ARE
THERE DIFFERENCES IN ZONAL DISTRIBUTION AND TUMOR VOLUME OF PROSTATE CANCER
IN PATIENTS WITH A POSITIVE FAMILY HISTORY?
(
Download pdf )
doi: 10.1590/S1677-55382010000500007
Clinical UrologyWADE J. SEXTON, PHILIPPE E. SPIESS, LOUIS L. PISTERS, SCOTT CARPENTER, LYDIA T. MADSEN, ROBIN ZAGONE, XUEMEI WANG, PATRICIA TRONCOSO
Genitourinary Oncology Program (WJS, PES), H. Lee Moffitt Cancer Center, Tampa, Florida, USA and Departments of Urology (LLP, SC, LTM, RZ), Quantitative Sciences (XW), and Pathology (PT), The University of Texas M. D. Anderson Cancer Center, Houston, Texas, USA
ABSTRACT
Purpose:
To determine if there are any differences in the zonal distribution and
tumor volumes of familial and sporadic prostate cancers (PC) in men undergoing
radical prostatectomy.
Material and Methods: 839 patients underwent
a radical prostatectomy in the absence of prior neoadjuvant therapy between
1987 and 1996. Telephone interviews were conducted to obtain an updated
family history. A positive family history was defined as the diagnosis
of PC in at least one first degree relative. Prostatectomy specimens were
examined to determine the number of tumor foci, zonal origin of the dominant
tumor focus, tumor volume of the largest cancer focus, total tumor volume,
Gleason score and stage, and the surgical margin status. Results were
stratified according to family history and ethnicity.
Results: We successfully contacted 437 patients
(52%). Prostatectomy specimens from 55 patients were excluded from review
due to a history of prior transurethral resection of the prostate
(n = 26) or uncertain pathological stage (n = 29). Of the remaining 382
patients, 76 (20%) reported having a first-degree relative with PC. Statistical
analysis revealed no significant differences in the pathologic variables
between the two groups of patients with or without a family history of
PC.
Conclusions: Familial and sporadic PC share
similar characteristics. No histopathological differences account for
the increased positive predictive value of PC screening tests among patients
with a family history of PC.
Key
words: prostatic neoplasms; tumor burden; family characteristics;
tumor volume; pathology
Int Braz J Urol. 2010; 36: 571-82
INTRODUCTION
The reported
rate of positive family history among men with prostate cancer (PC) ranges
from 13% to 26% and approaches 40% for men diagnosed with the disease
before age 55 (1-3). Detection of cancer in multiple first-degree or second-degree
relatives coupled with early age at cancer diagnosis substantially increases
a man’s relative risk and absolute risk of developing PC (4).
Whereas a number of earlier studies evaluated family history and radical
prostatectomy (RP) pathology to determine the significance of a positive
family history as a risk factor for PC, they were directed largely at
the number of tumors, the pathologic stage, and outcome measures including
biochemical disease-free survival (DFS) and overall survival (5-9). Very
little information is available on the zonal distribution of cancers in
patients with a positive family history. Furthermore, there are no pathologic
data that explain the increased positive-predictive value reported for
abnormal digital rectal examination (DRE) and prostate-specific antigen
(PSA) tests in high-risk patients, such as those with a positive family
history (10,11). We as well do not currently know if there exist differences
in terms of tumor volumes between patients with and without a family history
of PC?
We hypothesized that RP specimens from patients with a family history
of PC might reveal different tumor characteristics compared with specimens
from patients with sporadic PC and that the different characteristics
could account for the higher diagnostic yield of prostate biopsies performed
in patients with risk factors for disease. To address this hypothesis,
we specifically examined the zonal distribution of cancer foci, tumor
location, and tumor volume differences of our surgically managed patients
with and without a family history of PC. We also analyzed corresponding
clinicopathologic data, such as age, preoperative PSA levels, pathologic
stage, Gleason score, and surgical margin status in patient groups stratified
by family history.
MATERIALS AND METHODS
A research
protocol was designed and approved by our Institutional Review Board prior
to the initiation of this study. From January 1987 to December 1996, 839
patients underwent RP at The University of Texas M. D. Anderson Cancer
Center, with no prior neoadjuvant therapy. The patients were followed
for at least 6 years from the time of RP. Family history data were obtained
by telephone interview to update the patient’s file and to capture
additional family members affected with PC since the index patient’s
surgical procedure. The patients and immediate family members who were
successfully contacted were included in the study. Positive family history
was defined as having at least 1 first-degree relative (e.g., brother,
son, father) who had a diagnosis of PC. Only a few patients with possible
hereditary prostate cancer (HPC) were included in the analysis. HPC is
believed to be transmitted in an autosomal dominant fashion, and it is
postulated that HPC accounts for 5% to 10% of all cases of PC (and possibly
30% to 45% of early onset cancers). To be classified as having HPC, patients
had to fulfill one of the following clinical criteria: PC occurring in
3 successive generations (maternal or paternal side), 3 cases of PC diagnosed
within a nuclear family, or 2 men within a single family diagnosed with
PC before age 55 (4,12).
The prostatectomy specimens were evaluated as previously reported (13,14).
Each tumor focus was outlined on the histologic sections, and the total
number of tumor foci as well as the zonal origin of each tumor focus were
recorded. Zones were categorized as peripheral zone (PZ), transition zone
(TZ), or multiple zones (MZ). An area of carcinoma was considered a different
focus if it was separated by a low-power field diameter (4.5 mm) from
the nearest adjacent focus, as reported previously (15). Tumor location
was recorded according to standardized guidelines (Figure-1). Each tumor
focus was graded according to the Gleason grading system (16). The Gleason
score assigned to the specimen was that of the tumor focus with the highest
Gleason score. The volume of each tumor focus was calculated according
to the three-dimensional volume estimation method (17). The 2002 American
Joint Committee on Cancer tumor-node-metastasis staging system was used
to define organ-confined cancers (T2), established extraprostatic extension
(T3a), and cancer involvement of the seminal vesicles (T3b). Patients
with positive margins without extraprostatic extension were categorized
as having T2 tumors regardless of margin status.
Patients’ demographic and clinical characteristics were summarized
by family history status. Fisher’s exact test was performed to assess
the association between categorical variables. The Wilcoxon rank sum test
was performed to assess the difference in continuous variables between
patients with and those without a family history of PC. In order to assess
the predictive effect of family history and ethnicity simultaneously,
a multinomial logistic regression model was fit for the outcome variable
of “site of dominant tumor focus”, which can take the values
of “PZ”, “TZ”, or “MZ”. The fitted
multinomial logistic regression model can be written as follows:
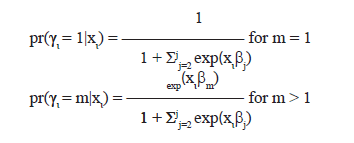
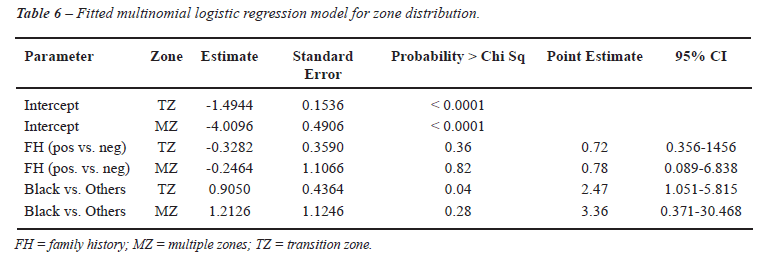
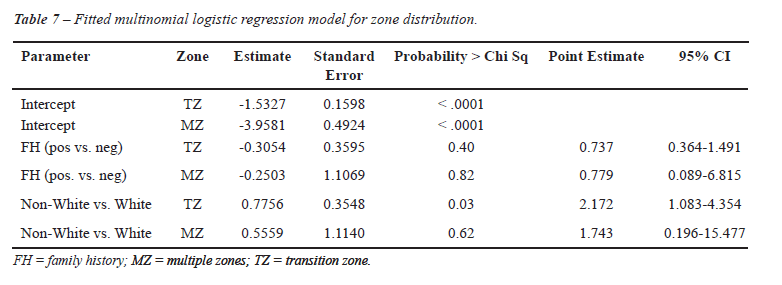
where yi is the response variable for subject i, and can take values of 1, 2, …, m. In our case, the site of dominant tumor focus could be “P”, “T” or “M”; therefore, m = 3. Xi is vector of covariates for subject i (here, are family history and ethnicity) and ßj is the vector of coefficient, with j ranges from 2 to J. Note: J = m. All statistical analyses were carried out in SAS or Splus software program.
RESULTS
Patient Characteristics
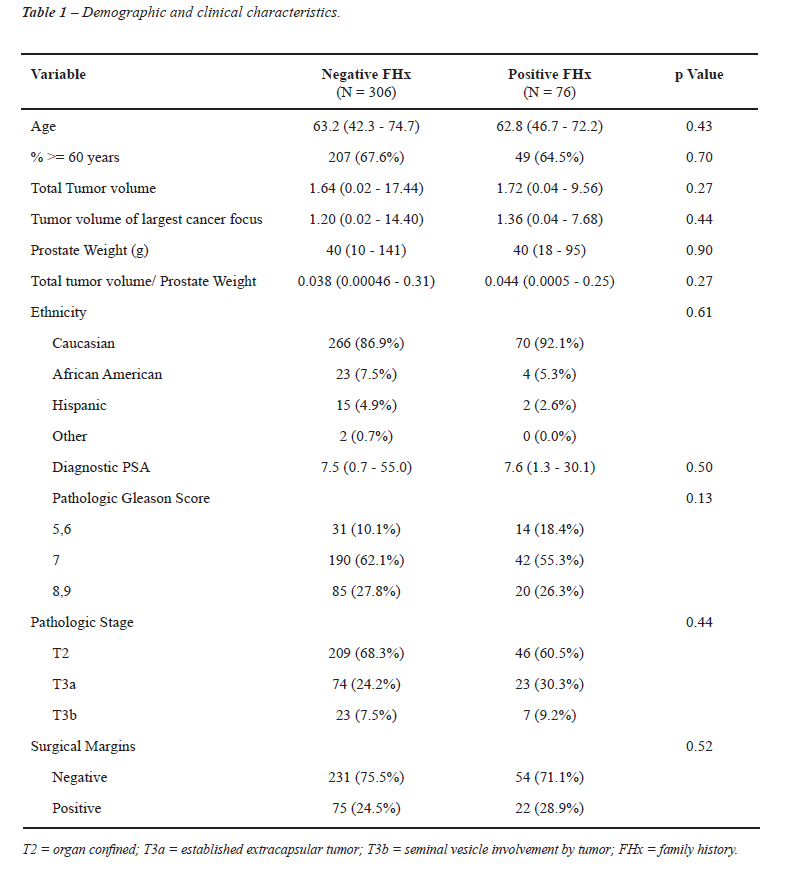
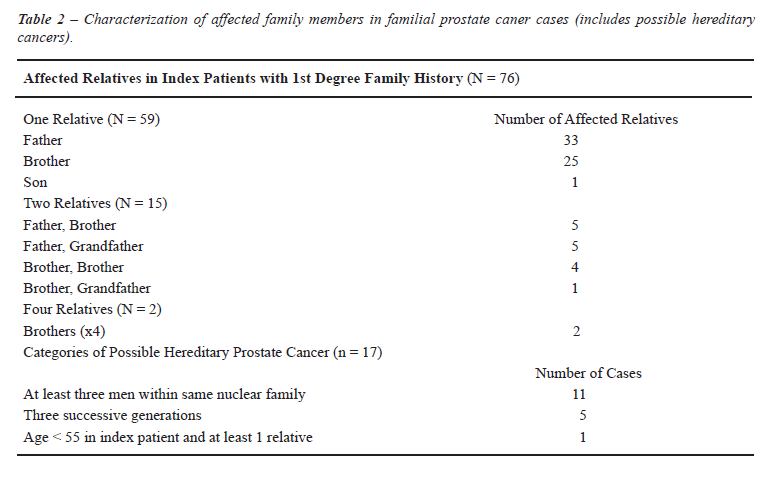
We successfully contacted 437 (52%) patients. Specimens from 55 patients were excluded from review due to the patient’s history of prior transurethral prostatectomy (n = 26), uncertain or missing pathologic data (n = 29), leaving 382 patients with complete clinical and pathologic data. Seventeen patients (4%) were believed to have HPC on the basis of the aforementioned clinical criteria. Table-1 lists clinical information and demographics of the patients with and without a family history of PC. There was no difference in any of the clinical or pathologic features including total tumor volume and tumor volume of the largest tumor focus among patients with and without a family history of PC. Table-2 characterizes the affected family members in familial prostate cancer and hereditary prostate cancer cases.
Association between Family History, Ethnicity, and Pathologic Features
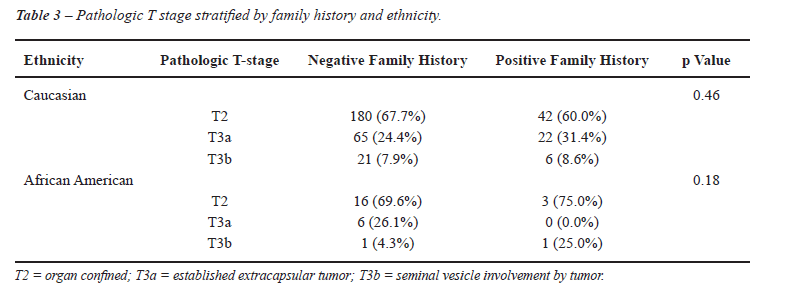
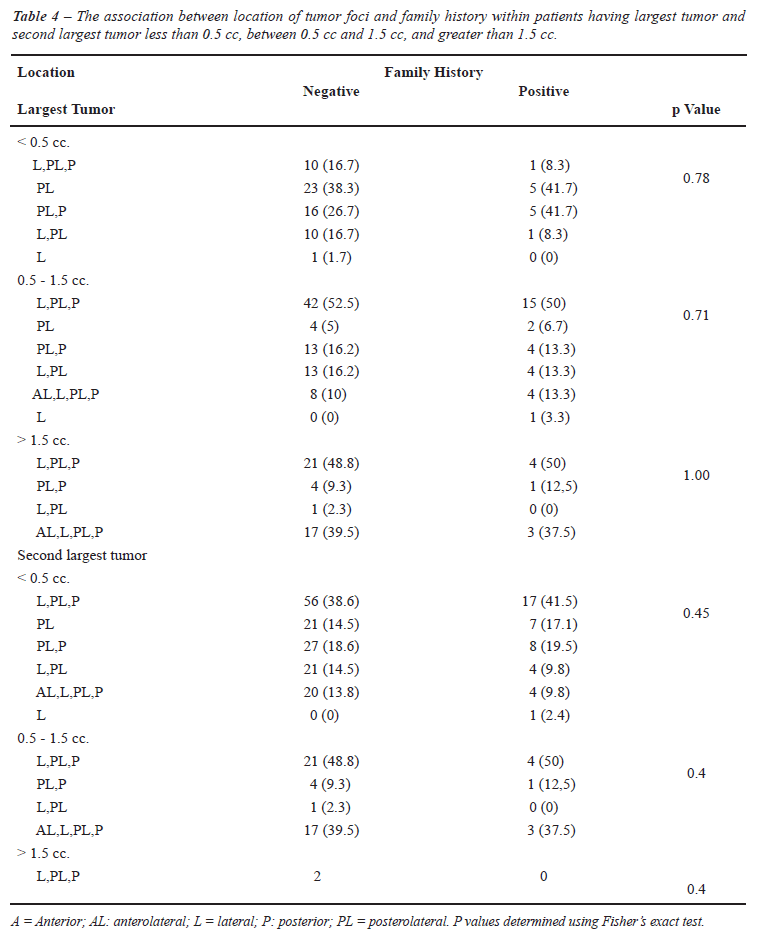
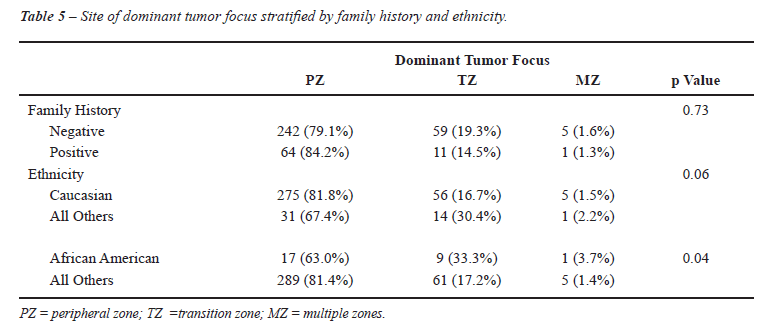
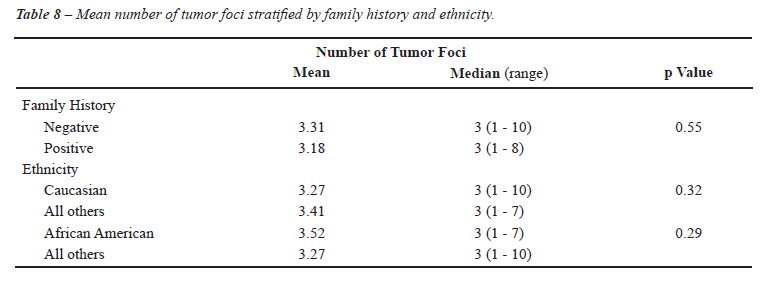
The association between family history of PC and pathologic stage was evaluated in Caucasian and other ethnicities (Table-3). No significant association was detected in either Caucasian (p = 0.46) or African American patients (p = 0.18). To further clarify the potential association between location of the largest cancer foci and ethnicity, we assessed the location of cancer foci by familial history of prostate cancer (Table-4). We were unable to show an association between the location of the largest cancer focus and familial history of prostate cancer, with substratification of the volume of the largest cancer focus (< 0.5 cc, 0.5 to 1.5 cc, and > 1.5 cc). Similarly, we attempted to determine if there was an association between the location of the second largest cancer focus and family history of prostate cancer (Table-4), with no statistically signification association noted. Table-5 shows the location of the dominant tumor focus according to the patient’s reported family history and race. Positive family history of PC did not significantly impact (p = 0.73) the dominant tumor focus. On multinomial logistic regression analysis, the lack of association between ethnicity and site of dominant tumor focus was preserved (Tables 6 and 7). Furthermore, we compared the number of tumor foci based on either family history or ethnicity (Table-8). The mean and median number of tumor foci did not show a significant difference based on family history of PC or ethnicity.
COMMENTS
Most physicians
recommend early screening for PC in populations at high risk for the disease.
High-risk populations include men with a positive family history of PC
and African American men (18). The risk is even greater for men with multiple
risk factors or for those whose affected relatives were diagnosed with
cancer at younger ages (less than 55-60 years of age) (4).
Several studies have sought the patterns of RP pathology that could be
unique to patients with a family history of PC. Studies have focused on
the number of tumors, Gleason score, pathologic stage, and biochemical
DFS and cancer-specific survival rates (1,5-9). However, there is very
little information about the zonal distribution of prostate cancer in
patients with a positive family history. Similarly, tumor volume has been
shown to be an independent predictor of biochemical failure yet there
is little to no information on the association between either tumor volume
(either total or of the dominant focus of prostate cancer) among patients
with or without a family history of PC (13,19).
Prostate cancer is characterized as a multifocal disease, and the majority
of cancers are located in the PZ area most commonly targeted and sampled
during prostate biopsies. Catalona et al. (10) reported that there is
a higher positive predictive value of PC screening tests (PSA and DRE)
for detecting PC in patients with established risk factors such as family
history and African American ethnicity while controlling for both age
and PSA. It seems intuitive that prostatectomy specimens from patients
with risk factors for disease might contain more dominant tumor foci located
peripherally in targeted biopsy zones, a greater number of tumor foci,
or a greater percentage of cancer per prostate specimen to account for
the significantly higher yield of prostate biopsies. However, our pathology
results do not support the hypothesis that differences in the zonal origin
of the dominant tumor focus or differences in the number of tumor foci
could account for higher cancer detection rates in patients with PC risk
factors. There were no significant differences in the mean number of tumor
foci based on family history or on ethnicity. Most dominant tumor foci
were located peripherally and family history did not alter the distribution
of the dominant tumor foci between the PZ, TZ, or MZ. Contrary to family
history status, when the locations of the dominant tumor foci were stratified
according to ethnicity, non-Caucasians were demonstrated to have more
dominant tumor foci originating within the transition zone compared to
Caucasian patients.
In an attempt to clarify if the location of the largest and second largest
tumor foci were different based on family history of PC, we were unable
to identify an association between site of these predominant tumor foci
and familial history of PC. This may have had important clinical implications
in terms of the transrectal ultrasound guided prostatic biopsy schemes
to apply to patients based on familial history of PC. However, due to
the lack of an association between the site of the largest and second
largest tumor focus and family history of PC, an extensive biopsy scheme
consisting of 10 to 12 biopsy cores should be obtained in all patients
in whom prostatic biopsies are clinically indicated.
Racial variation in the location of dominant tumor foci has been reported
previously. Pettaway and colleagues found that 35% of all tumor foci in
African American men were of TZ origin compared with 21% of the tumor
foci in a group of Caucasian patients matched according to pathologic
stage. In this same study, the median number of tumor foci and the overall
prostatic tumor volumes (stratified by pathologic stage and zone of origin
of the cancers) were not statistically different (20). Pettaway et al.
hypothesized that the TZ epithelium in African American patients might
be more susceptible to malignant transformation compared with the TZ epithelium
of Caucasian men (20).
A smaller study failed to reveal significant differences in tumor multifocality
and the zonal distribution of tumor foci in patients with a positive family
history. Bastacky et al. (21) examined the morphologic characteristics
of 81 radical prostatectomy specimens from selected small groups of patients
with sporadic PC, familial PC, and HPC. In this study, patients with sporadic
PC had a mean 7.3 tumor foci per gland compared with a mean 5.2 tumor
foci per gland in the familial PC group. On the contrary, patients with
familial PC had a larger volume of cancer per gland compared with the
sporadic PC group (3.3 cm3 vs. 2.4 cm3). However, these results failed
to reach statistical significance likely due to the small number of patients
in each of the three groups.
Although patients with a positive family history of PC are at higher risk
of developing the disease, most studies suggest that these patients have
either equivalent (12) or more favorable disease characteristics, such
as lower Gleason scores (1,7,21) or less extracapsular tumor extension
(9). Furthermore, there is no convincing evidence that survival is better
or worse for patients with a positive family history of PC undergoing
RP (5,8,9) or primary radiation therapy (22) compared to patients with
sporadic PC. Surprisingly, only one group reported differences in biochemical
DFS following RP or primary radiation therapy favoring patients with sporadic
PC. Even in this study, there were no clinical or pathologic differences
to account for the inferior biochemical outcome for patients with a positive
family history (6). Regardless of the disease outcome, no study to date
(including our report) has been able to demonstrate different pathologic
or morphologic characteristics in prostate specimens from patients with
a positive family history compared with patients without a family history
of PC that would account for an approximately 10% increase in the positive
predictive value of current diagnostic screening tests (PSA and DRE) on
PC detection (10,11,23). Consistent with previous reports, our study revealed
no significant differences between patients with a family history of PC
compared to patients without a family history of PC in terms of age, preoperative
PSA, Gleason score, pathologic stage, and surgical margin status. We did
not examine disease-specific or biochemical DFS. Based on the pathologic
results from both patient groups, it is unlikely that there would be meaningful
differences in these outcome measures.
Several potential biases with this study deserve mention. Although our
data were stratified according to ethnicity, few non-Caucasian patients
were included in the overall analysis. Patients included in this study
all underwent RP. Thus, our population may not represent the full spectrum
of familial and sporadic PC. Other potential risk factors, such as obesity
and diet, may significantly affect cancer initiation, promotion, and progression,
and were not accounted for in this analysis (24,25). Regarding family
history, we did not confirm reported positive family histories with pathology
reports of affected relatives. Neither were we able to accurately determine
the exact age of most of the affected family members at the time of their
own diagnosis. However, recognizing some of these potential biases at
study onset, we did not rely on medical record family history data for
study inclusion, thus excluding nearly half of the patients undergoing
RP during the study period. We attempted to clarify and update family
history data via telephone interviews with the patients or with their
immediate family members. However, an accurate assessment of the significance
of family history related to the development of PC will be limited as
long as the definition of a positive family history is based on clinical
criteria and not on molecular fingerprints or biomarkers that might establish
a true familial link to PC among affected individuals.
CONCLUSIONS
Patients
with familial and sporadic PC have pathologically and morphologically
similar tumors. In our study group, there were no clinically significant
differences in the zonal origin and location of the dominant tumor foci,
tumor volume, or number of tumor foci to account for the reported increased
positive predictive value of diagnostic screening tests in patients with
a family history of PC.
Acknowledgements
We thank Cindy Soto BS, HT (ASCP) and Ina Prokhorova, MD, HT, HTL (ASCP) for expert technical assistance with the pathologic specimens, Brooke Reynolds for assistance in preparation of the manuscript, and Kim Vu for assistance in preparation of the illustrations.
CONFLICT OF INTEREST
None declared.
REFERENCES
- Keetch DW, Humphrey PA, Smith DS, Stahl D, Catalona WJ: Clinical and pathological features of hereditary prostate cancer. J Urol. 1996; 155: 1841-3.
- Bratt O, Kristoffersson U, Lundgren R, Olsson H: Familial and hereditary prostate cancer in southern Sweden. A population-based case-control study. Eur J Cancer. 1999; 35: 272-7.
- Carter BS, Beaty TH, Steinberg GD, Childs B, Walsh PC: Mendelian inheritance of familial prostate cancer. Proc Natl Acad Sci U S A. 1992; 89: 3367-71.
- Bratt O: Hereditary prostate cancer: clinical aspects. J Urol. 2002; 168: 906-13.
- Azzouzi AR, Valeri A, Cormier L, Fournier G, Mangin P, Cussenot O: Familial prostate cancer cases before and after radical prostatectomy do not show any aggressiveness compared with sporadic cases. Urology. 2003; 61: 1193-7.
- Kupelian PA, Kupelian VA, Witte JS, Macklis R, Klein EA: Family history of prostate cancer in patients with localized prostate cancer: an independent predictor of treatment outcome. J Clin Oncol. 1997; 15: 1478-80.
- Kotsis SV, Spencer SL, Peyser PA, Montie JE, Cooney KA: Early onset prostate cancer: predictors of clinical grade. J Urol. 2002; 167: 1659-63.
- Grönberg H, Damber L, Tavelin B, Damber JE: No difference in survival between sporadic, familial and hereditary prostate cancer. Br J Urol. 1998; 82: 564-7.
- Norrish AE, McRae CU, Cohen RJ, Jackson RT: A population-based study of clinical and pathological prognostic characteristics of men with familial and sporadic prostate cancer. BJU Int. 1999; 84: 311-5.
- Catalona WJ, Antenor JA, Roehl KA, Moul JW. Screening for prostate cancer in high risk populations. J Urol. 2002; 168: 1980-3; discussion 1983-4.
- Aprikian AG, Bazinet M, Plante M, Meshref A, Trudel C, Aronson S, et al.: Family history and the risk of prostatic carcinoma in a high risk group of urological patients. J Urol. 1995; 154: 404-6.
- Carter BS, Bova GS, Beaty TH, Steinberg GD, Childs B, Isaacs WB, et al.: Hereditary prostate cancer: epidemiologic and clinical features. J Urol. 1993; 150: 797-802.
- Babaian RJ, Troncoso P, Steelhammer LC, Lloreta-Trull J, Ramirez EI: Tumor volume and prostate specific antigen: implications for early detection and defining a window of curability. J Urol. 1995; 154: 1808-12.
- Sanchez-Ortiz RF, Troncoso P, Babaian RJ, Lloreta J, Johnston DA, Pettaway CA: African-American men with nonpalpable prostate cancer exhibit greater tumor volume than matched white men. Cancer. 2006; 107: 75-82.
- Troncoso P, Babaian RJ, Ro JY, Grignon DJ, von Eschenbach AC, Ayala AG: Prostatic intraepithelial neoplasia and invasive prostatic adenocarcinoma in cystoprostatectomy specimens. Urology. 1989; 34(6 Suppl): 52-6.
- Gleason D: Histologic grading and clinical staging of prostate carcinoma. In: Tannenbaum M (ed.), Urologic Pathology. The Prostate. Philadelphia, Lea & Febiger. 1977: pp. 171-97.
- Chen ME, Johnston D, Reyes AO, Soto CP, Babaian RJ, Troncoso P: A streamlined three-dimensional volume estimation method accurately classifies prostate tumors by volume. Am J Surg Pathol. 2003; 27: 1291-301.
- Crawford ED: Epidemiology of prostate cancer. Urology. 2003; 62(6 Suppl1): 3-12.
- Nelson BA, Shappell SB, Chang SS, Wells N, Farnham SB, Smith JA Jr, et al.: Tumour volume is an independent predictor of prostate-specific antigen recurrence in patients undergoing radical prostatectomy for clinically localized prostate cancer. BJU Int. 2006; 97: 1169-72.
- Pettaway CA, Troncoso P, Ramirez EI, Johnston DA, Steelhammer L, Babaian RJ: Prostate specific antigen and pathological features of prostate cancer in black and white patients: a comparative study based on radical prostatectomy specimens. J Urol. 1998; 160: 437-42.
- Bastacky SI, Wojno KJ, Walsh PC, Carmichael MJ, Epstein JI: Pathological features of hereditary prostate cancer. J Urol. 1995; 153: 987-92.
- Ray ME, Dunn RL, Cooney KA, Sandler HM: Family history of prostate cancer and relapse after definitive external beam radiation therapy. Int J Radiat Oncol Biol Phys. 2003; 57: 371-6.
- Narod SA, Dupont A, Cusan L, Diamond P, Gomez JL, Suburu R, et al.: The impact of family history on early detection of prostate cancer. Nat Med. 1995; 1: 99-101.
- Amling CL, Riffenburgh RH, Sun L, Moul JW, Lance RS, Kusuda L, et al.: Soderdahl DW, Donahue TF, Foley JP, Chung AK, McLeod DG. Pathologic variables and recurrence rates as related to obesity and race in men with prostate cancer undergoing radical prostatectomy. J Clin Oncol. 2004; 22: 439-45.
- Rohrmann S, Roberts WW, Walsh PC, Platz EA: Family history of prostate cancer and obesity in relation to high-grade disease and extraprostatic extension in young men with prostate cancer. Prostate. 2003; 55: 140-6.
____________________
Accepted
after revision:
March 17, 2010
_______________________
Correspondence address:
Dr. Wade J. Sexton
Genitourinary Oncology Program
Moffitt Cancer Center
12902 Magnolia Drive
Tampa, Florida, 33612-9416, USA
Fax: + 1 81 3745-8494
E-mail: wade.sexton@Moffitt.org
EDITORIAL COMMENT
This study revealed no significant differences between patients with familial histories compared with the patients without familial histories of prostate cancer in relation their age, preoperative PSA, Gleason score, pathological stage, and surgical status of the margin. However, important data that could be answered is related to the disease-specific survival and biochemical disease-free survival. Otherwise, patients with familial PC had a larger volume of cancer per gland compared with the sporadic prostate cancer group - 3.3 cm3 vs. 2.4 cm3. This tumoral volume data justifies more attention for men with a familial history of prostate cancer, since the larger the tumoral volume, the larger the chances of biochemical recurrence. In cases of smaller tumoral volume (2.6 cc) normally the PSA is lower than 4.
REFERENCE
- Zhu H, Roehl KA, Antenor JA, Catalona WJ: Biopsy of men with PSA level of 2.6 to 4.0 ng/mL associated with favorable pathologic features and PSA progression rate: a preliminary analysis. Urology. 2005; 66: 547-51.
Dr.
Marcos F. Dall’Oglio
University of Sao Paulo, USP
Sao Paulo, Brazil
E-mail: marcosdallogliouro@terra.com.br
REPLY BY THE AUTHORS
We agree with the editorial comments by Dr. Dall’Oglio that it would be interesting to analyze any differences in the survival characteristics (biochemical-free survival and disease specific survival) of patients with sporadic prostate cancer compared to patients with a family history of prostate cancer. As discussed in the manuscript however, there is no convincing evidence to date that a positive family history impacts survival in those who have received therapy for clinically localized prostate cancer. Furthermore, we found no difference in tumor volume (p = 0.27) in patients with a positive family history of prostate cancer (1.72 cm3) vs. patients with sporadic prostate cancer (1.64 cm3) as demonstrated in Table 1. Bastacky and colleagues (reference #21 in the manuscript) found a non-statistically significant difference in the tumor volume of patients with familial prostate cancer compared to patients with sporadic prostate cancer (3.3 cm3 vs. 2.4 cm3).
The
Authors