THE
BEGINNING OF THE 21ST CENTURY: A PARADIGM SHIFT IN THE SURGICAL MANAGEMENT
OF RENAL CELL CARCINOMA IN SOUTH AMERICA
(
Download pdf )
MARCOS F. DALL’OGLIO, ALEXANDRE CRIPPA, CESAR CAMARA, JOSE PONTES-JUNIOR, JOSE R. COLOMBO, ADRIANO J. NESRALLAH, LUIS C. N. OLIVEIRA, MIGUEL SROUGI
Division of Urology, University of Sao Paulo Medical School and Cancer Institute of Sao Paulo, ICESP, Sao Paulo, SP, Brazil
Clinical Urology
Vol. 36 (6):
670-677, November - December, 2010
doi: 10.1590/S1677-55382010000600004
ABSTRACT
Purpose:
The incidence of renal cell carcinoma (RCC) has been rising by 2.3 to
4.3% every year over the past three decades. Previously, RCC has been
known as the internist’s tumor; however, it is now being called
the radiologist’s tumor because 2/3 are now detected incidentally
on abdominal imaging. We compared patients who were treated toward the
end of the 20th century to those treated during the beginning of the 21st
century with regard to RCC size and type of surgical treatment.
Materials
and Methods: The study included 226 patients. For analysis of tumor size,
we considered a cut point of < 4 cm and > 4 cm. For analysis of
type of surgery performed, we considered radical and partial nephrectomy.
Results:
After the turn of the century, there was a reduction of 1.57 ±
0.48 cm in the size of the RCC that was operated on. Nephron sparing surgeries
were performed in 17% of the cases until the year 2000, and 39% of the
tumors were < 4 cm. From 2001, 64% of the tumors measured < 4 cm
and 42% of the surgeries were performed using nephron sparing techniques.
Mean tumor size was 5.95 cm (± 3.58) for the cases diagnosed before
year 2000, and cases treated after the beginning of 21st century had a
mean tumor size of 4.38 cm (± 3.27).
Conclusions:
Compared with the end of the 20th century, at the beginning of the 21st
century due to a reduction in tumor size it was possible to increase the
number of nephron sparing surgeries.
Key
words: carcinoma; renal cell; incidence; diagnostic imaging;
urological surgical procedures
Int Braz J Urol. 2010; 36: 670-7
INTRODUCTION
The
incidence of the renal cell carcinoma (RCC) has been increasing by 2.3
to 4.3% per year over the last three decades in the United States (1).
Unfortunately, approximately 1/3 of the patients who have been diagnosed
with RCC will die due to progression to metastatic disease (1).
Since Bell’s classic study (2), the
first to relate RCC size to prognosis, there has been a variety of stage
modifications in the TNM system related to tumor size variations. This
information suggests that tumor growth significantly influences the prognosis
of this lethal disease. The majority of studies that have reported a large
number of patients indicate that the stratification size related to RCC
prognosis is between 4 and 5 cm (3,4).
Fortunately, most RCC cases diagnosed today
are incidental tumors with smaller sizes, and identified after ultrasonography
(US) or computed tomography (CT) examinations performed for other reasons
(5). There has been a reduction in size of recently diagnosed tumors from
7.8 to 5.3 cm, and an increase in organ confined disease, 47 to 78%, from
1989 to 1998 respectively (6). Due to the demonstration that nephron-sparing
surgery is effective in RCC, the number of nephron-sparing surgeries (NSS)
has also grown (7). At the turn of the century, the management of RCC
underwent a paradigm shift favoring nephron-sparing surgery in a large
part due to the identification of smaller-sized lesions, and similar oncologic
outcomes. The Mayo Clinic study showed that patients who underwent radical
nephrectomy presented a higher possibility to have elevated serum creatinine
levels and proteinuria higher than 2.0 ng/mL (8).
The goal of this study was to compare RCC
size between cases treated during the end of the 20th century to those
treated during the beginning of the 21st century. We also analyzed the
type of surgeries that were performed during both periods.
MATERIALS AND METHODS
During
the period between January 1995 and December 2005, 226 patients with RCC
who underwent surgery at our institution had their clinical data retrospectively
analyzed. Preoperative evaluation included blood and imaging exams such
as US, CT, and/or magnetic nuclear resonance, chest x-ray, bone scintillography,
and occasionally, urography.
Initially, we analyzed whether the clinical
presentation at the time of diagnosis was incidental or symptomatic. Then,
a single pathologist analyzed the anatomic and pathological variables
as follows: histology type, Fuhrman nuclear grade, presence of intra-tumoral
microvascular invasion, and tumor size. The study included calculation
of disease-free survival and specific cancer-survival curves with respect
to all the above variables. The study compared the tumor size over these
11 years and the surgical treatment, nephron sparing or radical surgeries.
The study also included an analysis of the individuals who underwent NSS.
The features of RCC treated during the last six years of the 20th century
were compared to the first five years of the 21st century.
The post-surgical follow-up of the individuals
was performed in a clinic, and afterward, there was a 3 months period
of confirmation by telephone of the current health status of the patient.
The clinical follow-up included a chest x-ray, abdominal CT scan and/or
US, and blood tests every 4 months during the first year, every half-year
from the second to the fifth year, then annually after this period.
The statistical analysis was based on Kaplan-Meier curves and the differences
in survival between the groups used the Log rank test. Results were considered
significant when the p-value was below 5% (p < 0.05).
RESULTS
Up to the year 2000, the majority of tumors had a size greater than > 4 cm (61%). However, after the turn of the century, there was a change in tumor size that underwent resection. After 2001, 64% of the tumors were < 4 cm, and the nephron-sparing surgery was duplicated as shown in Table-1.

Table-2 shows the average, median, and standard
deviations of the tumor size from patients who underwent surgery from
1995 to 2005.
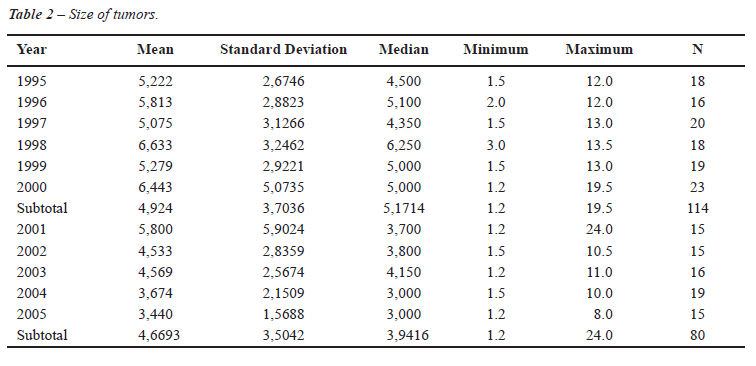
The types of surgeries performed during
the study period are shown in Table-3. It is important to emphasize that
during the period of 1995-2000 the NSS and radical nephrectomy were 17%
and 83%, while during 2001-2005 it was 42% and 58%, respectively.
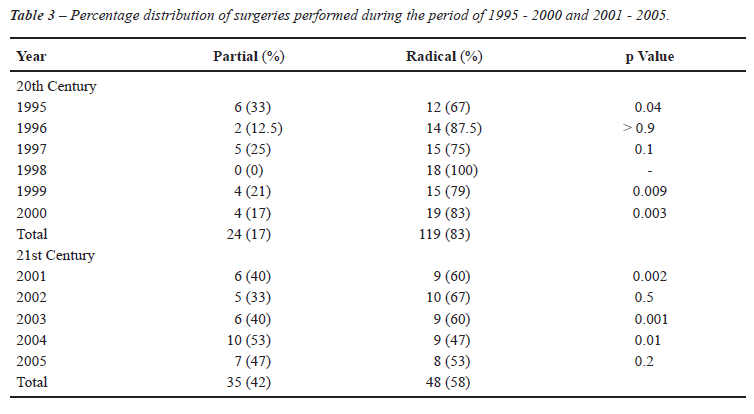
Figure-1 shows the profile of tumor sizes
over the years, allowing for the perception of a gradual reduction in
the tumor size over this period.
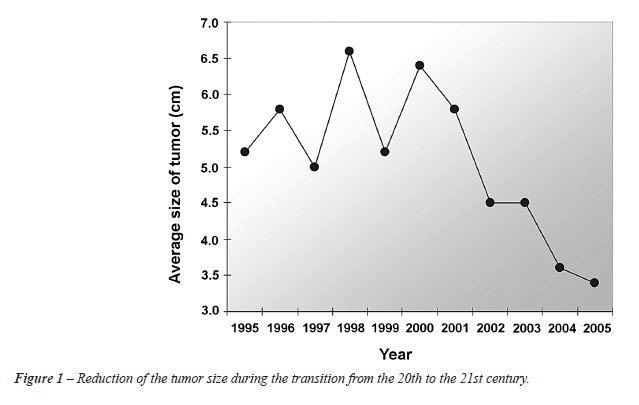
Table-4 shows RCC sizes in four subgroups
noting the increase in the incidence of tumors with sizes < 3 cm and
between 3-4 cm, as well as the reduction in the incidence of the tumor
sizes in subgroups from 4.1 - 7 cm, and > 7 cm after the year 2000.
The demographic and anatomic/pathological
data of patients who underwent NSS is described in Table-5.

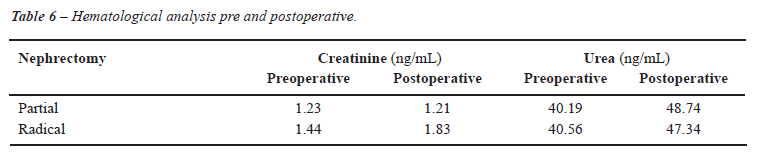
The main finding is that majority of tumors
were T1 (95%), 78% incidental and low degree. The creatinine serum and
urea pre and postoperative are demonstrated in Table-6.
COMMENTS
Our
study shows that there has been a paradigm shift in the surgical treatment
of RCC in Brazil. A significant reduction in the average tumor size has
taken place, 5.1 cm from the end of the past century, to 3.9 cm after
the year 2001. This has increased (25%) the indication for nephron sparing
surgery by 17% until 2000 and 42% after 2001.
For a long time, tumor size has been considered
one of the most important independent prognostic factors for RCC. This
has resulted in a number of frequent publications addressing this issue
(9,10), as well as, encouraging continuous proposals for changes in the
staging of the disease (5). These frequent changes in the RCC staging,
always related to the tumor size, certainly assure that this is the major
prognostic factor, and that it most faithfully defines the disease’s
behavior.
The survival outcome for tumors < 7 cm
that were re-resected by radical nephrectomy is similar; however, the
possibility of RCC recurrence varies from 2.6% to 9% in T1a and T1b tumors,
respectively (11). In this study, we found that the average survival of
patients with T1a tumors was 91%, while in T1b tumors, 79% were free from
the disease in 5 years. The size of the tumor is so relevant in RCC that
the growth of 1 cm in the RCC size increases the possibilities of cancer
progression by 17%, according to the important editorial by Marshall (12).
Patients with RCC greater than 5 cm have a five-fold greater chance of
dying due to the disease when compared to those with tumors with less
than 5 cm (risk ratio = 4.93) (12).
Although RCC used to be referred to as the
internist’s tumor, it may now be more appropriate to refer to it
as the radiologist’s tumor, because 60% of renal tumors are detected
incidentally during abdominal imaging obtained for other reasons. In this
context, laparoscopic NSS proved to be effective and safe in the treatment
of renal tumors (13). Currently, robotic NSS is already a reality, with
one important series performing surgeries for tumors from 1.4 to 3.6 cm
(14). Despite the development of alternative ablative techniques for solid
kidney lesions, surgical excision remains the cornerstone in treating
RCC.
In our study, we found that after the turn
of the century, there was a significant difference between the size of
the tumors and the type of surgery performed. It was only after 2001 that
the median tumor size decreased below 4 cm, 39% of cases before 2001 compared
to 64% after. On the other hand, the percentage of nephron sparing surgeries
increased significantly from 17% to 42%.
The size of renal tumors at the time of
diagnosis has been decreasing over the years, with a reduction in the
average size resected lesions from 7.8 to 5.3 cm, from 1989 to 1998 (7).
For this reason, a migration of the RCC stage has taken place according
to Kane et al. (15). This migration has occurred particularly in the pT1
stage where the median RCC size decreased from 4.1 in the year 1993, to
3.6 cm in 2003. The survival gain for patients treated in 1993 to 1998
rose by 3.3%. It is worth pointing out that between the years 1993 and
2004, the proportion of patients with RCC in Stage I grew from 43% to
57%; on the other hand, the proportion of patients with pT4 stage decreased
during the same period from 27.4% to 18.7% (16).
With a normal contralateral kidney, the
cumulative incidence of renal insufficiency (defined as a serum creatinine
level of > 2.0 mg/dL) at 10 years has been reported to be significantly
higher after radical nephrectomy than after partial nephrectomy (22% vs.
12% (17). Proteinuria was also more common after radical nephrectomy (55%
vs. 35%) (17). Also, metachronous renal tumors in the contralateral kidney
can occur in up to 10% of patients (18), underscoring the importance of
avoiding unnecessary nephron loss. Recent findings suggest that NSS is
greatly underused in the USA because, in a large nationwide hospital database,
only 9.6% of patients with surgically treated renal tumors underwent partial
nephrectomy (19). At many academic centers, partial nephrectomy comprises
60-70% of the operations for RCC (20). However, when using the nationwide
inpatients sample, these authors reported that only 7.5% of kidney tumor
operations in the USA from 1988 to 2002 were partial nephrectomies (19).
In England, a similar underuse of partial nephrectomy was reported in
2002 with only 4% out of 2671 nephrectomies performed (21).
One of the limitations of this study is
that it is a retrospective analysis; however, the fact that we have a
reliable database of individuals treated by a same group of surgeons from
a single institution is a positive point. We believe that with the increasing
diagnosis rate of solid renal lesions with progressively smaller sizes,
the indications for nephron sparing therapies will increase significantly,
favoring the preservation of the renal function and improving RCC outcomes.
CONCLUSIONS
Comparing patients diagnosed with RCC after the beginning of the 21st century to those diagnosed before, the patients diagnosed later were more likely to undergo nephron-sparing surgery increasing the probability of avoiding later chronic renal insufficiency.
ACKNOWLEDGEMENT
Adriana Sañudo performed the statistical analysis.
CONFLICT OF INTEREST
None declared.
REFERENCES
- Jemal A, Siegel R, Ward E, Murray T, Xu J, Smigal C, et al.: Cancer statistics, 2006. CA Cancer J Clin. 2006; 56: 106-30.
- Bell ET: Classification of renal tumors with observations on frequency of various types. J Urol. 1938; 39: 238-43.
- Lau WK, Cheville JC, Blute ML, Weaver AL, Zincke H: Prognostic features of pathologic stage T1 renal cell carcinoma after radical nephrectomy. Urology. 2002; 59: 532-7.
- Hafez KS, Fergany AF, Novick A: Nephron sparing surgery for localized renal cell carcinoma: impact of tumor size on patient survival, tumor recurrence and TNM staging. J Urol. 1999; 162: 1930-3.
- Zisman A, Pantuck AJ, Chao D, Dorey F, Said JW, Gitlitz BJ, et al.: Reevaluation of the 1997 TNM classification for renal cell carcinoma: T1 and T2 cutoff point at 4.5 rather than 7 cm. better correlates with clinical outcome. J Urol. 2001; 166: 54-8.
- Homma Y, Kawabe K, Kitamura T, Nishimura Y, Shinohara M, Kondo Y, et al.: Increased incidental detection and reduced mortality in renal cancer--recent retrospective analysis at eight institutions. Int J Urol. 1995; 2: 77-80.
- Lee CT, Katz J, Shi W, Thaler HT, Reuter VE, Russo P: Surgical management of renal tumors 4 cm. or less in a contemporary cohort. J Urol. 2000; 163: 730-6.
- Patard JJ, Pantuck AJ, Crepel M, Lam JS, Bellec L, Albouy B, et al.: Morbidity and clinical outcome of nephron-sparing surgery in relation to tumour size and indication. Eur Urol. 2007; 52: 148-54.
- Kontak JA, Campbell SC: Prognostic factors in renal cell carcinoma. Urol Clin North Am. 2003; 30: 467-80.
- Dall’Oglio MF, Srougi M, Nesrallah L, Leite KM, Hering F, Bomfim Ade C, et al.: Must the TNM staging of the renal cell carcinoma be modified again? Rev Assoc Med Bras. 2003; 49: 86-90.
- Patard JJ, Shvarts O, Lam JS, Pantuck AJ, Kim HL, Ficarra V, et al.: Safety and efficacy of partial nephrectomy for all T1 tumors based on an international multicenter experience. J Urol. 2004; 171: 2181-5.
- Marshall FF: Renal cell carcinoma - prognostic factors. J Urol. 2003; 170: 2233.
- Aron M, Haber GP, Gill IS: Laparoscopic partial nephrectomy. BJU Int. 2007; 99: 1258-63.
- Rogers CG, Singh A, Blatt AM, Linehan WM, Pinto PA: Robotic partial nephrectomy for complex renal tumors: surgical technique. Eur Urol. 2008; 53: 514-21.
- Nguyen MM, Gill IS, Ellison LM: The evolving presentation of renal carcinoma in the United States: trends from the Surveillance, Epidemiology, and End Results program. J Urol. 2006; 176: 2397-400; discussion 2400.
- Kane CJ, Mallin K, Ritchey J, Cooperberg MR, Carroll PR: Renal cell cancer stage migration: analysis of the National Cancer Data Base. Cancer. 2008; 113: 78-83.
- Lau WK, Blute ML, Weaver AL, Torres VE, Zincke H: Matched comparison of radical nephrectomy vs nephron-sparing surgery in patients with unilateral renal cell carcinoma and a normal contralateral kidney. Mayo Clin Proc. 2000; 75: 1236-42.
- Uzzo RG, Novick AC: Nephron sparing surgery for renal tumors: indications, techniques and outcomes. J Urol. 2001; 166: 6-18.
- Hollenbeck BK, Taub DA, Miller DC, Dunn RL, Wei JT: National utilization trends of partial nephrectomy for renal cell carcinoma: a case of underutilization? Urology. 2006; 67: 254-9.
- Russo P, Huang W: The medical and oncological rationale for partial nephrectomy for the treatment of T1 renal cortical tumors. Urol Clin North Am. 2008; 35: 635-43.
- Nuttall M, Cathcart P, van der Meulen J, Gillatt D, McIntosh G, Emberton M: A description of radical nephrectomy practice and outcomes in England: 1995-2002. BJU Int. 2005; 96: 58-61.
____________________
Accepted
after revision:
January 14, 2010
_______________________
Correspondence
address:
Dr. Marcos F. Dall’Oglio
Rua Barata Ribeiro, 398 / 501
Sao Paulo, SP, 01308-000, Brazil
Fax: + 55 11 3159-3618
E-mail: marcosdallogliouro@terra.com.br
EDITORIAL COMMENT
The paper by Dall’Oglio et al. nicely depicts that a paradigm shift has occurred in the surgical management of localized renal masses in Brazil paralleling similar changes worldwide particularly for lesions = 4 cm. At the present time, nephron-sparing surgery remains the “gold standard” for the management of small renal masses (SRM), with clear benefits in terms of cardiovascular toxicity while minimizing the risk of post-operative dialysis requirement. There is an unquestionable change in our underlying surgical approach to SRM in that the clinical question often asked is: when should a partial nephrectomy not be performed? In my clinical practice, over 70% of patients with renal masses less than 7 cm are treated by partial nephrectomy (in the absence of absolute indications for partial nephrectomy i.e. solitary kidney, underlying renal insufficiency, or bilateral renal masses). Whether the partial nephrectomy is performed using an open, pure laparoscopic, or robotic assisted laparoscopic approach is simply a technical consideration taking size, location, and surgical expertise into account. We cannot get away from the simple fact that for SRM, a partial nephrectomy (irrespective of its approach) is a better treatment choice for patients than radical nephrectomy. Recently, percutaneous (and laparoscopic) ablative techniques (i.e. radiofrequency ablation and cryoablation) have been proposed as a treatment alternative for SRM in well selected cases (typically lesions less than 2.5 cm and away from the renal hilum) understanding such treatment alternatives do not have long-term data (beyond 10 years) and require routine serial imaging following the ablative procedure. With these clear limitations, I feel percutaneous (and laparoscopic) ablative procedures should only be offered to a select subset of patients (1). With evolving technology and imaging modalities, newer treatment alternatives will become readily available to patients with SRM however partial nephrectomy has set the bar and we must never loose sight of the clear benefits it offers to our patient population. The impetus lies on the scientific community to develop imaging modalities or validate percutaneous renal biopsy strategies able to distinguish benign from malignant renal neoplasms such that treatment can be geared to those requiring definitive intervention.
REFERENCE
1. Spiess PE: Surgical management of locally recurrent renal cell carcinoma post-renal cryoablation: Importance of stringent selection criteria. Urol Oncol. 2010; 28: 241-2.
Dr.
Philippe E. Spiess
Assistant Professor of Urology Oncology
H. Lee Moffitt Cancer Center
Tampa, Florida, USA
E-mail: philippe.spiess@moffitt.org