RECTOURETHRAL
FISTULAS
(
Download pdf )
TOMÁS HANUS
Department of Urology, General Teaching Hospital, First Medical School, Charles University, Postgraduate School of Medicine, Prague, Czech Republic
ABSTRACT
A
recent review of the literature shows an increasing number of studies
on this subject, most of which are retrospective and presented solely
from a clinical point of view. Some of them are no more than case-reports.
There is a lack of valid epidemiologic data about the incidence of rectourethral
fistulas. According to the literature, these fistulas result from many
different causes.
Rectourethral fistulas may have a congenital
or acquired origin. Acquired cases include inflammatory, neoplastic, or
traumatic etiologies. The diagnostic algorithm is clear and very classical.
The new diagnostic approach represents an MRI investigation of the male
urethra. It is important to recognize this diversity of etiology because
each type requires a different surgical strategy. The aim of the surgical
approach is the closure of all types of fistulas. Spontaneous closure
of the recto-urethral fistulas after double diversion or by means of a
one-stage procedure is possible only in a few cases. In most cases, the
treatment proceeds in three stages (double diversion–urinary and
bowel, closure technique, undiversion). An endoscopic approach using biological
sealants seems to be promising. Few urologists and general surgeons have
attained wide experience in the management of rectourethral fistulas.
No single procedure has been proven most effective or even universally
applicable.
Key words:
urethra; rectum; abnormalities; fistula; reconstructive surgical procedures
Int Braz J Urol. 2002; 28: 338-45
CLASSIFICATION OF RECTOURETHRAL FISTULAS
Culp & Calhoon (1) described five basic groups of rectourethral fistulas according to the etiology: congenital, iatrogenic, traumatic, neoplastic, inflammatory.
CONGENITAL RECTOURETHRAL FISTULAS
Endo
et al. (2) described the results of a group study of the Japanese Study
Group of Anorectal Anomalies (JSGA) to determine the relative incidence
of specific types of anorectal anomaly in Japan. This study also discusses
the relationship between the levels of the fistula and blind end of the
rectum, low type deformity, rare types, and associated anomalies. A total
of 1,992 patients (1,183 boys and 809 girls), registered during the period
of 1976 to 1995, were analyzed according to the pathogenesis of anorectal
malformation in the field of molecular genetics. Rectovesical fistula
and complete covered anus were not infrequent deformities in this series.
It is suggested that at least 20% of rectourethral fistula should be categorized
as intermediate or low deformity from the viewpoint of the position of
the rectal pouch. A significant preponderance of Down’s syndrome
in cases of deformities without fistula suggests that further investigation
of associated anomalies in comparison with other congenital diseases may
provide insights.
The purpose of Rintala’s study (3)
was to compare the long-term outcome of sacroperineal-sacroabdominoperineal
pull-through to that of posterior sagittal anorectoplasty). In boys with
high anorectal anomalies, posterior sagittal anorectoplasty is clearly
superior to sacroperineal and sacro-abdominoperineal pull-through in terms
of long-term bowel function and fecal continence.
ACQUIRED RECTOURETHRAL FISTULAS
In
1972, Smith & Venema (4) reported their 20-year experience with 160
patients who had undergone radical retropubic prostatectomy (RRP) with
an incidence of 15 rectal injuries. Only 4 fistulas developed in this
group.
The most common single causes of rectourethral
fistulas in the series of 23 male patients published by Tiptaft et al.
(5) were fracture of the pelvis and iatrogenic causes (including 2 cases
after transurethral prostatic surgery, 2 cases after open prostatectomy
and 3 cases after urethral instrumentation).
Noldus et al. (6) reported 23 rectal injuries
(3.9%) during 589 RRP and cystoprostatectomy (CP).
Eastham & Scardino (7) summarized the
incidence of rectal injury during RRP in 3834 patients as 0.7% in average
(range 0.2 – 2.9%). Incidence of urethrorectal fistula, as an immediate
perioperative complication of open perineal prostate surgery, is 1.4%
(8).
Nyam et al. reviewed records of all patients
who had rectourethral fistula diagnosed between January 1981 and December
1995. Of the 16 male patients identified, only, all but three had had
intervention for their prostatic malignancy performed elsewhere. All patients
were interviewed by telephone for follow-up. The average age of those
sixteen patients was 68 years. The follow-up covered 80 months in general.
Adenocarcinoma of the prostate in 15 patients and recurrent transitional
cell epithelioma of the bladder in one patient were the underlying malignant
diseases. Seven patients had a radical retropubic prostatectomy, two had
radical retropubic prostatectomy after radiation, two had brachytherapy,
and three were treated by a combination of radiation and brachytherapy.
One patient formed a fistula after cystectomy and dilation of a stricture.
This heterogeneous group of patients received multiple therapies including
initial colostomy (7 patients), transanal repair (2 patients), parasacral
repair (2 patients), transperineal repair (2 patients), colo-anal anastomosis
(3 patients), and muscle transposition (3 patients). Four of those patients
required a permanent stoma.
Zippe (9) reviewed preliminary results on
prostate cryosurgery and reported a 2% to 5% incidence of rectourethral
fistulas. Complications of transrectal ultrasound - guided radical prostate
cryosurgery ought to be lower. Porter (10) mentions no urethroperineal
fistula and only 2.5% of urethrorectal fistula in 210 patients.
Montorsi et al. (11) reported one complication
of prostatorectal fistula after transrectal prostatic hyperthermia (430C)
in patients with advanced prostatic cancer (ten sessions of 60 min each).
Fistula was cured by leaving a urethral catheter in place for one month.
Kleinberg et al. (12) summarizes the results
of thirty-one patients with stage T1 or T2 prostatic carcinoma following
computed tomography - guided transperineal (125) I implants with 47 mCi
as the median total activity used. Only one patient developed a prostate
- rectal fistula attended to by means of an ileal conduit.
Fengler & Abcarian (13) published the
cases of eight patients with recto-urinary fistula in the course of a
treatment for prostate cancer (3 fistulas after radiation therapy alone,
3 after prostatectomies and 2 after both surgery and radiation therapy.
Chang et al. (14) published a case of prostatic
malakoplakia masquerading as a rectal tumor due to formation of a fistulous
tract to the rectal muscular layers.
Cools et al. (15) reported a very uncommon
fistula between the large bowel and the prostatic urethra due to Crohn’s
disease. Felipetto (16) had to treat prostatic cutaneous fistula as a
complication of Pseudomonas prostatitis.
DIAGNOSTICS OF RECTOURETHRAL FISTULAS
Rectourethral fistulas may be strongly suspected from the patient’s history (fecaluria, abnormal urethral discharge, pneumaturia, leakage of urine from the rectum during micturition). The most important diagnostic steps are rectal examination, proctoscopy, careful urethroscopy, intraurethral injection of methylene blue dye, radiopaque contrast agent placed into the bladder and then voided which usually appears in the rectum on X-ray (17-19).
TREATMENT OF RECTOURETHRAL FISTULAS
In
many cases, small fistulas will heal “spontaneously” with urinary
and/or fecal diversion. Therefore, an initial trial of conservative therapy
is reasonable. Select patients with chronic fistulas who are poor surgical
candidates may be treated conservatively with antibiotics, pads and symptomatic
care. Timing of repair is often individualized -mainly according to the
etiology, delay of diagnosis, size of fistula, the first or subsequent
repairs, the general condition of the patient, etc. Diversion of urine
(suprapubic cystostomy) is mandatory as well as correction of any stricture
of urethra distal to the fistula. Fecal diversion, temporary interruption
of the fecal stream-colostomy, is used by some as a mandatory part of
double diversion or selectively. Gibbons (20) requires a diverting end
- colostomy for 3-4 months. As surgeons became more experienced, bowel
preparations became standardized, and effective antibiotics were developed,
the enthusiasm for colostomy diminished. Currently, colostomy is recommended
when antibiotics alone cannot control the inflammation and infection associated
with the fistula or when the fistula involves radiated tissue. Low residue
diet contributes to healing. Suitable drainage (perineal, urethral splinting)
is very important. Two-layer suture of urethra and rectum with suture
lines at right angles and with interposition of soft tissue e.g. omentum
(21) or gracilis muscle-Ryan et al. (22) or scrotal flap (23) or using
other adequate approaches (transabdominal, transvesical, direct exposure
of rectourethral fistula through a transperineal incision, transanal,
transrectal trans-sphincteric approach or a flap technique with the intact
anal sphincter (5,24), or Latzko procedure (6).
Approaches to the urethrorectal fistula
repair are listed in Table-1.

Perineal Approach
In
1917, Young & Stone dissected the rectum away from sphincters, divided
the fistula, closed the urethra, and mobilized the rectum further cephalad
in such a way as to pull the affected rectum caudad out of the anus where
it was then transected and discarded, suturing the proximal rectum to
the anal skin. Lewis did not interpose sphincter between the repairs,
but did suture the elevator muscle fibers together in the anterior midline
when possible.
In 1958, Goodwin et al. (25) reported a
series of 22 rectourethral fistulas approached perineally. Extensive mobilization
of the rectum posteriorly and the bladder anteriorly through wide perineal
exposure allowed interposition of the elevator ani muscles between the
urinary tract and rectum.
Posterior-Sagittal Approach
In
1885, Kraske described a posterior midline incision extending to the left
paramedian aspect of the coccyx and sacrum that required partial sacrectomy
in addition to coccygectomy. His method did not involve division of the
sphincters, but rather sweeping the rectum laterally to ultimately accomplish
resection and reanastomosis of a tumor-bearing rectal segment, thereby
preserving fecal continence.
In 1962, Kilpatrick & Thompson used
this approach when the rectum was completely mobilized circumferentially
proximal and distal to the fistula. The rectourethral fistula was then
divided, sparing as much as possible on the urethral aspect. The rectal
opening was excised and closed in two layers; the urethra was repaired
and stented with a catheter.
Posterior
(Parasacrococcygeal)
Trans-sphincteric Approach
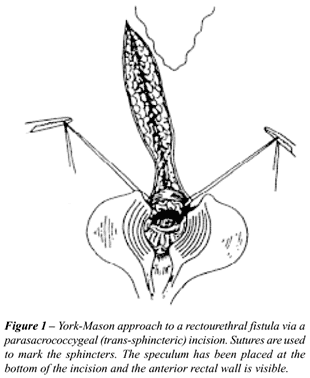
In
1969, Kilpatrick & Mason (26) improved this method and advocated a
more radical procedure of dividing all of the rectal sphincters to give
direct access to rectourethral fistula. The procedure (the so-called York
Mason approach) is simpler than some complicated transabdominal or transperineal
approaches to rectourethral fistula. It is still used because it allows
direct visualization of the fistula via parasacrococcygeal (trans-sphincteric)
incision especially to fistulae in the mid to lower rectum (13). After
skin incision, the mucocutaneous junction is marked with sutures and internal
sphincter is exposed. Division of the sphincter mechanism and posterior
rectal wall allows exposure of the fistula. Each sphincter muscle is tagged
with color-coded sutures. The next step of this procedure is the incision
around the fistula, followed by the excision of the fistulous tract, exposing
catheter in prostatic urethra. The undermining of rectal wall allows a
sufficient mobilization. After the closure of prostatic urethra, the full-thickness
rectal wall flaps sutured “vest over pants” technique is recommended.
It is important to note that the suture lines do not overlap each other.
The procedure is completed by suture of rectal wall and approximation
of sphincter muscle (Figure-1).
Fengler & Abcarian (13) succeeded in
healing rectourethral fistula in all of 8 patients when they adopted the
York Mason approach.
Bukowski et al. (27) treated 7 acquired
recurrent rectourethral fistulas (3 after prostatectomy, 3 after trauma
and one after perineal abscess) using the York Mason technique.
Fourniér et al. (28) reported a similar
success in the treatment of one case of urethro-prostate-rectal fistula
after gun wound.
Transanal Approach
Parks
& Motson (24) popularized the addition of a full thickness local flap
of anterior rectal wall as an aid to fistula repair through the intact
anal canal (Figure-2). They modified the transanal technique by denuding
the rectal mucosa lateral and distal to the fistula, thereby exposing
the circular rectal wall away from Denonvilliers´ fascia proximal
to the fistula for four centimeters. Tiptaft et al. (5) used a special
anal retractor for this surgery.
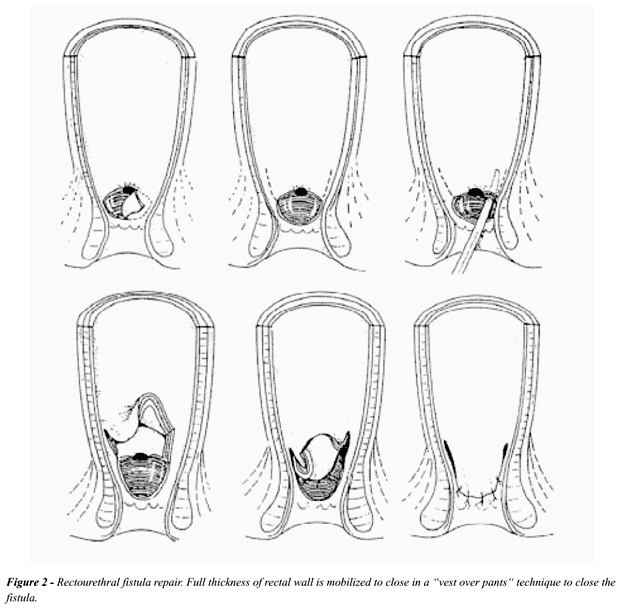
During the Latzko procedure, a fistula is
closed in three-layer fashion with absorbable suture. A transurethral
catheter is placed for 3 weeks. Noldus et al. (6) reported 23 patients
(3.9%) with rectal injury in a total of 589 RRP and CP. Of these 23 patients,
12 developed a rectourethral fistulas, whereas 7 fistulas closed spontaneously
by leaving the transurethral catheter in for a longer period. The remaining
5 fistulas were all successfully closed by the transanal Latzko procedure.
Anterior Transanorectal Approach
In 1973, Gecelter performed a midline perineal incision to gain access to the urinary tract after placing the patient in exaggerated lithotomy position. The sphincters were incised anteriorly, carefully placing tagging sutures, and the rectal incision was carried to the fistulous tract, which was excised and repaired in multiple layers with transposition of tissue as available.
Combined Approach
Al-Ali et al. (19) treated 30 men with rectourethral fistulas caused by war wounds. The method of posterior trans-sphincteric-anterior rectal wall advancement was the treatment of choice. Double diversion (end sigmoid colostomy and suprapubic cystostomy) were performed in all patients. Double diversion alone resulted in “spontaneous” rectourethral fistulas healing in 47% of the patients, but 53% of them required reconstruction. Early repair was recommended for large fibrous fistulas. The procedure was performed one month after double diversion. Undiversion was done after two months, when the urethra and anorectal canal were normal.
Endoscopic Approach
Wilbert
et al. reported 2 cases of rectourethral fistulas, which were repaired
endoscopically. The patients were positioned prone and rectoscope mounted
to the operating table was inserted into the rectum.The fistula was visualized
and the opening excised to the level of the perirectal tissues with cautery.
The rectal wall was mobilized full thickness with scissors and closed
primarily in two layers. The patients were then placed in lithotomy position
and the urethral side of the fistula was coagulated. Fibrin injection
can be applied if necessary.
In 2001, Bardari et al. (29) used biological glue (Glubran®) to close
one prostato-perineal fistula complicating a Miles abdomino-perineal resection
of rectum and one persistent neobladder-ileal fistula. The biologic sealant
was administrated endoscopically through an open-end ureteral catheter
6F.
Other Approaches
Youssef
et al. (30) successfully treated 12 male patients, ranging from 7 to 65
years old, who presented urethrorectal fistula from 1990 to 1997 using
the perineal subcutaneous dartos pedicled flap procedure. Urethrorectal
fistulas resulted from crush pelvic injury (6 cases) and gunshot (2 cases),
or were developed after prostatectomy (4 cases). The fistula was associated
with urethral stricture in 4 cases. A perineal approach was used in all
cases of urethrorectal fistula and combined with the trans-symphyseal
approach in the 4 patients with posterior urethral stricture. They interposed
a subcutaneous dartos pedicled flap as a vascularized tissue flap between
the repaired rectum and urethra. No leakage or perineal collection developed
and there was no fistula recurrence. Follow-up ranged from 9 to 42 months.
This technique of a perineal subcutaneous dartos pedicled flap may fulfill
the requirements for a successful repair of urethrorectal fistula.
Felipetto et al. (16) closed the prostatic
cutaneous fistula (as a complication of Pseudomonas prostatitis) with
human fibrin sealant (Tissucol).
In a prospective study, Venkatesh &
Ramanujam (31) discussed the efficacy of autologous fibrin glue application
in the healing or closure of recurrent anorectal fistulas. Autologous
cryoprecipitate was used as fibrin glue in all patients. This group included
complex anorectal fistulas, rectovaginal fistulas, and urethro-vesico-rectal
fistulas. Patients were given bowel preparation and intravenous antibiotics
before the procedure. Fistulas were curetted before injection of fibrin
glue. The success rate reached 60 percent. Patients with urinary tract
fistulas and acquired immunodeficiency syndrome failed to respond. No
major complications ensued. The results of fibrin glue application in
this small series of recurrent fistulas are inspiring. This fibrin glue
can be used as an outpatient procedure since it is a minimally invasive
technique, without any significant complications. It can be offered as
an alternative to conventional surgery in recurrent fistulas.
CONCLUSIONS
The possibility of euro-rectal fistula must be acknowledged. A careful history of the patient has to be taken into account. Standard urologic evaluation is mandatory. Proctoscopy, coloscopy and cystoscopy are inevitable investigations. There is a choice of surgery techniques for the closure of fistulas. Surgery of rectourethral fistulas is technically demanding and requires collaboration with rectal surgeons.
REFERENCES
- Culp OS, Calhoon HW: A variety of rectourethral fistulas: experiences with 20 cases. J Urol. 1964; 91: 560.
- Endo M, Hayashi A, Ishihara M, Maie M, Nagasaki A, Nishi T, Saeki M: Analysis of 1,992 patients with anorectal malformations over the past two decades in Japan. Steering Committee of Japanese Study Group of Anorectal Anomalies. J Pediatr Surg. 1999; 34: 435-41.
- Rintala RJ, Lindahl HG: Posterior sagittal anorectoplasty is superior to sacroperineal-sacroabdominoperineal pull-through: a long-term follow-up study in boys with high anorectal anomalies. J Pediatr Surg. 1999; 34: 334-7.
- Smith AM, Veenema RJ: Management of rectal injury and rectourethral fistulas following radical retropubic prostatectomy. J Urol. 1972; 108: 778.
- Tiptaft RC, Motson RW, Costello AJ, Paris AME, Blandy JP: Fistulae involving rectum and urethra: the place of Park‘s operations. Brit J Urol. 1983; 55: 711-5.
- Noldus J, Graefen M, Huland H: An “old technique” for a new approach for repair of rectourinary fistulas. J Urol. 1997, 157 (suppl. 4): 1547.
- Eastham JA, Scardino PT: Radical Prostatectomy. In: Walsh PC, Retik AB, Vaughan ED, Wein AJ (eds.). Campbell’s Urology. Philadelphia, WB Saunders Co., 7th ed.,1997, p. 2554.
- Thomas R, Davis R, Ahuja S: Toward out-patient radical prostatectomy: a cost effective cost management of patients with localized prostate cancer. Brit J Urol. 1997; 80 (suppl. 2): 261.
- Zippe CD: Cryosurgery of the prostate: technique and pitfalls. Urol Clin North Am. 1996; 23: 147-63.
- Porter AT, Littrup P, Grignon D, Forman J, Montie JE: Radiotherapy and Cryotehrapy for Prostate Cancer. In: Walsh PC, Retik AB, Vaughan ED, Wein AJ (eds.). Campbell’s Urology. Philadelphia, WB Saunders Co., 7th ed., 1997 p. 2605.
- Montorsi F, Guazzoni G, Bergamaschi F, Galli L, Colombo R, Consonni P et al.: Transrectal prostatic hyperthermia and advanced prostatic cancer: Clinical results of one year follow up. Acta Urol. Ital, 1992; 6 (Suppl. 6): 471-4.
- Kleinberg L, Wallner K, Roy J, Zelefsky M, Arterbery VE, Fuks Z et al.: Treatment-related symptoms during the first year following transperineal (125)I prostate implantation. Int J Radiat Oncol Biol Phys. 1994; 28: 985-90.
- Fengler SA, Abcarian H: The York Mason approach to repair of iatrogenic rectourinary fistulae. Am J Surg. 1997; 173: 213-7.
- Chang KM, Lee RC, Ciu AW, Wang JH, Chiang H: Malakoplakia of the prostate forming a fistulous tract to rectum: a case report. Chung-Hua-I-Hsueh-Tsa-Chih-Taipei. 1996; 58: 439-43.
- Cools P, Vanderputte S, Van der Stighelen Y, Colemont L, Denis B: Rectourethral fistula due to Crohn’s disease. Acta Urol Belg. 1996; 64: 47-8.
- Felipetto R, Vigano L, Cecchi M, Florentini L, Minervini R: Use of fibrin sealant in the treatment of prostatic cutaneous fistula case of Pseudomonas prostatitis. Int Urol Nephrol. 1995; 27: 563-5.
- Blandy JP, Singh M: Fistulae involving the adult male urethra. Br J Urol. 1972; 44: 632.
- Chiou RK, Anderson JC, Tran R, Patterson RH, Wobig R, Taylor RJ et al.: Evaluation of urethral strictures and associated abnormalities using resolution and color Doppler ultrasound. Urology. 1996; 47: 102-7.
- Al-Ali M, Kashmoula D, Saoud IJ: Experience with 30 posttraumatic rectourethral fistulas: presentation of posterior transsphincteric anterior rectal wall advancement. J Urol. 1997; 158: 421-4.
- Gibbons RP: Radical Perineal Prostatectomy. In: Walsh PC, Retik AB, Vaughan ED, Wein AJ (eds.). Campbell‘s Urology. Philadelphia, WB Sanders Co., 7th ed., 1997, pp. 2589-603.
- Turner-Warwick R: The use of the omental pedicle graft in urinary reconstruction. J Urol. 1976; 116: 341.
- Ryan JA Jr, Beebe HG, Gibbons RP: Gracilis muscle flap for closure of rectourethral fistula. J Urol. 1979; 122: 1242.
- Venable DD: Modification of the anterior perineal transanorectal approach for complicated prostatic urethrorectal fistula repair. J Urol. 1989; 142: 381-4.
- Parks AG, Motson RW: Peranal repair of rectoprostatic fistula. Brit J Surg. 1983; 70: 725.
- Goodwin WE, Turner RD, Winter CD: Rectourinary fistula: principles of management and a technique of surgical closure. J Urol. 1958; 80: 246.
- Kilpatrick FR, Mason AY: Postoperative recto-prostatic fistula. Brit J Urol. 1969; 41: 649-51.
- Bukowski TP, Chakrbarty A, Powell IJ, Frontera R, Perlmutter AD, Montie JE: Acquired rectourethral fistula: Method of repair. J Urol. 1995; 153: 730.
- Fourniér R, Traxer O, Lande P, Tuech JJ, Vergos M: Posterior trans-anal-sphincter approach in the management of urethro-prostate-rectal fistula. J Urol [Paris]. 1996; 102: 75-8.
- Bardari F, D´Urso L, Leggero R, Coppola P, Muto G: Conservative treatment of iatrogenic urinary fistulas: the value of a new synthetic biological glue (GLUBRAN). Eur Urol. 2001; 39 (suppl .5): 297.
- Youssef AH, Fath Alla M, El-Kassaby AW: Perineal subcutaneous dartos pedicled flap as a new technique for repairing urethrorectal fistula. J Urol. 1999; 161: 1498-500.
- Venkatesh KS, Ramanujam P: Fibrin glue application in the treatment of recurrent anorectal fistulas. Dis Colon Rectum, 1999; 42: 1136-9.
_______________________
Received:
January 22, 2002
Accepted: February 22, 2002
_______________________
Correspondence address:
Dr. Tomáš Hanuš
Department of Urology, Charles University
Ke Karlovu 6
128 08 Prague 2
Czech Republic