SONOGRAPHIC
DETECTION OF RENAL AND URETERAL STONES. VALUE OF THE TWINKLING SIGN
(
Download pdf )
MICHAEL MITTERBERGER, FRIEDRICH AIGNER, LEO PALLWEIN, GERMAR-MICHAEL PINGGERA, RICHARD NEURURER, PETER REHDER, FERDINAND FRAUSCHER
Departments of Radiology II (FA, LP, FF) and Urology (MM, GMP, RN, PR), Medical University Innsbruck, Innsbruck, Austria
ABSTRACT
Purpose:
To compare the detection of urinary stones using standard gray scale ultrasound
for diagnostic accuracy using the color Doppler “twinkling sign”.
Materials and Methods: Our study population
consisted of forty-one patients who demonstrated at least one urinary
stone on unenhanced CT evaluation of the kidneys or ureters. Each patient
was evaluated using gray scale ultrasound and color Doppler imaging by
an observer who was blinded to the CT results.
Results: Seventy-seven stones were present
in 41 patients, including 47 intrarenal stones, 5 stones in the renal
pelvis, 8 stones at the ureteropelvic junction, 5 ureteral stones and
12 stones at the ureterovesical junction. Based upon gray scale sonography
the diagnosis of stone was made with confidence in 66% (51/77) of locations.
Based upon Doppler sonography using the twinkling sign, the diagnosis
of stone was made with confidence in 97% (75/77) of locations. Clustered
ROC analysis demonstrated that the Doppler twinkling sign (Az = 0.99)
was significantly better than conventional gray scale criteria (Az = 0.95)
for the diagnosis of urinary stones (p = 0.005, two-sided test).
Conclusions: The color Doppler twinkling
sign improves the detection, confidence and overall accuracy of diagnosis
for renal and ureteral stones with minimal loss of specificity.
Key
words: urolithiasis; ultrasound; Doppler; computed tomography
Int Braz J Urol. 2009; 35: 532-41
INTRODUCTION
Detection
of urinary stones on ultrasound (US) may be problematic when the stones
are obscured by ultrasonic beam-attenuating tissue, such as renal sinus
fat, mesenteric fat, and bowel, or when their posterior acoustic shadowing
is weak (1-3). Despite the technical advances of US, radiologists have
difficulty confirming or excluding the presence of urinary stones when
the gray-scale findings are indeterminate.
The twinkling sign is a color-flow US artifact described behind calcifications
and presenting as a random color encoding in the region were shadowing
would be expected on gray-scale images (4). Recent studies have reported
that the twinkling sign may be useful for detection of urinary stones
(5-7).
The present study compares standard gray scale ultrasound with color Doppler
ultrasound (twinkling sign) for the detection of urinary stones, initially
diagnosed by unenhanced computed tomography (CT).
MATERIALS AND METHODS
Patients
Forty-one patients (24 males, 17 females; mean age: 50 ± 8.7 years, range: 19-74 years) with urinary stones (52 renal stones, 25 ureteral stones) were included in our study. All patients were evaluated for the presence of urinary stones by unenhanced spiral computed tomography (CT), which served as the “gold standard” for the diagnosis of urinary stones (8). In addition, plain abdominal radiography was performed in all patients. Our study had Institutional Review Board Approval and written informed consent was obtained from all patients prior to the US studies.
Ultrasound Technique
US examinations were done within 24 hours after CT. US examinations were
performed by one of two experienced radiologists (A.K; F.F.), who were
blinded to the CT findings. Gray-scale, color and power Doppler US, and
pulsed-wave spectral Doppler US were performed in all 41 patients. All
studies were performed with the Acuson Sequoia 512; (Acuson, MountainView,
Cal.) with a transmit frequency of 2.5 to 6.0 MHz. Gray-scale US criteria
included visualization of a hyperechoic structure with posterior acoustic
shadowing within the kidney and/or the ureter. For visualization of posterior
acoustic shadowing, focal zones were positioned at the depth of the stone,
or slightly deeper than the stone.
Color Doppler US was performed using a red-and-blue
color map and power Doppler US using a pink color map with a standardized
Doppler protocol to detect the twinkling artifact. For color Doppler,
gain was set to the point just below the threshold for color noise and
the pulse repetition frequency (= velocity scale) was set to 64 cm/sec
to eliminate color flow signals from renal blood flow. The color window
size was adjusted to cover the whole renal sinus. Doppler imaging began
with continuous sweeps of the renal sinus in the longitudinal and transverse
planes. For investigation of the ureter, the color window size was adjusted
for each part of the ureter as it was visualized. Evaluation of the ureter
began with the UPJ (ureteropelvic junction). The course of the ureter
was follow caudally from the UPJ to image the proximal ureter. The mid-ureter
was examined at the level where the ureter crossed the iliac vessels.
The distal-ureter was evaluated through a full urinary bladder. Whenever
a twinkling sign was present, a Doppler spectrum was obtained to exclude
arterial or venous flow. At each level, gray scale evaluation was followed
by color and power Doppler imaging for a twinkling sign. For each kidney/ureter
unit, gray-scale and Doppler findings were recorded at 8 locations: intrarenal
upper pole, intrarenal mid-sinus, intrarenal lower-pole, renal pelvis,
UPJ, mid-ureter, distal-ureter and ureterovesical junction. Each location
was evaluated whether or not there was a stone appearance for each gray-scale
and Doppler (color and power).
US findings were evaluated by consensus interpretation of the two radiologists
(AK, FF). The gray-scale US appearance of urinary stones was analyzed
for size, echo difference between stone and adjacent tissue, and posterior
acoustic shadowing. Stone size was determined on gray-scale US alone.
The location of the stones was determined either on gray-scale US or color
Doppler US findings. Echo difference between stone and adjacent tissue
was recorded as marked, slight, or indistinct. Posterior acoustic shadowing
was noted as absent, weak or strong. On color and power Doppler images,
the presence, appearance, and intensity of the twinkling sign was assessed.
The intensity of the color signal was recorded as 0 (= absent), 1 (= weak,
present) and 2 (= strong, present). Furthermore, the length of the twinkling
sign was classified and a length of > 1 cm was defined as 2 (= strong
present). At pulsed-wave spectral Doppler US, the pattern of the spectrum
was analyzed.
Computed Tomography Technique
All patients underwent an unenhanced helical CT examination using a Somatom Plus 4 unit (Siemens, Erlangen, Germany). Single breathhold, continuous, transverse helical acquisition was performed from the top of the kidneys to the base of the bladder with a 5-mm collimation, a 2:1 pitch, 120 kVp, 280 mAs, and a reconstruction at 2.5-mm intervals. No oral or intravenous contrast was administered. A typical examination lasted less than 30 seconds. The images were analyzed at a workstation that was capable of reconstruction processing. An independent radiologist reviewed each CT examination for the presence of stones in the kidneys and ureters. Each stone was classified as located in one of the eight previously described positions.
Statistical Analysis
The ultrasound
findings were compared with the findings on unenhanced CT. Stone size
was measured on axial CT images (maximum transverse diameter) and compared
with the size measured on gray-scale US (maximum transverse diameter).
In order to compare the detection rate of urinary stones by gray- scale
and Doppler imaging we used ROC analysis. Given the clustered nature of
our observations (2 kidneys per patient with 8 locations per kidney/ureter)
imaging findings were not independent for each patient. We therefore used
a clustered ROC analysis for this comparison with 16 clustered observations
per patient (9). In these few cases when two stones were detected by US
at a single location (n = 3) the finding was tabulated as a single observation.
The confidence of diagnoses was made according to the greatest confidence
among the stones in that location for each gray- scale and Doppler sonography.
A p-value of less than 0.05 was considered statistically significant (10).
RESULTS
The 77 urinary
stones had a mean size of 0.5 cm ± 0.3 on unenhanced CT. The mean
size of the 52 renal stones was 0.5 cm ± 0.2 (range, 0.3 - 1.0
cm) and of the 25 ureteral stones was 0.4 cm ± 0.3 (range, 0.3
- 1.0 cm). Forty-seven renal stones (90%) were located in calices and
5 in the renal pelvis (10%). Eight ureteral stones (32%) were located
at the UPJ, 2 (8%) in the mid-ureter, 3 (12%) in the distal-ureter, and
12 (48%) at the ureterovesical junction.
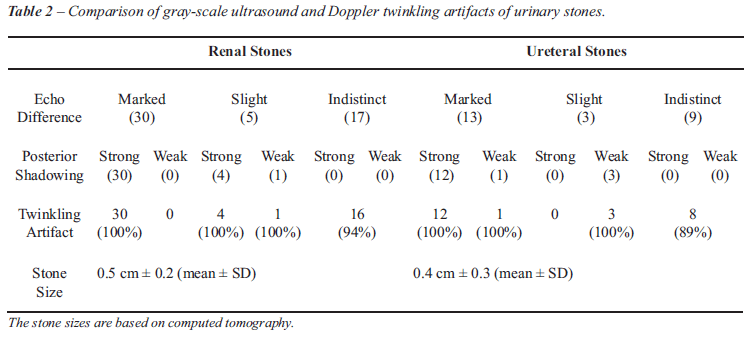
On gray-scale US 43 of 77 urinary stones (56%) showed marked echo difference
(30 renal stones, 13 ureteral stones), 8 urinary stones (10%; 5 renal
stones, 3 ureteral stones) showed slight echo difference, and 26 urinary
stones (34%; 17 renal stones, 9 ureteral stones) showed indistinct echo
difference (= no definite criteria for a urinary stone), respectively.
Forty-six urinary stones (60%) showed strong posterior acoustic shadowing,
and 5 urinary stones (7%) had weak posterior acoustic shadowing. The mean
size of the 51 urinary stones detected on gray-scale US was 0.7 cm ±
0.5 on. The mean size of the 35 renal stones was 0.7 cm ± 0.5 (range,
0.3 - 1.4 cm) and of the 16 ureteral stones was 0.6 cm ± 0.4 (range,
0.3 - 1.2 cm).
The twinkling sign was generated from 75 of 77 urinary stones (97%). One
renal stone (2%) and 1 ureteral stone (4%) did not demonstrate this Doppler
sign. As described in previous studies, the twinkling sign appeared as
a rapidly changing color complex seen persistently behind urinary stones,
like a comet’s tail.
Fifty-one of 52 of renal stones (98%) and 24 of 25 ureteral stones (96%)
demonstrated the signs. Forty-seven of 49 stones (96%) less or equal than
0.4 cm, and 100% of stones (n = 28) with a size greater than 0.4 cm had
the twinkling sign (Table-1 and Figures 1-3). Four ureteral stones with
indistinct echo difference showed the twinkling sign. Seventeen of 20
urinary stones (85%) with indiscrete posterior acoustic shadowing showed
twinkling sign. Sixty (80%) of 75 stones with twinkling sign had signals
with strong intensity (Table-2).

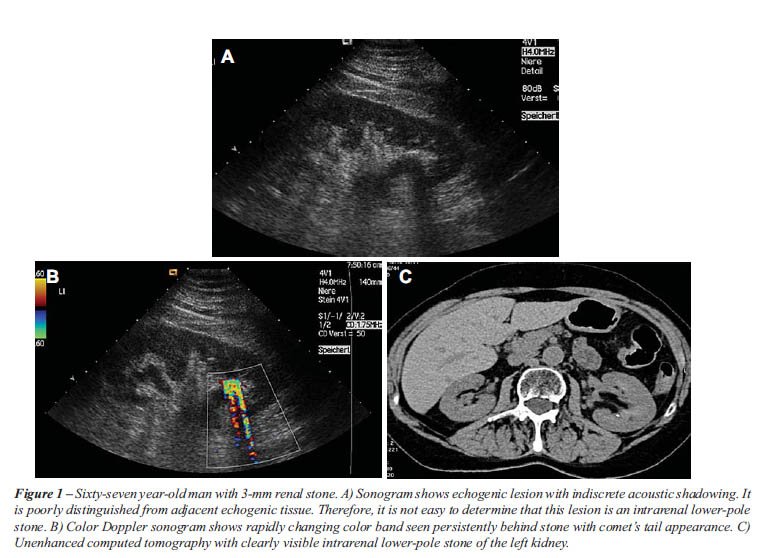
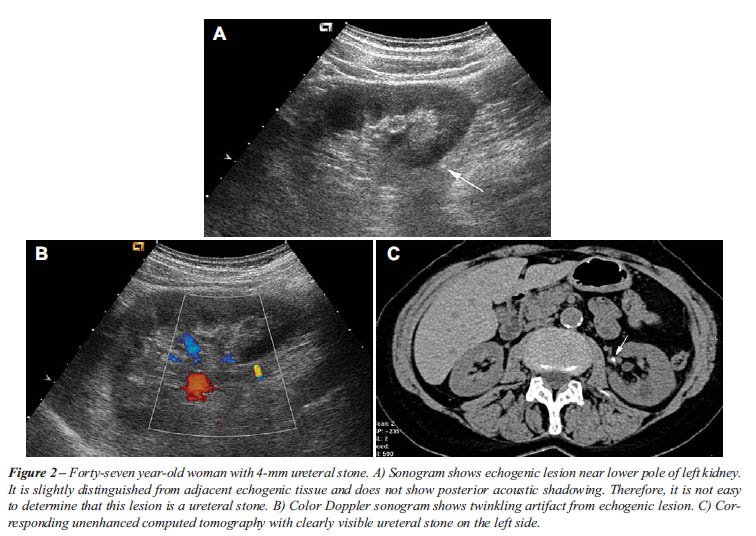
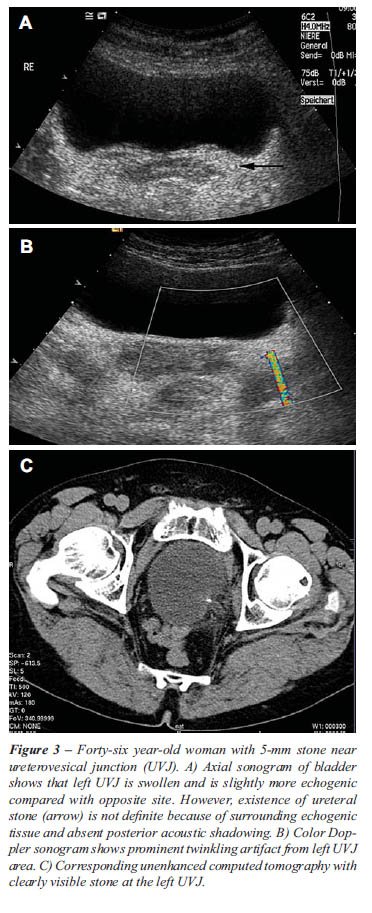
Based upon gray-scale criteria 10 false positive stones were suggested
within an intrarenal location in while one false positive stone was suggested
at a UPJ. One of the false positive intrarenal stones also demonstrated
a false positive twinkling sign.
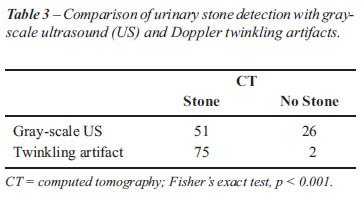
Gray- scale US detected 35 of 52 renal stones (67%) and 16 of 25 ureteral
stones (64%). Overall, gray scale US demonstrated a sensitivity of 66%
(51/77) while the twinkling sign demonstrated a sensitivity of 97% (75/77)
(Table-3). A twinkling sign was present in 51 of 52 (98%) of renal stones
and in 24 of 25 ureteral stones (96%). Based upon clustered ROC analysis
twinkling sign significantly improved the detection of urinary stones
(Az = 0.099) compared with gray-scale US (Az = 0.095), (p < 0.005).
Table-2 shows the location of urinary stones by size (= 0.4 cm and >
0.4 cm; based on CT size measurement) detected with gray-scale US and/or
with twinkling sign.
Forty-six of 77 urinary stones (60%) were visible on plain abdominal film.
All 46 of these stones showed a twinkling sign. The 2 urinary stones with
no twinkling sign were radiolucent on plain abdominal films.
COMMENTS
Sonographic
detection of urinary stones is relative easy for stones with both distinct
echogenicity and posterior acoustic shadowing (11). However, in many cases
it is difficult to determine whether a urinary stone is present because
of its indistinct echogenicity and indiscrete posterior acoustic shadowing.
Indistinct echogenicity of stones results from surrounding echogenic tissue,
such as prominent renal sinus fat, mesenteric fat, and bowel. When a renal
stone is poorly distinguished from echogenic renal sinus fat and has an
indiscrete posterior acoustic shadowing, it may be difficult to determine
its presence on US. In one prior study, three radiologists interpreted
31 ultrasonograms with a sensitivity of 81% and a specificity of 86% for
detecting renal stones (3).
The present study was performed to determine whether the color Doppler
twinkling sign might improve the sonographic diagnosis of urinary stones.
Almost all stones (97%; 75 of 77) showed a color Doppler twinkling sign
and 60 of 75 stones (80%) with twinkling signs had signals with strong
intensity. The conclusion of the present study is that the twinkling sign
has potential usefulness in clinical practice, especially to confirm the
presence of stones with indistinct echo difference and indistinct posterior
acoustical shadowing.
Rahmouni et al. found color Doppler twinkling artifacts originating from
parenchymal calcifications, including bladder calculi (4). They explained
that when the US beam is incident to a rough interface composed of sparse
reflectors, a twinkling sign is generated by the phase shifts resulting
from multiple reflections in the medium. Because urinary stones become
larger particles by aggregation or agglomeration of primary crystal forms,
they are predominantly composed of a highly reflecting crystalline aggregate
of varying chemical composition with a mucoproteinous organic matrix (12).
On the basis of the explanation of Rahmouni et al., the twinkling artifacts
from urinary stones are likely to be generated by random strong reflections
and multiple inner reflections of the incidental US beam at a rough interface
formed by a crystalline aggregate of stones (4). In a phantom study of
Lee et al., the twinkling sign originated from a fixed site of each stone
during repeated scanning (7). This finding indicates that the twinkling
sign is related to some structural factor in the stone.
Rahmouni et al. suggested that the artifact could be influenced by ultrasonic
beam attenuation of tissues interposed between the probe and a calcification
(4). Lee et al. reported that 4 of 20 renal stones and 2 of 16 ureteral
stones did not show any twinkling sign (7). They suggested that ureteral
stones may be influenced more than renal stones by ultrasonic attenuation
of interposed tissues because the ureter is deep-seated below abundant
fatty tissue without a proper acoustic window. In the present study only
two urinary stones showed no twinkling sign. Furthermore, Lee et al. reported
that they could not find any correlation between the location of stones
and the genesis of the signs, which is in line with our findings (7).
Our findings suggest, that color Doppler twinkling sign is more affected
by the architecture of the stones than by beam attenuation from interposed
tissue.
Lee et al. further reported that the location of focal zone can influence
the occurrence and intensity of the sign (7). When the focal zone was
placed below urinary stones, artificial color signal was prominent and
strengthened in our phantom study (results not presented). Our standardized
protocol placed the focal zone slightly below the area of interest. Using
this focal position we found a twinkling sign in almost all stones (75
of 77). Based on our results we support standardized US settings with
the focal depth slightly below the stone of interest.
Lee et al. noted that one of the limitations of their study was that they
did not determine whether the detection of the twinkling signs would actually
improve the detection of stones. In our study we evaluated the improvement
in the detection of urinary stones using the twinkling sign. With CT findings
as our “gold standard”, we were able to improve significantly
the detection of urinary stones with twinkling sign as compared with gray-
scale US (p < 0.005). Only two stones (= 0.4 cm) did not show a twinkling
sign and these stones were not visible on gray-scale US. Both stones were
radiolucent and the urine analysis was suggested uric acid stones. This
finding is in line with the finding of Chelfouh et al. who reported absence
of the twinkling sign from uric acid stones. Based upon our observations,
it is possible that the twinkling sign might be helpful in the differentiation
of urinary stone composition and morphology. However, this should be evaluated
in further studies.
Echogenic foci with color signs can be seen in the area of the renal sinus
and do not always suggest stones. Renal artery calcification should be
considered in the differential diagnosis, especially in patients with
long-standing diabetes, hypertension, or other systemic diseases associated
with atherosclerotic vascular disease (13). Real-time scanning can help
differentiate arterial calcifications from renal calculi because arterial
calcifications are seen to pulsate. However, twinkling sign may also develop
from calcifications of renal tumor, renal cyst, and renal parenchyma.
These calcifications usually can be differentiated from renal stones on
the basis of their location on real-time scanning and the patient’s
history.
We note several limitations of our study. The main limitation of the study
is that only 8% of the ureteral stones were located in the mid ureter.
Since it is known that this mid-ureteral stones are often the hardest
to be localized by sonography, the good results of stone detection by
sonography may differ, when more mid ureteral stones would have been included.
Further, the US examination was interpreted by conserves review of two
radiologists. We have no data about intra- and interobserver-variability.
Second, we used only one US unit (Acuson Sequoia 512). As reported by
Aytac et al. the twinkling sign depends on the color sensitivity and the
acoustic output of the US unit (6). Therefore, with different US units
these results might be not reproducible. In spite of these limitations,
we conclude that the color Doppler twinkling sign was frequent and characteristic
enough to provide a useful additional finding for urinary stones on US.
CONCLUSIONS
The color Doppler twinkling sign improves the detection and confidence of diagnosis for renal and ureteral stones with minimal loss of specificity.
CONFLICT OF INTEREST
None declared.
REFERENCES
- McConnell JD: Ultrasonography of the kidney. Semin Urol. 1994; 12: 333-40.
- King W 3rd, Kimme-Smith C, Winter J: Renal stone shadowing: an investigation of contributing factors. Radiology. 1985; 154: 191-6.
- Kimme-Smith C, Perrella RR, Kaveggia LP, Cochran S, Grant EG: Detection of renal stones with real-time sonography: effect of transducers and scanning parameters. AJR Am J Roentgenol. 1991; 157: 975-80.
- Rahmouni A, Bargoin R, Herment A, Bargoin N, Vasile N: Color Doppler twinkling artifact in hyperechoic regions. Radiology. 1996; 199: 269-71.
- Chelfouh N, Grenier N, Higueret D, Trillaud H, Levantal O, Pariente JL, et al.: Characterization of urinary calculi: in vitro study of “twinkling artifact” revealed by color-flow sonography. AJR Am J Roentgenol. 1998; 171: 1055-60.
- Aytaç SK, Ozcan H: Effect of color Doppler system on the twinkling sign associated with urinary tract calculi. J Clin Ultrasound. 1999; 27: 433-9.
- Lee JY, Kim SH, Cho JY, Han D: Color and power Doppler twinkling artifacts from urinary stones: clinical observations and phantom studies. AJR Am J Roentgenol. 2001; 176: 1441-5.
- Sheafor DH, Hertzberg BS, Freed KS, Carroll BA, Keogan MT, Paulson EK, et al.: Nonenhanced helical CT and US in the emergency evaluation of patients with renal colic: prospective comparison. Radiology. 2000; 217: 792-7.
- Obuchowski NA: Nonparametric analysis of clustered ROC curve data. Biometrics. 1997; 53: 567-78.
- Altmann D: Practical statistics for medical research. London, Chapman and Hall. 1991; pp. 416-8.
- Middleton WD, Dodds WJ, Lawson TL, Foley WD: Renal calculi: sensitivity for detection with US. Radiology. 1988; 167: 239-44.
- Segal AB: MP. Radiological characteristics of urolithiasis. In: Pollack HM, (ed.), Clinical urography. Philadelphia, WB Saunders. 1990; pp. 1758.
- Kane RA, Manco LG: Renal arterial calcification simulating nephrolithiasis on sonography. AJR Am J Roentgenol. 1983; 140: 101-4.
____________________
Accepted
after revision:
May 11, 2009
_______________________
Correspondence
address:
Dr. Michael Mitterberger
Department of Urology
University Hospital of Innsbruck
Anichstrasse 35
6020, Innsbruck, Austria
Fax: + 0043 512 504-24873
E-mail: Michael.Mitterberger@i-med.ac.at
EDITORIAL COMMENT
This is an interesting study with some provocative findings, which should stimulate further research in this diagnostic area. The main limitations of the findings, partly pointed out by the authors, are related to the issues of variability (inter and intra-observer), scanning machine, experience of the observer, availability and comparability in the emergency out-of-hours setting, and the lack of sufficient numbers of patients with stones in the mid-ureter, or distal ureter above the ureterovesical junction and below the iliac vessels, as these are often the hardest to visualize and often confused with pelvic phleboliths. The fact that the color-Doppler studies were always performed after the normal sonography added an element of observer verification bias. The authors also failed to state if there were any differences between color and power Doppler. However, there is sufficient encouragement from the authors’ findings to stimulate a multicenter larger double-blinded study with randomization and some negative controls, to define the true diagnostic role of this technique in wider practice, as ultimately, ultrasound is a more observer dependent technique than non-contrast computed tomography.
Dr.
Anup Patel
Department of Urology
St. Mary’s Hospital
Imperial College School of Medicine
London, United Kingdom
E-mail: anup.patel@imperial.ac.uk
REPLY BY THE AUTHORS
For the
diagnostic imaging of patients with suspected urinary tract stones, excretory
urography has been the gold standard. In recent years unenhanced helical
computed tomography (CT) has been introduced as a quick alternative. Additional
information regarding renal function may be obtained by combining CT with
contrast infusion. As an alternative a plain film of the kidneys, ureters
and bladder (KUB) combined with ultrasonography (US) can be used (1).
The major disadvantage of X-ray and CT imaging is the high radiation doses
given to the patient during the investigation. In the United States, the
number of CT scans has been quadrupled since 1992. Physicians are referring
their patients for so many imaging tests that as many as 2% of cancers
may be attributable to radiation exposure during CT scanning (2). The
cumulative effective doses of radiation from imaging procedures increased
with advancing age. In a recent paper about low-dose ionizing radiation
exposure from medical imaging procedures, Fazel et al. concluded that
the current pattern of use of medical imaging in the United States is
exposing many patients to substantial doses of ionizing radiation (3).
Therefore, strategies for optimizing and ensuring appropriate use of these
procedures in the general population should be developed.
In our opinion, there is an urgent need to reduce radiation exposure in
imaging, especially for repeated or redundant imaging like in patients
with urinary stones. US has the advantage of being universally available,
does not expose the patient to radiation, requires no intravenous contrast
medium, and is independent of the kidney function. The present study shows,
that the twinkling sign using the color Doppler US improves the detection
of urinary stones in comparison with gray-scale US and almost achieves
the same results as unenhanced CT. Therefore, in our opinion color Doppler
US with the twinkling sign can become a main alternative diagnostic imaging
tool for patients with suspected urinary stones or for follow-up and may
replace more expensive, invasive or harmful imaging procedures.
REFERENCES
1. Preminger
GM, Tiselius HG, Assimos DG, Alken P, Buck AC, Gallucci M, et al.: Guideline
for the management of ureteral calculi. Eur Urol 2007; 52: 1610-31.
2. Lauer MS: Elements of danger - the case of medical imaging. N Engl
J Med. 2009; 361: 841-3.
3. Fazel R, Krumholz HM, Wang Y, Ross JS, Chen J, Ting HH, et al.: Exposure
to low-dose ionizing radiation from medical imaging procedures. N Engl
J Med. 2009; 361: 849-57.
The
Authors