positive in colonic adenocarcinoma and negative in urothelial carcinoma
with glandular differentiation, has been shown to be expressed in enteric-type
urothelial adenocarcinoma (11). High molecular weight keratins, including
CK 1, 5, 10 and 14, have been shown to be highly expressed in urothelial
carcinomas and only rarely expressed in colonic and prostatic adenocarcinomas
(12). It has also been reported that HCG producing tumor cells can be
seen in urothelial carcinoma.
AMACR was first identified as a prostate
cancer marker (13). Further studies showed its expression in the majority
of colonic adenocarcinomas (14). In addition, AMACR is also positive in
prostatic mucinous adenocarcinoma and ductal adenocarcinoma, although
the reactivity may be reduced focally. AMACR is typically negative or
weakly positive in urothelial carcinoma and positive in a small percentage
of adenocarcinomas of the bladder (15). The negative AMACR stain in this
high grade urothelial-type adenocarcinoma supports its urothelial differentiation
and is inconsistent with prostatic ductal adenocarcinoma or colonic adenocarcinoma.
In the case reported here, the finding of a small subset of HCG positive
cells also supports the urothelial origin or differentiation of this tumor.
Curtis et al. (2) proposed that a panel
of antibodies to PSA, PSAP, CK7, CK20 and HMWK would be useful in differentiating
urothelial-type adenocarcinoma, metastatic colorectal adenocarcinoma,
and conventional prostatic acinar adenocarcinoma. Urothelial-type adenocarcinomas
should be positive for HMWK and CK7 and may be positive for CK20, but
should not express PSA and PSAP. Colorectal carcinoma should be positive
for CK20 but negative for all other markers. Prostatic acinar or ductal
adenocarcinoma should express PSA and PSAP but be negative for all other
markers. Our tumor strongly expressed CK20 and CEA, and was focally positive
for CDX2. This profile may suggest a colorectal primary. However, our
tumor also expressed HMWK and CK7, which is not typical of colon cancer.
More importantly, the patient had a completely negative workup for primary
colon cancer. Hence we feel that our case is most consistent with urothelial-type
adenocarcinoma of the prostate gland showing a degree of enteric differentiation.
Although our tumor did not contain mucin, it had other characteristics
commonly seen in colorectal carcinoma including extensive dirty necrosis
and a focal cribriform growth pattern.
The etiology of this malignant tumor is
unknown. Although urethritis glandularis has been proposed to be a precursor
lesion for urothelial-type adenocarcinoma, our patient did not have evidence
of urethritis glandularis. In fact, the sampled prostatic urethral mucosa
was histologically unremarkable. One possibility is that the tumor arose
from a prostatic duct, which may be lined by urothelial-type mucosa. Of
interest, our patient also had a small focus of conventional acinar-type
prostatic adenocarcinoma, which was also described in two of the four
previous case reports. Therefore, an alternative possibility is that this
tumor may be derived from a stem cell which underwent biphasic differentiation
to urothelial-type adenocarcinoma and conventional prostatic acinar adenocarcinoma.
Due to the rarity of these tumors, it is
difficult to predict their behavior. One previously reported patient died
of disease with liver metastasis within one year of initial diagnosis
(2). Another patient experienced local recurrence 4 years status-post
simple prostatectomy (1). The two other patients showed no evidence of
disease one year1 and 16 months (2) status-post radical prostatectomy.
Our patient developed both local recurrence as well as multiple metastases
to lung, liver, and bone within 4 months of prostatectomy. It was observed
that the metastatic tumors of this patient were morphologically and immunohistochemically
identical to the urothelial-type adenocarcinoma of the prostate. Therefore,
it is important to note that these tumors may behave in an aggressive
manner.
In conclusion, we have described a rare
high grade urothelial-type adenocarcinoma arising in the prostate. This
primary prostatic tumor, mimicking metastatic colorectal adenocarcinoma
due to its striking morphologic resemblance as well as overlapping immunohistochemical
phenotype, should be recognized for appropriate clinical management. Our
study supports the previous notion that high molecular weight keratin
may be the key marker for differentiating these tumors from colorectal
metastasis.
CONFLICT OF INTEREST
None declared.
REFERENCES
- Tran KP, Epstein JI: Mucinous adenocarcinoma of urinary bladder type arising from the prostatic urethra. Distinction from mucinous adenocarcinoma of the prostate. Am J Surg Pathol. 1996; 20: 1346-50.
- Curtis MW, Evans AJ, Srigley JR: Mucin-producing urothelial-type adenocarcinoma of prostate: report of two cases of a rare and diagnostically challenging entity. Mod Pathol. 2005; 18: 585-90.
- Malpica A, Ro JY, Troncoso P, Ordonez NG, Amin MB, Ayala AG: Nephrogenic adenoma of the prostatic urethra involving the prostate gland: a clinicopathologic and immunohistochemical study of eight cases. Hum Pathol. 1994; 25: 390-5.
- Nicolaisen GS, Williams RD: Primary transitional cell carcinoma of prostate. Urology. 1984; 24: 544-9.
- Sawczuk I, Tannenbaum M, Olsson CA, deVere White R: Primary transitional cell carcinoma of prostatic periurethral ducts. Urology. 1985; 25: 339-43.
- Grignon DJ, Ro JY, Ayala AG, Johnson DE, Ordonez NG: Primary adenocarcinoma of the urinary bladder. A clinicopathologic analysis of 72 cases. Cancer. 1991; 67: 2165-72.
- Wang HL, Lu DW, Yerian LM, Alsikafi N, Steinberg G, Hart J, et al.: Immunohistochemical distinction between primary adenocarcinoma of the bladder and secondary colorectal adenocarcinoma. Am J Surg Pathol. 2001; 25: 1380-7.
- Epstein JI, Lieberman PH: Mucinous adenocarcinoma of the prostate gland. Am J Surg Pathol. 1985; 9: 299-308.
- Christensen WN, Steinberg G, Walsh PC, Epstein JI: Prostatic duct adenocarcinoma. Findings at radical prostatectomy. Cancer. 1991; 67: 2118-24.
- Wang NP, Zee S, Zarbo RJ, Bacchi CE, Gown AM: Coordinate expression of cytokeratins 7 and 20 defines unique subsets of carcinomas. Appl Immunohistochem 1995; 3: 99–107.
- Tamboli P, Mohsin SK, Hailemariam S, Amin MB: Colonic adenocarcinoma metastatic to the urinary tract versus primary tumors of the urinary tract with glandular differentiation: a report of 7 cases and investigation using a limited immunohistochemical panel. Arch Pathol Lab Med. 2002; 126: 1057-63.
- Yang XJ, Lecksell K, Gaudin P, Epstein JI: Rare expression of high-molecular-weight cytokeratin in adenocarcinoma of the prostate gland: a study of 100 cases of metastatic and locally advanced prostate cancer. Am J Surg Pathol. 1999; 23: 147-52.
- Xu J, Stolk JA, Zhang X, Silva SJ, Houghton RL, Matsumura M, et al.: Identification of differentially expressed genes in human prostate cancer using subtraction and microarray. Cancer Res. 2000; 60: 1677-82.
- Jiang Z, Fanger GR, Banner BF, Woda BA, Algate P, Dresser K, et al.: A dietary enzyme: alpha-methylacyl-CoA racemase/P504S is overexpressed in colon carcinoma. Cancer Detect Prev. 2003; 27: 422-6.
- Suh N, Yang XJ, Tretiakova MS, Humphrey PA, Wang HL: Value of CDX2, villin, and alpha-methylacyl coenzyme A racemase immunostains in the distinction between primary adenocarcinoma of the bladder and secondary colorectal adenocarcinoma. Mod Pathol. 2005; 18: 1217-22.
____________________
Accepted
after revision:
March 5, 2006
_______________________
Correspondence address:
Dr. Brian P. Adley
Department of Pathology, Feinberg 7-325
Northwestern Univ, Feinberg Sch Med
251 E. Huron Street
Chicago, Illinois, 60611, USA
Fax: + 1 312 926-3127
E-mail: b-adley@northwestern.edu
EDITORIAL COMMENT
This
is a very well written paper on a very rare tumor. Adenocarcinoma of the
urinary tract may correspond to 3 possibilities: glandular differentiation
of a conventional urothelial carcinoma (more frequently), secondary involvement
(more frequently from the prostate or colorectal region), and primary
(more rarely). Clinical work-up as well as morphology characteristics
and immunohistochemistry help in the differential diagnosis.
Primary
adenocarcinomas may also arise from the urethra or distal prostatic ducts.
In this case the differential diagnosis is much more difficult because
the great majority of the cases are conventional adenocarcinomas of the
prostate and the pathologist is not aware of the possibility of dealing
with a so rare condition. The differential must be done with a thorough
clinical work-up to exclude adenocarcinoma of the rectum or colon and,
on morphology, only by immunohistochemistry.
Only
4 cases were previously published of a primary urothelial-type adenocarcinoma
of the prostate. The final diagnosis is mainly based on immunohistochemistry.
Dr. Athanase
Billis
Full-Professor of Pathology
State University of Campinas, Unicamp
Campinas, São Paulo, Brazi
E-mail: athanase@fcm.unicamp.br